Ch 12 Gases I Physical Properties A Ideal















- Slides: 21

Ch. 12 - Gases I. Physical Properties

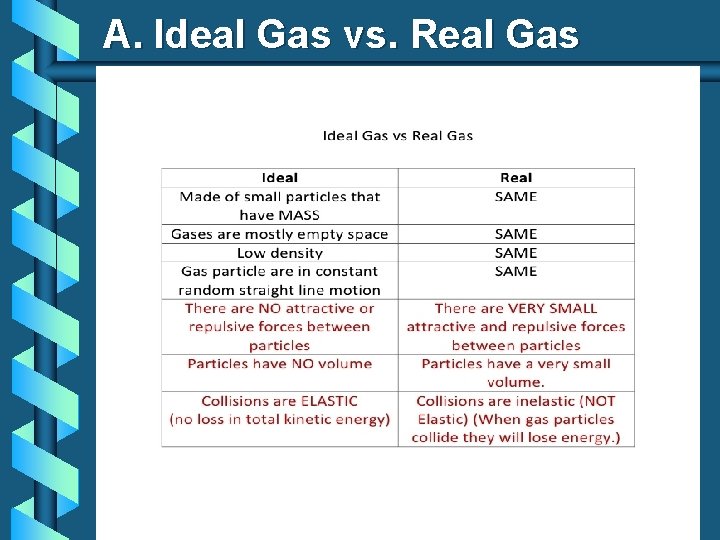
A. Ideal Gas vs. Real Gas b Kinetic Molecular Theory (KMT) • The theory that explains the behavior of gases at the molecular level. • This theory makes some assumptions about a theoretical gas called an ideal gas.

A. Ideal Gas vs. Real Gas

A. Ideal Gas vs. Real Gas behavior is most ideal… • at low pressures • at high temperatures • in nonpolar atoms/molecules Ø (see picture)

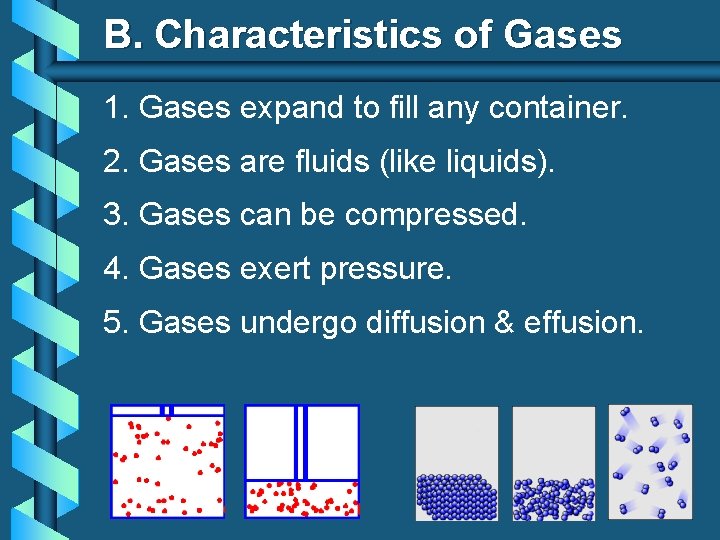
B. Characteristics of Gases 1. Gases expand to fill any container. 2. Gases are fluids (like liquids). 3. Gases can be compressed. 4. Gases exert pressure. 5. Gases undergo diffusion & effusion.

B. Characteristics of Gases b Diffusion • Spreading of gas molecules throughout a container until evenly distributed. b Effusion • Passing of gas molecules through a tiny opening in a container.

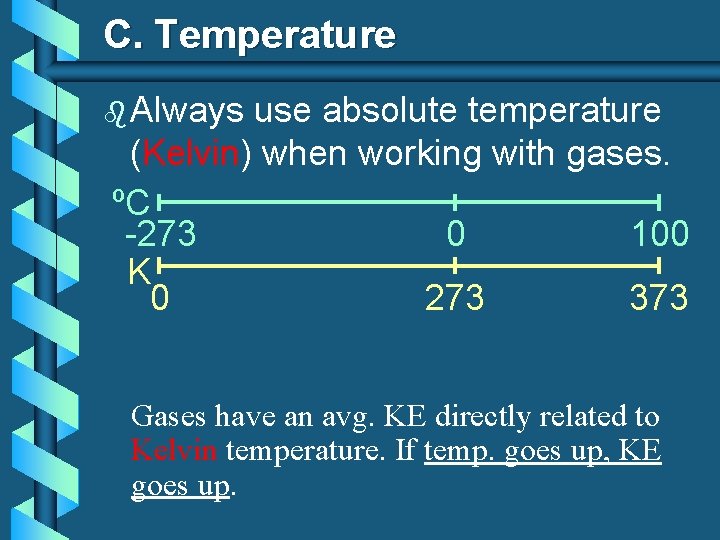
C. Temperature b Always use absolute temperature (Kelvin) when working with gases. ºC -273 0 100 K 0 273 373 Gases have an avg. KE directly related to Kelvin temperature. If temp. goes up, KE goes up.

D. Pressure SI unit for pressure is the Pascal: 1 Pa =Newton(N)/m 2 Which shoes create the most pressure?

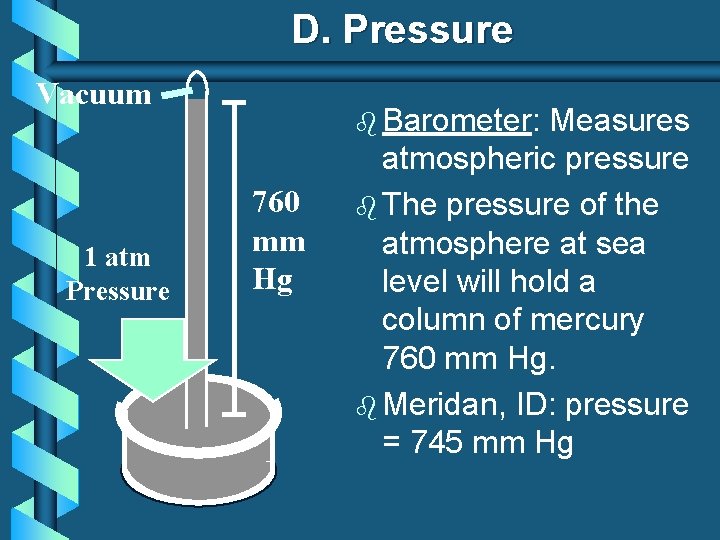
D. Pressure Vacuum 1 atm Pressure b Barometer: 760 mm Hg Measures atmospheric pressure b The pressure of the atmosphere at sea level will hold a column of mercury 760 mm Hg. b Meridan, ID: pressure = 745 mm Hg

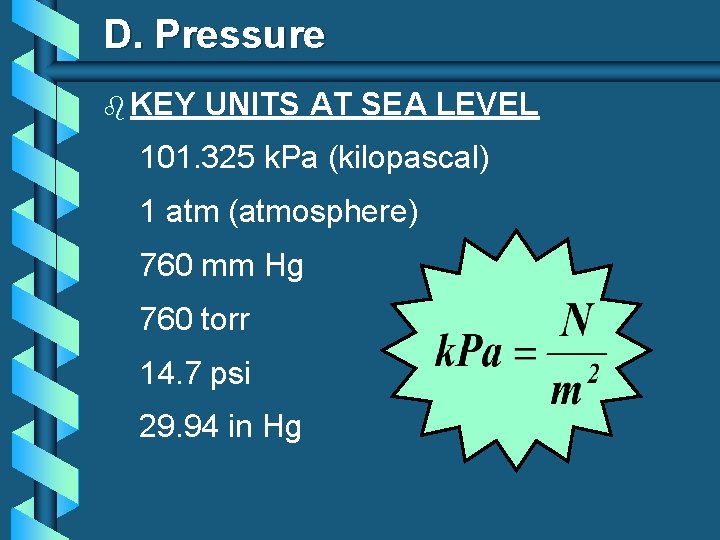
D. Pressure b KEY UNITS AT SEA LEVEL 101. 325 k. Pa (kilopascal) 1 atm (atmosphere) 760 mm Hg 760 torr 14. 7 psi 29. 94 in Hg

D. Pressure 1. The column of mercury in a barometer is 745 mm high. What is the atmospheric pressure in k. Pa? 745 mm Hg 101. 325 k. Pa 760 mm Hg = 99. 3 k. Pa

E. STP Standard Temperature & Pressure 0°C Or 273 K 1 atm Or 101. 325 k. Pa Or …

F. The Gas Laws: A series of mathematical relationships that describe the behavior of gases in regards to Pressure, Volume, & Temperature

Boyle’s Law b The pressure and volume of a gas are inversely related • at constant mass & temp Show Video PV = k P V

Charles’ Law b The volume and absolute temperature (K) of a gas are directly related • at constant mass & Show Video pressure & this V T

Gay-Lussac’s Law b The pressure and absolute temperature (K) of a gas are directly related • at constant mass & Show volume Video P T

Avogadro’s Law b Equal volumes of gases contain equal numbers of moles (n) • at constant temp & pressure • true for any gas Show video V n

Combined Gas Law P 1 V 1 = P 2 V 2 n 2 T 2 n 1 T 1 P 1 V 1 = P 2 V 2 What other equations? Play video!

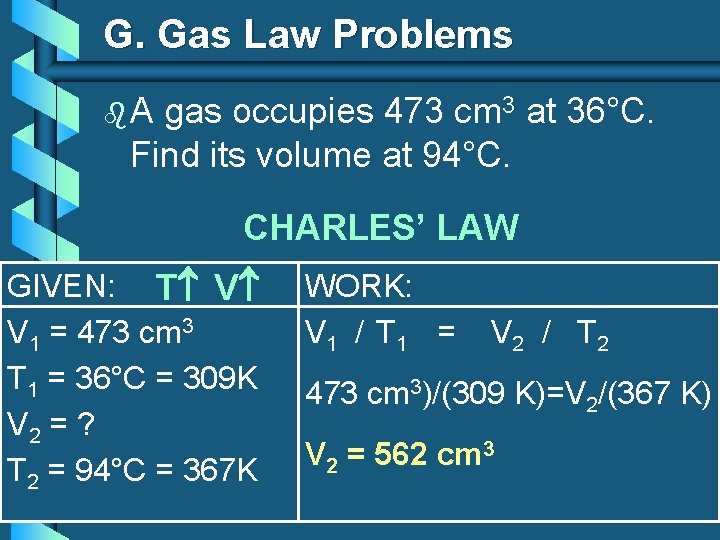
G. Gas Law Problems b. A gas occupies 473 cm 3 at 36°C. Find its volume at 94°C. CHARLES’ LAW GIVEN: T V V 1 = 473 cm 3 T 1 = 36°C = 309 K V 2 = ? T 2 = 94°C = 367 K WORK: V 1 / T 1 = V 2 / T 2 473 cm 3)/(309 K)=V 2/(367 K) V 2 = 562 cm 3

G. Gas Law Problems b. A gas occupies 100. m. L at 150. k. Pa. Find its volume at 1. 50 x 103 mm Hg. BOYLE’S LAW GIVEN: P V WORK: V 1 = 100. m. L P 1 V 1 = P 2 V 2 P 1 = 150. k. Pa (150. k. Pa)(100. m. L)=(200. k. Pa)V 2 = ? V 2 = 75. 0 m. L 3 P 2 = 1. 50 x 10 mm. Hg = 200. k. Pa

G. Gas Law Problems b. A gas occupies 7. 84 cm 3 at 71. 8 k. Pa & 25°C. Find its volume at STP. COMBINED GAS LAW GIVEN: P T V WORK: V 1 = 7. 84 cm 3 P 1 V 1 = P 2 V 2 T 1 P 1 = 71. 8 k. Pa (71. 8 k. Pa)(7. 84 cm 3)/(298 K) T 1 = 25°C = 298 K =(101. 325 k. Pa) V 2 /(273 K) V 2 = ? P 2 = 101. 325 k. Pa V 2 = 5. 09 cm 3 T 2 = 273 K