Physical and Chemical Properties Physical Properties Are properties





















- Slides: 25

Physical and Chemical Properties

Physical Properties � Are properties that do NOT change the chemical nature of matter � They are usually what we can observe with our senses. (sight, smell, hear, touch, taste) �A substance’s physical properties allow us to identify the substance without causing a change in the composition of the substance.

Lustre � How shiny or dull a material is � Example Silver has high lustre vs. Chalk is dull

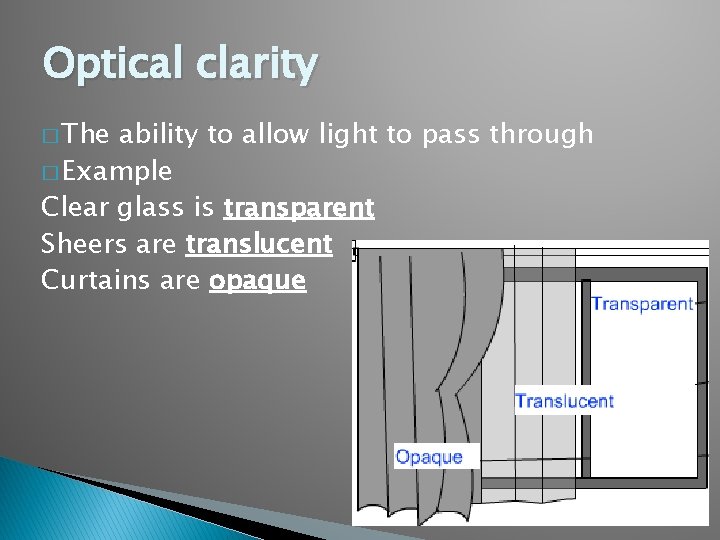
Optical clarity � The ability to allow light to pass through � Example Clear glass is transparent Sheers are translucent Curtains are opaque

Brittleness �How breakable or flexible a material is �Example Glass is brittle but modeling clay is flexible

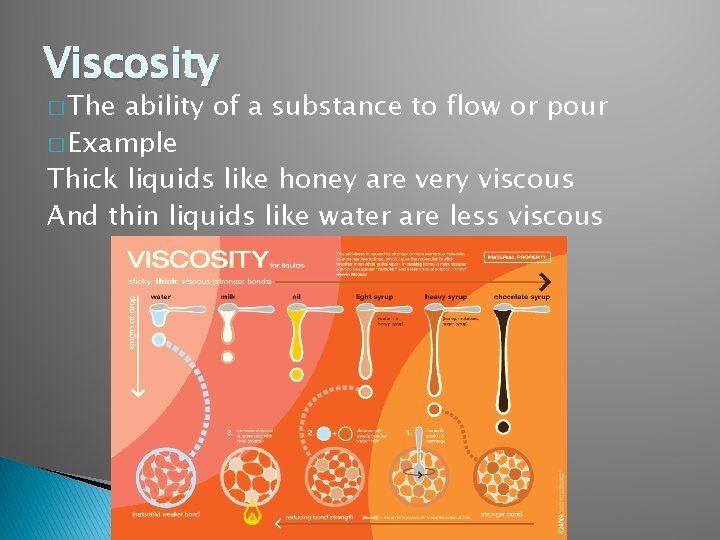
Viscosity � The ability of a substance to flow or pour � Example Thick liquids like honey are very viscous And thin liquids like water are less viscous

Hardness �A measure of the resistance of a solid to being scratched. � Example Diamond is very hard and chalk is very soft

Malleability � The ability of a substance to be hammered into a thin sheet or moulded. � Examples Aluminum foil is malleable but glass will break if hammered

Ductility � The ability of a substance to be pulled into thin strands or wires � Example Copper is very ductile

Electrical Conductivity � The ability of a substance to let an electric current pass through it � Example Copper has high conductivity but rubber has no conductivity

State of Matter � If a substance is a solid, liquid or a gas

Melting Point �The temperature a substance changes state from a solid to a liquid �Example Ice melts at 0 o. C

Boiling Point � The temperature a substance changes state from a liquid to a gas. � Example Water boils at 100 o. C

Freezing Point � The temperature a substance changes state from a liquid to a solid. � Example Water freezes at 0 o. C

Crystal form �Solids can exist in many different forms: crystals, powder, cubes and many other shapes �Examples Salt is shaped like cubes

Solubility � The ability of a substance to dissolve in a solvent � Example Sugar is soluble in water but oil is not soluble in water

Density � The amount of matter per unit volume � Example Rock is more dense than water and foam

Other physical properties �Colour �Odour �Taste

Chemical Properties � Are properties that change the chemical nature of matter. There will be a chemical reaction. � The are properties that relate to how the substance changes in composition or how they interact with other substances.

Combustibility � The ability of a substance to burn � Example Gasoline is combustible but water is not

Reaction with Acid � The ability of a substance to react with an acid � Example Copper does not react with an acid but zinc reacts quickly with acid

Reaction with Water � The ability of a substance to react with a water � Example Lithium reacts violently when placed in water, copper does not react with water

Reaction With Oxygen � The ability of a substance to react with a oxygen gas � Example Iron rusts when it reacts with oxygen in air

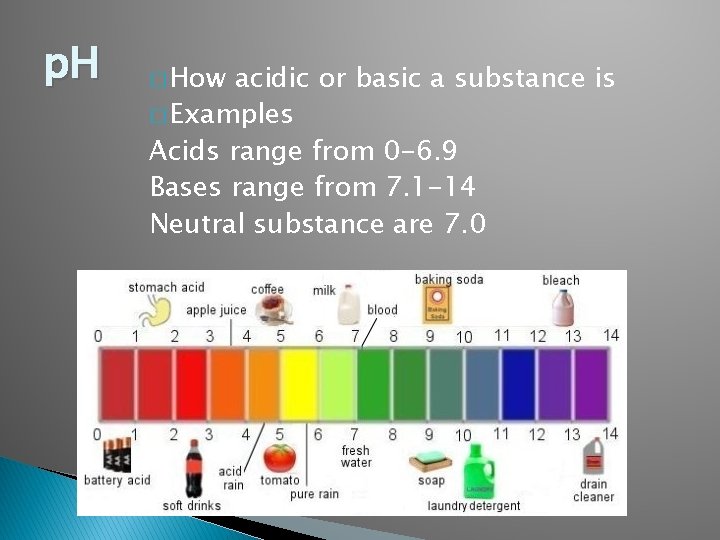
p. H � How acidic or basic a substance is � Examples Acids range from 0 -6. 9 Bases range from 7. 1 -14 Neutral substance are 7. 0

�HW: Practice Handout and page 182 #4, page 186 #2