NOBLE GASES by Mrs Shashi Jit Virdi ASSOCIATE

NOBLE GASES by : Mrs. Shashi Jit Virdi ASSOCIATE PROFESSOR Chemistry Department PGGCG, Sector 11 Chandigarh

Where are the noble gases? The elements in group 0, on the right of the periodic table, are called the noble gases. He helium Ne neon Ar argon Kr krypton Xe xenon Rn radon

NOBLE GASES ØThe group of noble gases is regarded as zero group of the periodic table. ØThese are gases at ordinary temperature and do not have chemical reactivity. ØDue to chemical inertness, these were called inter gases. ØThe elements Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe) and Radon (Rn) comprise noble gases.
Atomic World: Fast and Furious Helium 3, 000 miles per hour 1 mile/sec Air (oxygen + nitrogen 1, 000 miles per hour 7, 000, 000 collisions per second

Now-a-days, as number of compounds of these gases, particularly of xenon and krypton have been prepared, this shows that these gases are not completely inert. They are called noble gases instead of inert gases which signifies that these gases have some reactivity. Because of the low abundance of these gases on earth, they have also been called rare gases.

ØMendeleef had not left any space for the noble gases in the periodic Table ØObviously, he could not imagine the existence of elements which have almost no chemical reactivity.

Ø Ramsay, discoverer of these gases, proposed a new group for these elements on the basis of their atomic masses and lack of chemical reactivity. Ø This group is called zero group of the periodic table. The zero group also suggests chemical inertness or zero valency.

Before the discovery of noble gases, there was a sudden change from the most electronegative halogens (on right hand side) to most electropositive alkali metals (left hand side) in the periodic table. The addition of zero group between VII A group and IA group has solved this anomaly.
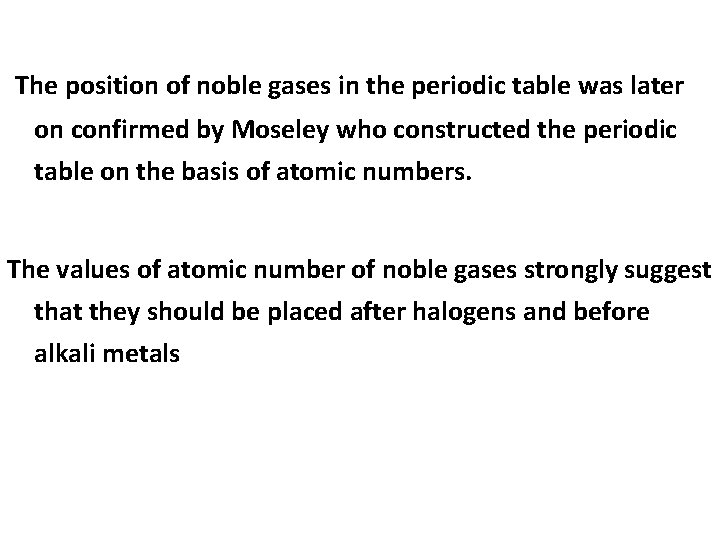
The position of noble gases in the periodic table was later on confirmed by Moseley who constructed the periodic table on the basis of atomic numbers. The values of atomic number of noble gases strongly suggest that they should be placed after halogens and before alkali metals
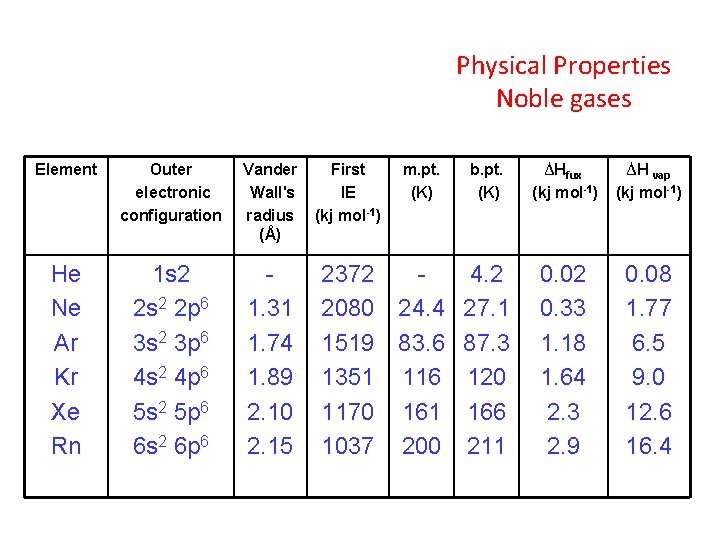
Physical Properties Noble gases Element Outer electronic configuration Vander Wall's radius (Å) He Ne Ar Kr Xe Rn 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 4 p 6 5 s 2 5 p 6 6 s 2 6 p 6 1. 31 1. 74 1. 89 2. 10 2. 15 First IE (kj mol-1) m. pt. (K) b. pt. (K) 2372 4. 2 2080 24. 4 27. 1 1519 83. 6 87. 3 1351 116 120 1170 161 166 1037 200 211 ∆Hfux (kj mol-1) ∆H vap (kj mol-1) 0. 02 0. 33 1. 18 1. 64 2. 3 2. 9 0. 08 1. 77 6. 5 9. 0 12. 6 16. 4

CHEMICAL INERTNESS OF NOBLE GASES Chemical Inertness of these gases is supported by the reasons: i) The atoms have stable completely field electronic shells ii) They have high ionisation energies iii) The noble have almost zero electron affinities. Therefore, they do not have any tendency to gain, lose or share electrons with other atoms.

Chemical properties of Nobel Gases ØThe atoms of inert gases have saturated shells, therefore they are chemically inert. ØRecent studies have shown that under certain specific condition, they enter into chemical combinations and form some rare chemical compounds. ØThe specific conditions and the types of compounds formed by these gases are disused below-

Ø Under excited condition: - Sparking Helium at low pressure in presence of mercury, tungsten etc. forms compounds like Hg. He 2, Hg. He 10, WHe 2. Ø Helium compounds are also fromed in discharge tubes like Bi. He 2, Fe. He, Pt 3 He, Pd. He. These compounds are not considered as true chemical compounds as He is absorbed on the surface.

• Compounds formed through co-ordination- Argon forms a number of unstable compound with varying no. of BF 3 molecules e. g. Ar. BF 3, Ar. 6 BF 3 • In these compounds, argon atoms donates a pair of electrons to Boron atom of BF 3. • In case of higher compounds fluorine atoms of BF 3 also donate pair of electrons.

c) Hydrates of noble gases: The hydrates of these gases are formed by compressing the gases with water e. g. , Xe. 6 H 2 O. d) Compounds formed by physical trapping (Clathrates) The inert gases Argon, Krypton and Xenon form solid compounds with certain organic molecules such as phenol and hydroquinone under pressure In such compounds the inert gas are enclosed in the crystal lattice of organic compounds known as clathrates or cage compounds.

Compounds of noble gases. Xenon forms a large no. of compounds with oxygen and fluorine in different oxidation states. These are xenon fluorides, xenon oxides and xenon oxifluorides. 1. Xe. F 2 Preparation. 1. Xenon di fluoride is best prepared by heating a mixture of 0 xenon and fluorine in molecular ratio of 2: 1 at 400 C in a sealed nickel tube. On cooling quickly, a colourless solid Xe. F 2 is obtained. Xe+F 2 Ni 4000 C Xe. F 2

Properties 1. Xenon difluoride is a colourless, crystalline solid which melts at 1290 C. 2. It reacts with hydrogen to give hydrogen fluoride and xenon. Xe. F 2 + H 2 Xe+2 HF

3. It gives substitution reactions with strong protonic acids. Xe. F 2 + HX FXe. X + HF FXe. X + HX Xe. X 2 + HF Where X= CIO-4 CF 3 COO-, SO 3 Fetc.

4. It hydrolyses slowly but completely in acidic, neutral or alkaline solutions. 2 Xe. F 2+2 H 2 O 2 Xe. F 2+4 Na. OH 2 Xe+4 HF+O 2 2 Xe+4 Na. F+O 2+2 H 2 O 5. It oxidizes iodine in the presence of BF 3 to give IF.

2. Xe. F 4 Preparation. Ø It is prepared by heating a mixture of xenon and fluorine, in a nickel vassal, at 4000 C under pressure of 5 -6 atm. Ø It can also be synthesized by passing an electric discharge through a mixture of xenon and fluorine at -78 OC. Properties of Xe. F 4 are: Ø It is a colorless, crystalline solid, with m. pt. 117. 10 c, sublimes readily. Ø Oxidized by hydrogen to HF at 300 C. Ø A stronger fluorinating agent than Xe. F 2

3. Xe. F 6 Preparation. 1. It is prepared by heating xenon with excess of fluorine (in the molar ratio of 1: 20) in a nickel vessel at 250 -3000 C under pressure of 50 -60 atm. Xe + 3 F 2 Xe. F 6 2. It can also be obtained by the oxidation of Xe. F 4 with O 2 F 2 under pressure. Xe. F 4 + O 2 F 2 -1300 c Xe. F 6 + O 2

Properties: - Crystalline substance, m. pt. 49. 50 C, Mostly volatile, all the fluorides of xenon are greenish yellow colour vapour. It is extremely reactive. Therefore, it cannot be stored in glass or quartz vessels because of the following reactions which finally give the dangerously explosive xenon trioxide. 2 Xe. F 6+Si. O 2 2 Xe. OF 4+Si. O 2 2 Xe. O 2 F 2+Si. O 2 2 Xe. OF 4 +Si. F 4 2 Xe. O 2 F 2+Si. F 4 2 Xe. O 3+Si. F 4 (explosive)
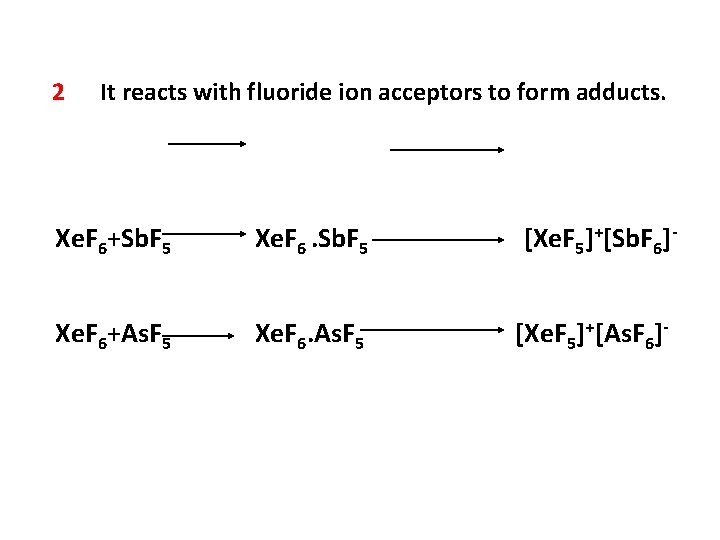
2 It reacts with fluoride ion acceptors to form adducts. Xe. F 6+Sb. F 5 Xe. F 6. Sb. F 5 [Xe. F 5]+[Sb. F 6]- Xe. F 6+As. F 5 Xe. F 6. As. F 5 [Xe. F 5]+[As. F 6]-

4. Xe. OF 4 Preparation : (i) Xenon Oxytetraflouride is prepared by partial hydrolysis of Xenon hexa flouride Xe. F 6 + H 2 O Xe. OF 4 + 2 HF (ii ) by the action of Xe. F 6 on silicon dioxide 2 Xe. F 6 + Si. O 2 Xe. OF 4 + Si. F 4
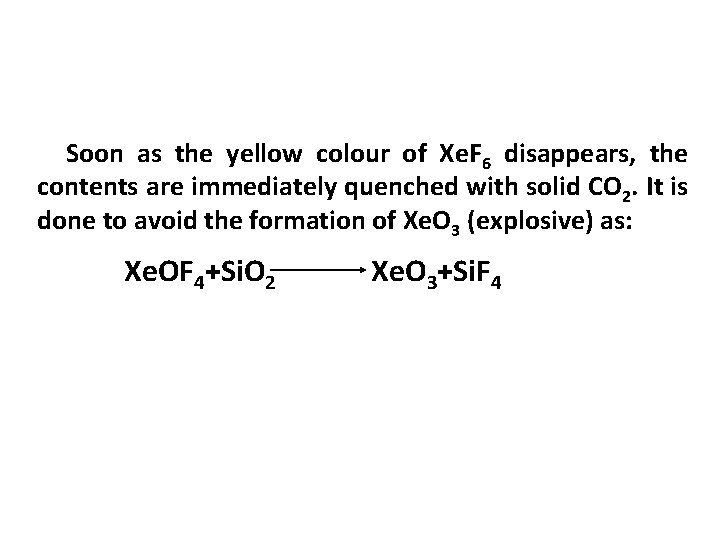
Soon as the yellow colour of Xe. F 6 disappears, the contents are immediately quenched with solid CO 2. It is done to avoid the formation of Xe. O 3 (explosive) as: Xe. OF 4+Si. O 2 Xe. O 3+Si. F 4

Properties. 1. It is a colourless compound melting at -46 o. C. 2 It is reduced by hydrogen to xenon. Xe. OF 4+3 H 2 Xe+H 2 O+4 HF 3 It reacts with water or silica to form another oxyfluoride, Xe. O 2 F 2, in ; which xenon remains in the same oxidation state. Further reaction gives explosive compound Xe. O 3
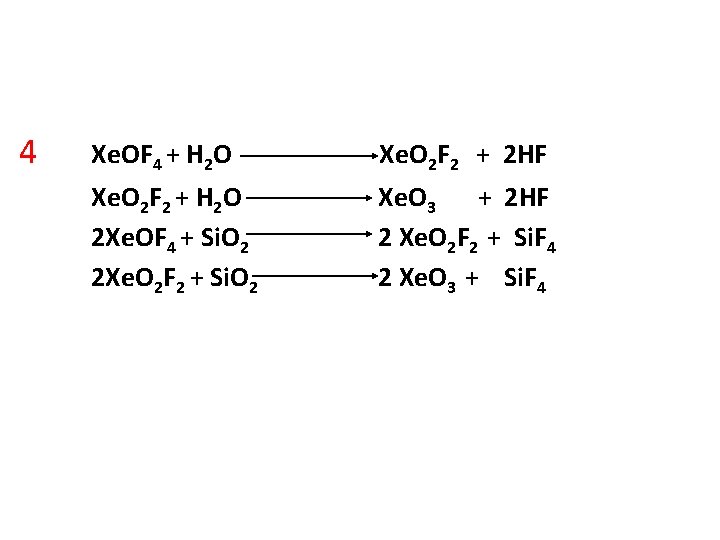
4 Xe. OF 4 + H 2 O Xe. O 2 F 2 + 2 HF Xe. O 2 F 2 + H 2 O 2 Xe. OF 4 + Si. O 2 2 Xe. O 2 F 2 + Si. O 2 Xe. O 3 + 2 HF 2 Xe. O 2 F 2 + Si. F 4 2 Xe. O 3 + Si. F 4

5. Xe. O 2 F 2 Preparation ; 1. by mixing Xe. O 3 and Xe. OF 4 at temperature close to 78 OC. Xe. O 3+Xe. OF 4 2 Xe. O 2 F 2 The compound is purified by fractional distillation. 2. It is also formed when Xe. OF 4 is hydrolyzed or reacted with silica. 2 Xe. OF 4+Si. O 2 Xe. OF 4+H 2 O 2 Xe. O 2 F 2+Si. F 4 Xe. O 2 F 2+ 2 HF

Properties. 1. It is a colourless solid. 2. Its melting point is 30. 8 OC. 3. It is easily hydrolyzed to give xenon trioxide. Xe. O 2 F 2 + H 2 O Xe. O 3 + 2 HF.

6. Xe. O 3 Preparation. Xenon trioxide is prepared by the hydrolysis of Xe. F 6 or Xe. F 4 6 Xe. F 4 + 12 H 2 O 2 Xe. O 3 + 4 Xe +24 HF+3 O 2 Xe. F 6 + 3 H 2 O Xe. O 3 + 6 HF It acts as a powerful oxidizing agent in acidic medium. For instance, it oxidizes Pu 3+ to Pu 4+ in the presence of H+ ions. 6 Pu+3 + Xe. O 3 + 6 H+ 6 Pu +4 + Xe + 3 H 2 O

Preparation : - 7. Xe O 4 It is prepared by action of conc. H 2 SO 4 on sodium or barium xenate (Na 4 Xe. O 6 or Ba 2 Xe O 6) at room temp. Na 4 Xe. O 6+ 2 H 2 SO 4 Xe. O 4 + 2 Na 2 SO 4 +2 H 2 O Properties: It is very unstable and decomposes to xenon and oxygen.
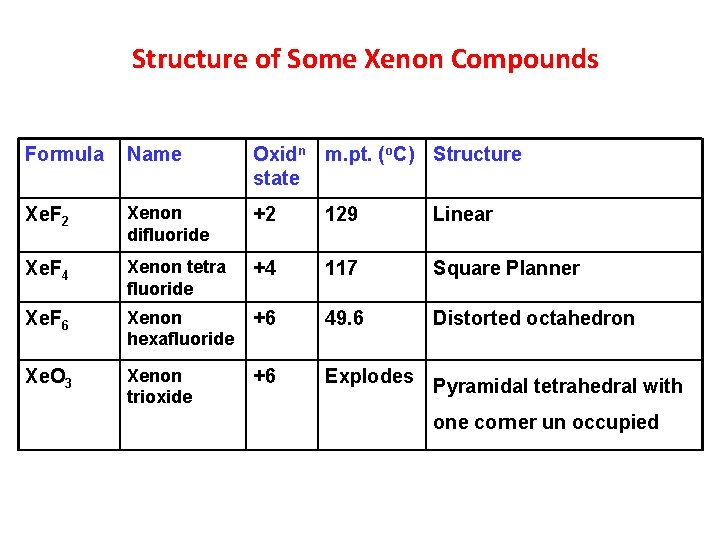
Structure of Some Xenon Compounds Formula Name Oxidn m. pt. (o. C) Structure state Xe. F 2 Xenon difluoride +2 129 Linear Xe. F 4 Xenon tetra fluoride +4 117 Square Planner Xe. F 6 Xenon hexafluoride +6 49. 6 Distorted octahedron Xe. O 3 Xenon trioxide +6 Explodes Pyramidal tetrahedral with one corner un occupied
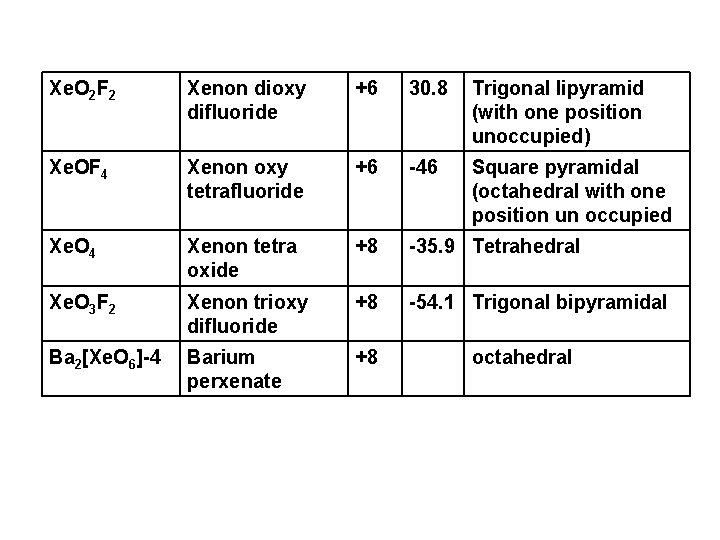
Xe. O 2 F 2 Xenon dioxy difluoride +6 30. 8 Trigonal lipyramid (with one position unoccupied) Xe. OF 4 Xenon oxy tetrafluoride +6 -46 Square pyramidal (octahedral with one position un occupied Xe. O 4 Xenon tetra oxide +8 -35. 9 Tetrahedral Xe. O 3 F 2 Xenon trioxy difluoride +8 -54. 1 Trigonal bipyramidal Ba 2[Xe. O 6]-4 Barium perxenate +8 octahedral

Structure and Bonding in Xenon Compounds Formula Structure No. of VSEPR of e lone (Explanation of structure) pairs Xe. F 2 Linear 5 3 Five electron pairs form trigonal bipyramidal with three lone pairs at equatorial positions Xe. F 4 Square Planner 6 2 Six electron pairs form octahedron with two positions occupied by lone pairs Xe. F 6 Distorted 7 octahedron 1 Pentagonal bipyramidal or octahedron with one lone pair Xe. O 3 Pyramidal 1 Three π bonds so that the remaining four electron pairs form a tetrahedron with one corner occupied by a lone pairs. 7 capped
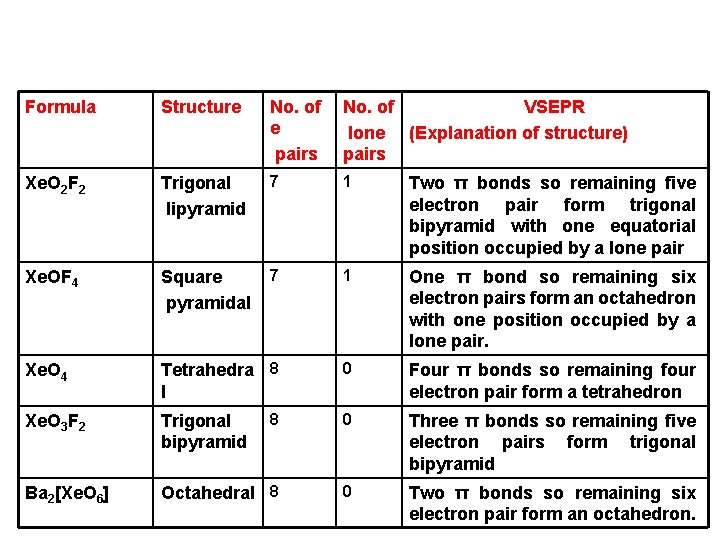
Formula Structure No. of e pairs No. of VSEPR lone (Explanation of structure) pairs Xe. O 2 F 2 Trigonal lipyramid 7 1 Two π bonds so remaining five electron pair form trigonal bipyramid with one equatorial position occupied by a lone pair Xe. OF 4 Square pyramidal 7 1 One π bond so remaining six electron pairs form an octahedron with one position occupied by a lone pair. Xe. O 4 Tetrahedra 8 l 0 Four π bonds so remaining four electron pair form a tetrahedron Xe. O 3 F 2 Trigonal bipyramid 8 0 Three π bonds so remaining five electron pairs form trigonal bipyramid Ba 2[Xe. O 6] Octahedral 8 0 Two π bonds so remaining six electron pair form an octahedron.
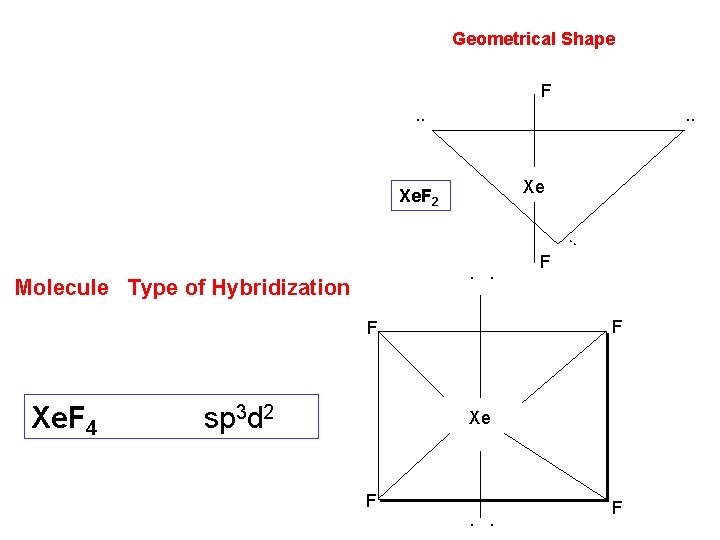
Geometrical Shape F. . Xe Xe. F 2 . . Molecule Type of Hybridization F F Xe. F 4 sp 3 d 2 F Xe F . . F
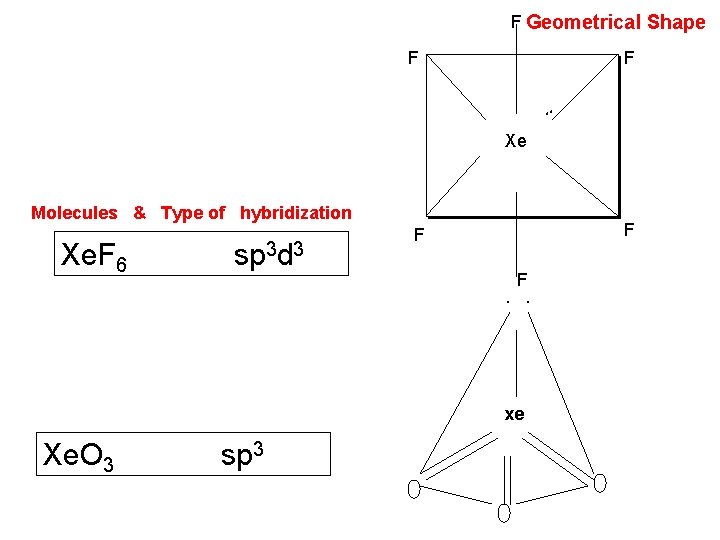
F Geometrical Shape F F . . Xe Molecules & Type of hybridization Xe. F 6 sp 3 d 3 F F F. . xe Xe. O 3 sp 3
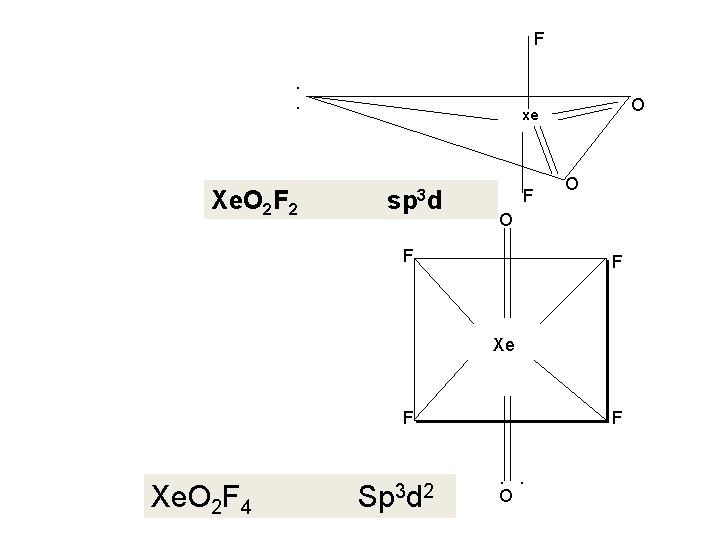
F. . Xe. O 2 F 2 O xe sp 3 d F O O F F Xe. OF Xe. O 2 F 44 3 d 3 d 22 sp Sp F F. . O
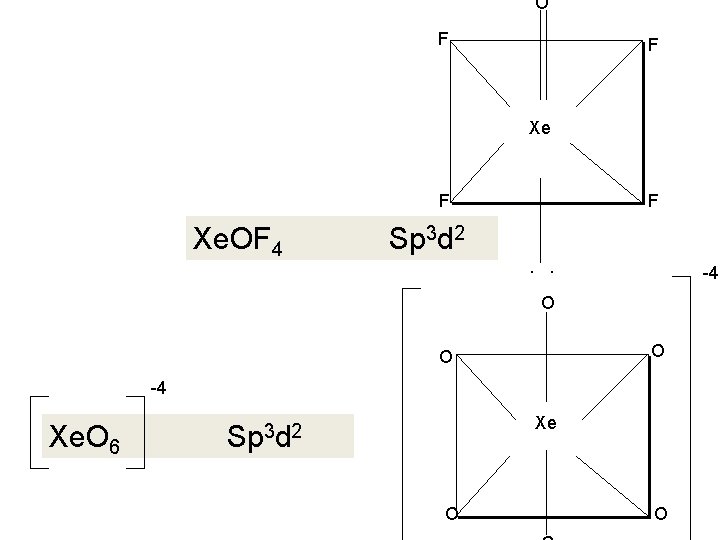
O F F Xe. OF 4 F Sp 3 d 2. . -4 O O O -4 Xe. O 6 Xe Sp 3 d 2 O O

Significance of Noble gases in development of theoretical chemistry. 1. In Elucidation of distribution of electrons in atom 2. In periodic classification 3. In the development of electronic theory of valency 4. In radioactivity.

Thank you
- Slides: 41