The Behavior of Gases Compressibility n Gases can

“The Behavior of Gases”

Compressibility n Gases can expand to fill its container, unlike solids or liquids n The reverse is also true: n They are easily compressed, or squeezed into a smaller volume n Compressibility is a measure of how much the volume of matter decreases under pressure

Compressibility n This is the idea behind placing “air bags” in automobiles n In an accident, the air compresses more than the steering wheel or dash when you strike it n The impact forces the gas particles closer together, because there is a lot of empty space

Compressibility n At room temperature, the distance between particles is about 10 x the diameter of the particle n Fig. 14. 2, page 414 n This empty space makes gases good insulators (example: windows, coats) n How does the volume of the particles in a gas compare to the overall volume of the gas?

Variables that describe a Gas n The four variables and their common units: 1. pressure (P) in kilopascals 2. volume (V) in Liters 3. temperature (T) in Kelvin 4. amount (n) in moles • The amount of gas, volume, and temperature are factors that affect gas pressure.

1. Amount of Gas n When we inflate a balloon, we are adding gas molecules. n Increasing the number of gas particles increases the number of collisions nthus, the pressure increases n If temperature is constant, then doubling the number of particles doubles the pressure

Pressure and the number of molecules are directly related n More molecules means more collisions, and… n Fewer molecules means fewer collisions. n Gases naturally move from areas of high pressure to low pressure, because there is empty space to move into – a spray can is example.

Common use? n A practical application is Aerosol (spray) cans n gas moves from higher pressure to lower pressure n a propellant forces the product out n whipped cream, hair spray, paint n Fig. 14. 5, page 416 n Is the can really ever “empty”?

2. Volume of Gas n In a smaller container, the molecules have less room to move. n The particles hit the sides of the container more often. n As volume decreases, pressure increases. (think of a syringe) n Thus, volume and pressure are inversely related to each other

3. Temperature of Gas Raising the temperature of a gas increases the pressure, if the volume is held constant. (Temp. and Pres. are directly related) n The molecules hit the walls harder, and more frequently! n Fig. 14. 7, page 417 n Should you throw an aerosol can into a fire? What could happen? n When should your automobile tire pressure be checked? n


The Gas Laws n OBJECTIVES: n. Describe the relationships among the temperature, pressure, and volume of a gas.

The Gas Laws n OBJECTIVES: n. Use the combined gas law to solve problems.

The Gas Laws are mathematical n The gas laws will describe HOW gases behave. n Gas behavior can be predicted by theory. n The amount of change can be calculated with mathematical equations. n You need to know both of these: theory, and the math

Robert Boyle (1627 -1691) • Boyle was born into an aristocratic Irish family • Became interested in medicine and the new science of Galileo and studied chemistry. • A founder and an influential fellow of the Royal Society of London • Wrote extensively on science, philosophy, and theology.

#1. Boyle’s Law - 1662 Gas pressure is inversely proportional to the volume, when temperature is held constant. Pressure x Volume = a constant Equation: P 1 V 1 = P 2 V 2 (T = constant)
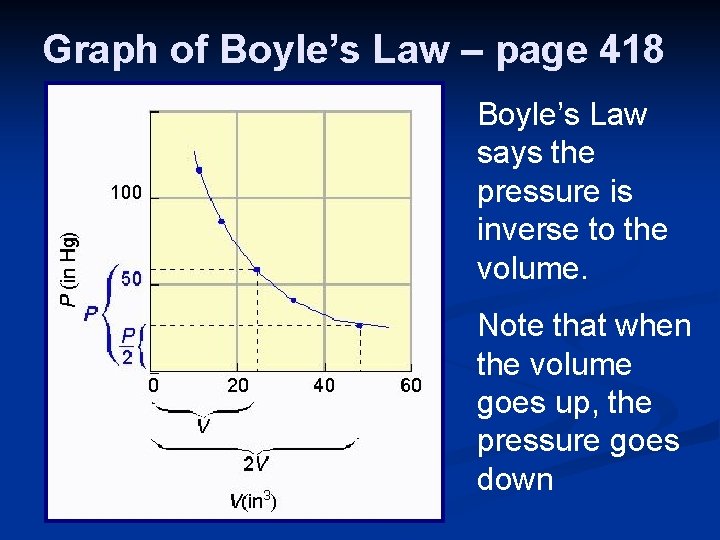
Graph of Boyle’s Law – page 418 Boyle’s Law says the pressure is inverse to the volume. Note that when the volume goes up, the pressure goes down

Jacques Charles (1746 -1823) • French Physicist • Part of a scientific balloon flight on Dec. 1, 1783 – was one of three passengers in the second balloon ascension that carried humans • This is how his interest in gases started • It was a hydrogen filled balloon – good thing they were careful!

#2. Charles’s Law - 1787 The volume of a fixed mass of gas is directly proportional to the Kelvin temperature, when pressure is held constant. This extrapolates to zero volume at a temperature of zero Kelvin.

Converting Celsius to Kelvin • Gas law problems involving temperature will always require that the temperature be in Kelvin. (Remember that no degree sign is shown with the kelvin scale. ) • Reason? There will never be a zero volume, since we have never reached absolute zero. Kelvin = C + 273 and °C = Kelvin - 273

Joseph Louis Gay-Lussac (1778 – 1850) French chemist and physicist v Known for his studies on the physical properties of gases. v In 1804 he made balloon ascensions to study magnetic forces and to observe the composition and temperature of the air at different altitudes. v

#3. Gay-Lussac’s Law - 1802 • The pressure and Kelvin temperature of a gas are directly proportional, provided that the volume remains constant. • How does a pressure cooker affect the time needed to cook food? (Note page 422) • Sample Problem 14. 3, page 423

#4. The Combined Gas Law The combined gas law expresses the relationship between pressure, volume and temperature of a fixed amount of gas. Sample Problem 14. 4, page 424

The combined gas law contains all the other gas laws! n If the temperature remains constant. . . n P 1 x V 1 T 1 = P 2 x V 2 T 2 Boyle’s Law

The combined gas law contains all the other gas laws! n If the pressure remains constant. . . n P 1 x V 1 T 1 = P 2 x V 2 T 2 Charles’s Law

u. The combined gas law contains all the other gas laws! u. If the volume remains constant. . . P 1 x V 1 T 1 = P 2 x V 2 T 2 Gay-Lussac’s Law

Ideal Gases n OBJECTIVES: n. Compute the value of an unknown using the ideal gas law.

Section 14. 3 Ideal Gases n OBJECTIVES: n. Compare and contrast real an ideal gases.

5. The Ideal Gas Law #1 Equation: P x V = n x R x T n Pressure times Volume equals the number of moles (n) times the Ideal Gas Constant (R) times the Temperature in Kelvin. n n. R = 8. 31 (L x k. Pa) / (mol x K) The other units must match the value of the constant, in order to cancel out. n The value of R could change, if other units of measurement are used for the other values (namely pressure changes) n

The Ideal Gas Law n We now have a new way to count moles (the amount of matter), by measuring T, P, and V. We aren’t restricted to only STP conditions: P x V n = R x T

Ideal Gases We are going to assume the gases behave “ideally”- in other words, they obey the Gas Laws under all conditions of temperature and pressure n An ideal gas does not really exist, but it makes the math easier and is a close approximation. n Particles have no volume? Wrong! n No attractive forces? Wrong! n

Ideal Gases n There are no gases for which this is true (acting “ideal”); however, n Real gases behave this way at a) high temperature, and b) low pressure. n. Because at these conditions, a gas will stay a gas! n. Sample Problem 14. 5, page

#6. Ideal Gas Law 2 P x V = m x R x T M n Allows LOTS of calculations, and some new items are: n m = mass, in grams n M = molar mass, in g/mol n n Molar mass = m R T P V

Density n Density is mass divided by volume m D= V so, m M P D= = V R T


Ideal Gases don’t exist, because: 1. Molecules do take up space 2. There attractive forces between particles - otherwise there would be no liquids formed

Real Gases behave like Ideal Gases. . . When the molecules are far apart. n The molecules do not take up as big a percentage of the space n n We can ignore the particle volume. n This is at low pressure

Real Gases behave like Ideal Gases… n When molecules are moving fast n. This is at high temperature n Collisions are harder and faster. n Molecules are not next to each other very long. n Attractive forces can’t play a role.

Section 14. 4 Gases: Mixtures and Movements n OBJECTIVES: n. Relate the total pressure of a mixture of gases to the partial pressures of the component gases.

Section 14. 4 Gases: Mixtures and Movements n OBJECTIVES: n. Explain how the molar mass of a gas affects the rate at which the gas diffuses and effuses.

#7 Dalton’s Law of Partial Pressures For a mixture of gases in a container, PTotal = P 1 + P 2 + P 3 +. . . • P 1 represents the “partial pressure”, or the contribution by that gas. • Dalton’s Law is particularly useful in calculating the pressure of gases collected over water.
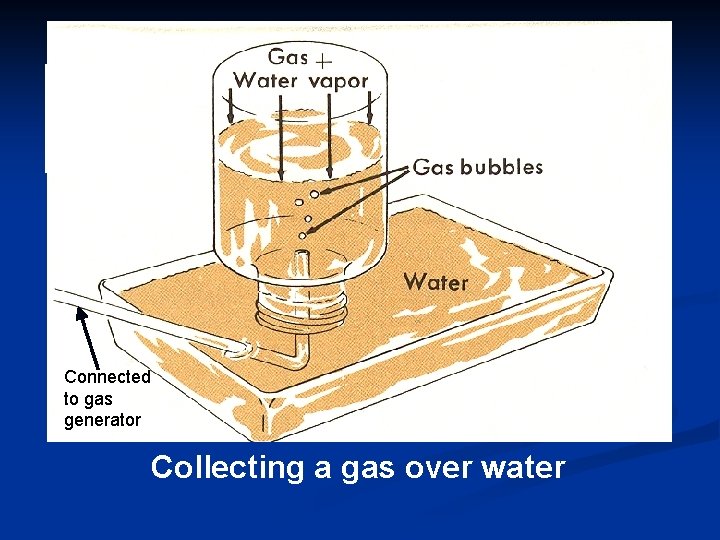
Connected to gas generator Collecting a gas over water
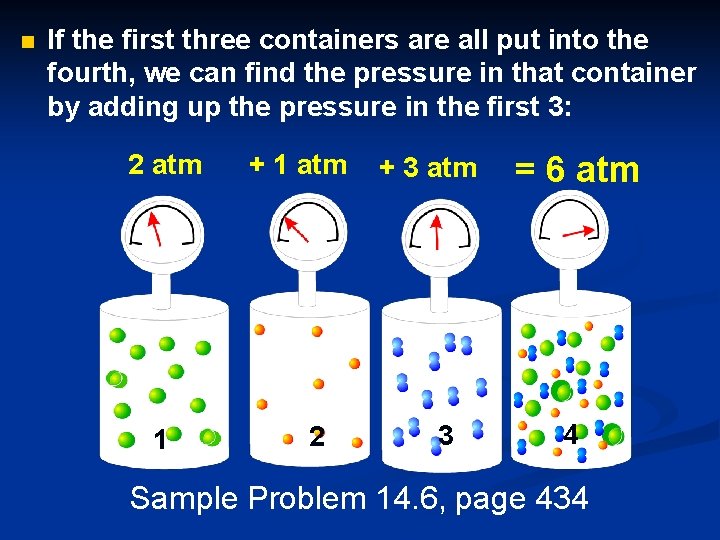
n If the first three containers are all put into the fourth, we can find the pressure in that container by adding up the pressure in the first 3: 2 atm 1 + 1 atm 2 + 3 atm 3 = 6 atm 4 Sample Problem 14. 6, page 434

Diffusion is: u. Molecules moving from areas of high concentration to low concentration. u. Example: perfume molecules spreading across the room. n Effusion: Gas escaping through a tiny hole in a container. n Both of these depend on the molar mass of the particle, which determines the speed.

• Diffusion: describes the mixing of gases. The rate of diffusion is the rate of gas mixing. • Molecules move from areas of high concentration to low concentration. • Fig. 14. 18, p. 435

Effusion: a gas escapes through a tiny hole in its container -Think of a nail in your car tire… Diffusion and effusion are explained by the next gas law: Graham’s

8. Graham’s Law Rate. A Rate. B = Mass. B Mass. A The rate of effusion and diffusion is inversely proportional to the square root of the molar mass of the molecules. n Derived from: Kinetic energy = 1/2 mv 2 n m = the molar mass, and v = the velocity. n

Graham’s Law Sample: compare rates of effusion of Helium with Nitrogen – done on p. 436 n With effusion and diffusion, the type of particle is important: n n n Gases of lower molar mass diffuse and effuse faster than gases of higher molar mass. Helium effuses and diffuses faster than nitrogen – thus, helium escapes from a balloon quicker than many other gases!
- Slides: 48