Chemistry 10 1 10 1 The Mole A








































- Slides: 43

Chemistry 10. 1

10. 1 The Mole: A Measurement of Matter u You could measure the amount of sand in a sand sculpture by counting each grain of sand, but it would be much easier to weigh the sand. You’ll discover how chemists measure the amount of a substance using a unit called a mole, which relates the number of particles to the mass.

10. 1 Measuring Matter u Measuring Matter – What are three methods for measuring the amount of something?

10. 1 Measuring Matter u You often measure the amount of something by one of three different methods—by count, by mass, and by volume.

10. 1

10. 1

10. 1

10. 1

for Sample Problem 10. 1 Problem Solving 10. 1 Solve Problem 1 with the help of an interactive guided tutorial.

Sample problem 10. 1 u u u We are solving mass of 0. 50 bushels of apples We know: 0. 20 bushels = 1 dozen apples = 2. 0 kg u Answer: u u u 0. 50 bushel x 1 dozen__ 0. 20 bushel = 5. 0 kg x 2. 0 kg__ 1 dozen

10. 1 u What is a mole?

10. 1 What is a Mole? u What Is a Mole? – How is Avogadro’s number related to a mole of any substance?

10. 1 What is a Mole? u. A mole of any substance contains Avogadro’s number of representative particles, or 6. 02 1023 representative particles. –The term representative particle refers to the species present in a substance: usually atoms, molecules, or formula units.

10. 1 What is a Mole? – Converting Number of Particles to Moles u. One mole (mol) of a substance is 6. 02 1023 representative particles of that substance and is the SI unit for measuring the amount of a substance. u. The number of representative particles in a mole, 6. 02 1023, is called Avogadro’s number.

10. 1 What is a Mole?

10. 2

10. 2

10. 2

10. 2

for Sample Problem 10. 2 Problem Solving 10. 4 Solve Problem 4 with the help of an interactive guided tutorial.

10. 1 What is a Mole? – Converting Moles to Number of Particles

Sample problem 10. 2 u? u Moles is 2. 17 x 1023 particles of Br We know 1 mole = 6. 02 x 1023 u Answer: u 2. 17 x 1023 X 1 mole = 0. 360 mol Br 6. 02 x 1023

10. 3

10. 3

10. 3

10. 3

for Sample Problem 10. 3 Problem Solving 10. 5 Solve Problem 5 with the help of an interactive guided tutorial.

Sample problem 10. 3 u u u u We We We are looking for the # of atoms in 1. 14 mol SO 3 know there are 4 molecules in SO 3 know 1 mole of SO 3 is 6. 02 x 1023 moles Answer: 1. 14 mol X 6. 02 x 1023 mol X 4 atoms molecule = 2. 75 x 1024

The Mass of a Mole of an Element 10. 1 u The Mass of a Mole of an Element – How is the atomic mass of an element related to the molar mass of an element?

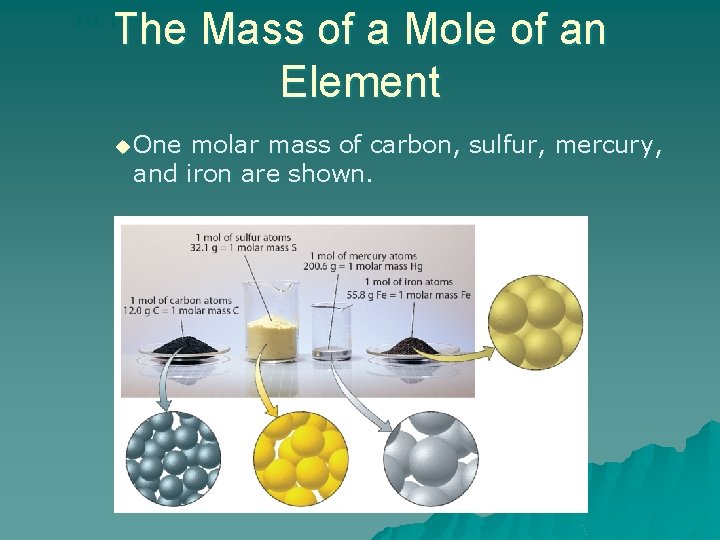
10. 1 The Mass of a Mole of an Element u. The atomic mass of an element expressed in grams is the mass of a mole of the element. –The mass of a mole of an element is its molar mass.

The Mass of a Mole of an Element 10. 1 u One molar mass of carbon, sulfur, mercury, and iron are shown.

Animation 11 – Find out how Avogadro’s number is based on the relationship between the amu and the gram.

The Mass of a Mole of an Element 10. 1

10. 1 The Mass of a Mole of a Compound u The Mass of a Mole of a Compound –How is the mass of a mole of a compound calculated?

10. 1 The Mass of a Mole of a Compound – To calculate the molar mass of a compound, find the number of grams of each element in one mole of the compound. Then add the masses of the elements in the compound.

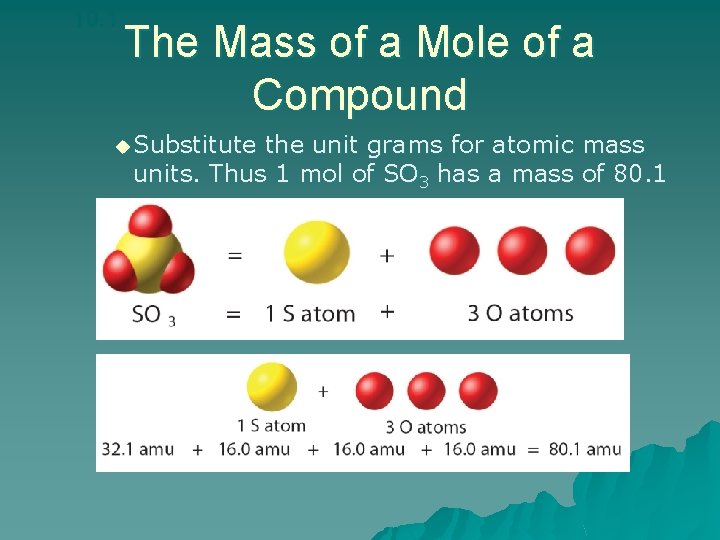
10. 1 The Mass of a Mole of a Compound u Substitute the unit grams for atomic mass units. Thus 1 mol of SO 3 has a mass of 80. 1 g.

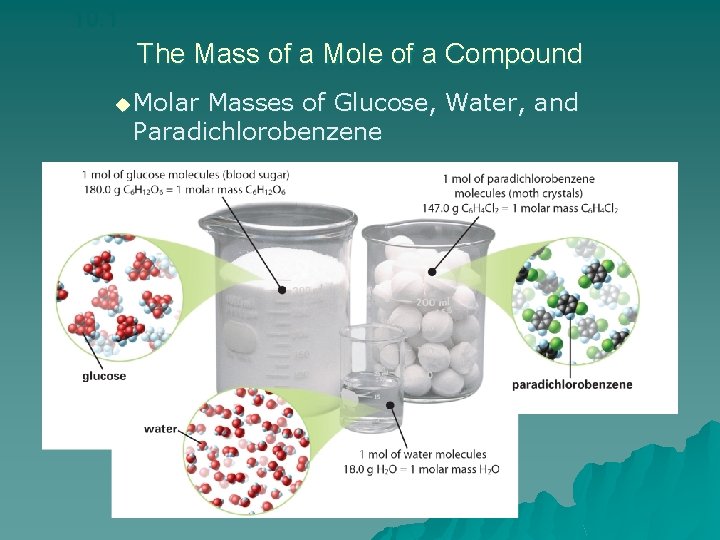
10. 1 The Mass of a Mole of a Compound u Molar Masses of Glucose, Water, and Paradichlorobenzene

10. 4

10. 4

10. 4

10. 4

for Sample Problem 10. 4 Problem Solving 10. 7 Solve Problem 7 with the help of an interactive guided tutorial.

END OF SHOW