THE MOLE CONTENTS What is a mole and














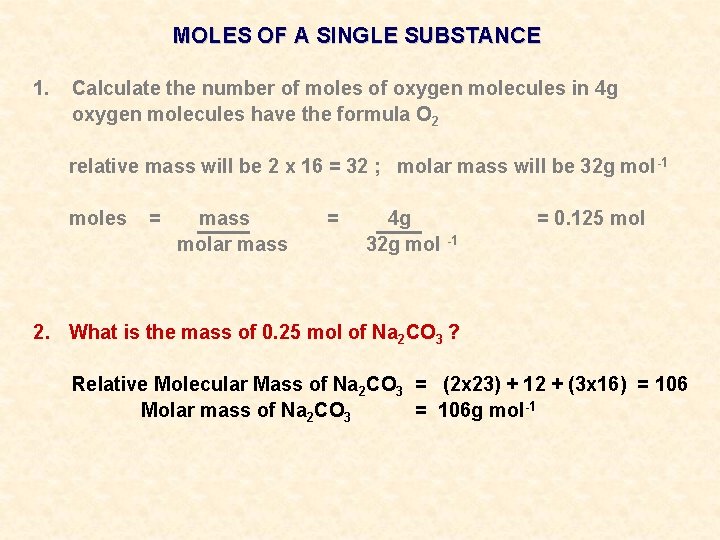
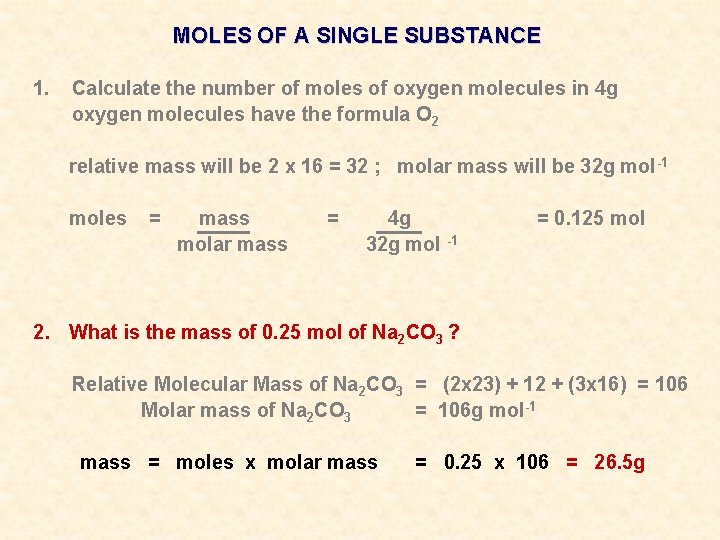
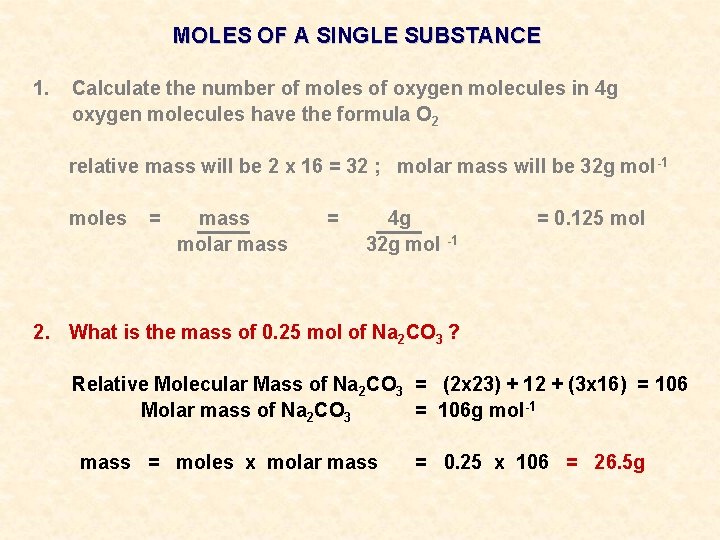
- Slides: 18

THE MOLE CONTENTS • What is a mole and why do we use it? • Calculating the number of moles of a single substance • Reacting mass calculations

THE MOLE Before you start it would be helpful to… • know how to balance simple equations • know how to re-arrange mathematical formulae DON’T BE LEFT IN THE DARK!

THE MOLE WHAT IS A MOLE ? it is the standard unit of amount of a substance it is just a number, a very big number it is a way of saying a number in words, just like. . . DOZEN for 12 SCORE for 20 GROSS for 144

THE MOLE WHAT IS A MOLE ? it is the standard unit of amount of a substance it is just a number, a very big number it is a way of saying a number in words, just like. . . DOZEN for 12 SCORE for 20 GROSS for 144 HOW BIG IS IT ? 60220000000000 (Approximately). . . THAT’S It is a lot easier to write it as. . . BIG !!! 6. 022 x 1023

THE MOLE WHAT IS A MOLE ? it is the standard unit of amount of a substance it is just a number, a very big number it is a way of saying a number in words, just like. . . DOZEN for 12 SCORE for 20 GROSS for 144 HOW BIG IS IT ? 60220000000000 (Approximately). . . THAT’S It is a lot easier to write it as. . . It is also known as. . . BIG !!! 6. 022 x 1023 AVOGADRO’S NUMBER It doesn’t matter what the number is as long as everybody sticks to the same value !

THE MOLE WHY USE IT ? Atoms and molecules don’t weigh much so it is easier to count large numbers of them. In fact it is easier to weigh substances. Using moles tells you. . . how many particles you get in a certain mass the mass of a certain number of particles DO I NEED TO KNOW ANYTHING ELSE ? Yes, it would help if you can balance equations AND Keep trying, you will get the idea. . . EVENTUALLY!

THE MOLE – AN OVERVIEW WHAT IS IT? The standard unit of amount of a substance just as the standard unit of length is a METRE It is just a number, a very big number It is also a way of saying a number in words like DOZEN for 12 GROSS for 144

THE MOLE – AN OVERVIEW WHAT IS IT? The standard unit of amount of a substance just as the standard unit of length is a METRE It is just a number, a very big number It is also a way of saying a number in words like DOZEN for 12 GROSS for 144 HOW BIG IS IT ? 60220000000000 (approx) - THAT’S BIG !!! It is a lot easier to write it as 6. 022 x 1023 And anyway it doesn’t matter what the number is as long as everybody sticks to the same value !

THE MOLE – AN OVERVIEW WHAT IS IT? The standard unit of amount of a substance just as the standard unit of length is a METRE It is just a number, a very big number It is also a way of saying a number in words like DOZEN for 12 GROSS for 144 HOW BIG IS IT ? 60220000000000 (approx) - THAT’S BIG !!! It is a lot easier to write it as 6. 022 x 1023 And anyway it doesn’t matter what the number is as long as everybody sticks to the same value ! WHY USE IT ? Atoms and molecules don’t weigh much so it is easier to count large numbers of them. In fact it is easier to weigh substances. Using moles tells you : - how many particles you get in a certain mass the mass of a certain number of particles

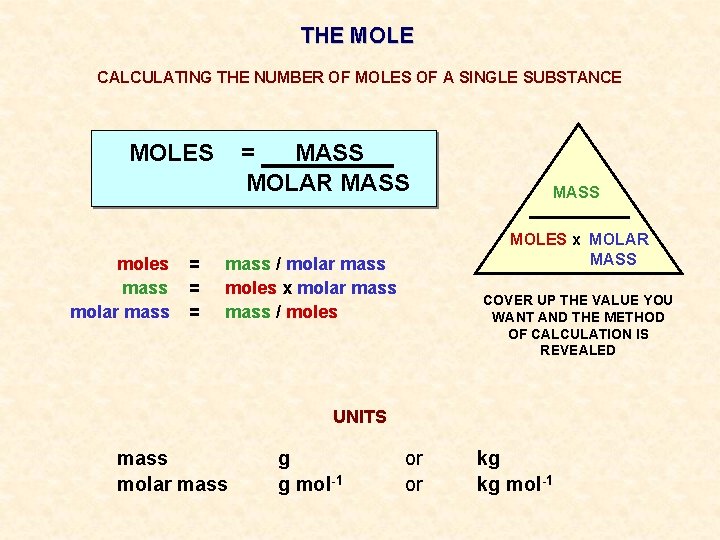
THE MOLE CALCULATING THE NUMBER OF MOLES OF A SINGLE SUBSTANCE MOLES moles mass molar mass = = MASS MOLAR MASS MOLES x MOLAR MASS mass / molar mass moles x molar mass / moles COVER UP THE VALUE YOU WANT AND THE METHOD OF CALCULATION IS REVEALED UNITS mass molar mass g g mol-1 or or kg kg mol-1

MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g

MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1

MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 = 0. 125 mol

MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 = 0. 125 mol

MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 2. What is the mass of 0. 25 mol of Na 2 CO 3 ? = 0. 125 mol
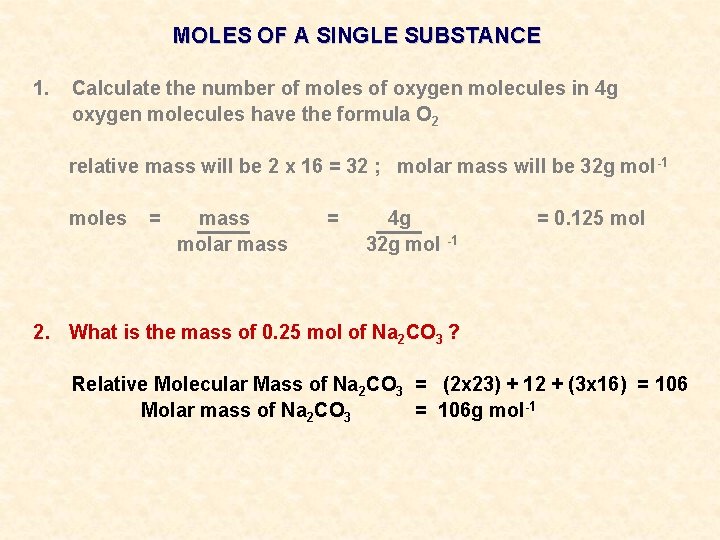
MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 = 0. 125 mol 2. What is the mass of 0. 25 mol of Na 2 CO 3 ? Relative Molecular Mass of Na 2 CO 3 = (2 x 23) + 12 + (3 x 16) = 106 Molar mass of Na 2 CO 3 = 106 g mol-1
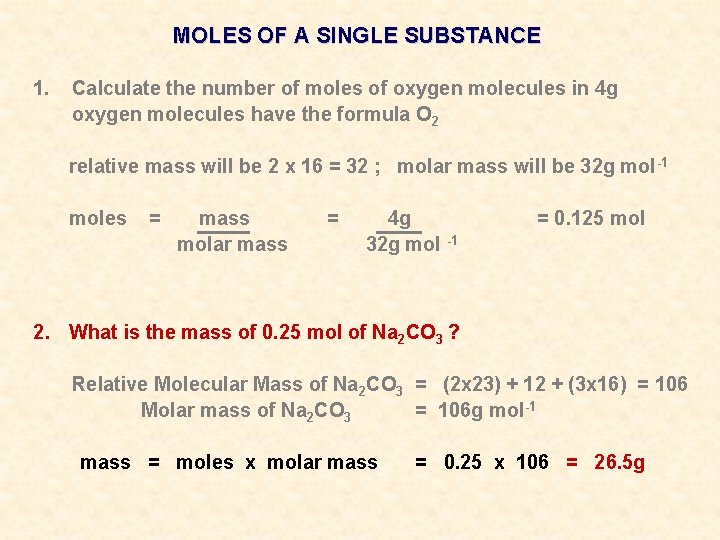
MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 = 0. 125 mol 2. What is the mass of 0. 25 mol of Na 2 CO 3 ? Relative Molecular Mass of Na 2 CO 3 = (2 x 23) + 12 + (3 x 16) = 106 Molar mass of Na 2 CO 3 = 106 g mol-1 mass = moles x molar mass = 0. 25 x 106 = 26. 5 g
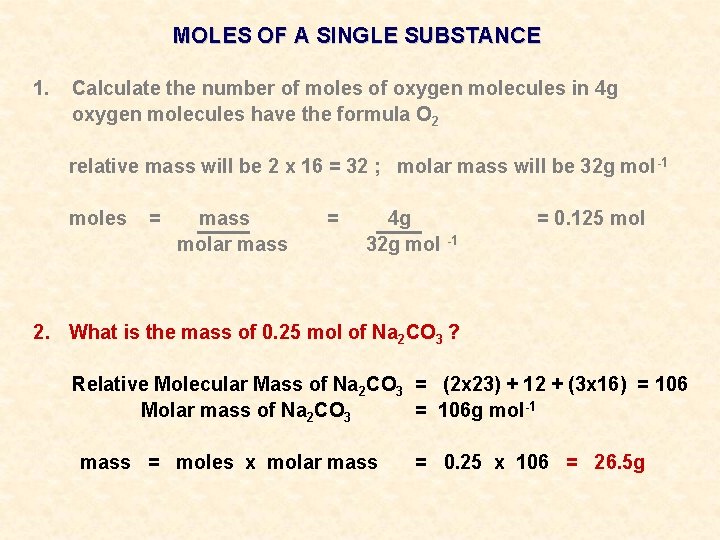
MOLES OF A SINGLE SUBSTANCE 1. Calculate the number of moles of oxygen molecules in 4 g oxygen molecules have the formula O 2 relative mass will be 2 x 16 = 32 ; molar mass will be 32 g mol -1 moles = mass molar mass = 4 g 32 g mol -1 = 0. 125 mol 2. What is the mass of 0. 25 mol of Na 2 CO 3 ? Relative Molecular Mass of Na 2 CO 3 = (2 x 23) + 12 + (3 x 16) = 106 Molar mass of Na 2 CO 3 = 106 g mol-1 mass = moles x molar mass = 0. 25 x 106 = 26. 5 g