The Mole Chemistry 11 The Mole The Mole






















































- Slides: 57

The Mole Chemistry 11

The Mole

The Mole Try it. . .

The Mole Problem. . . Another scientist enters the arena: Joseph Gay-Lussac Yet, another scientist enters the arena: Amedeo Avagadro

Volume of Gases and Mass? The Mole 1 L 24 1 kg What’s the volume of each? 1 L 24 2 kg How many particles in each? What’s the mass of each? L 11 L 24 24 3 kg

Volume of Gases and Mass? The Mole How many particles here? 1 L 24 2 kg What’s the volume of each? 1 L 24 4 kg How many particles in each? What’s the mass of each? 22 L L 48 48 kg 66 kg

The Mole Try it. . .

The Mole Think. . .

The Mole --- Molar Mass

The Mole --- Molar Mass Try it. . . Clue. . . Homework. . . Exercises 6 & 7

The Mole --- Molar Mass

The Mole --- Molar Mass Relating # of Moles and Mass Only use: mol

The Mole --- Molar Mass Try it. . . 1. . 2. . 3. .

The Mole --- Molar Mass Homework. . . Exercises 8, 9 & 10

The Mole --- Volume of a Gas Relating # of Moles and Volume of a Gas

The Mole --- Volume of a Gas Try it. . . Homework. . . Exercises 11 & 12

The Mole --- # of Particles Relating # of Moles and #of particles

The Mole --- # of Particles Try it. . . 1. . 2. .

The Mole --- # of Particles --- Molar Mass Homework. . . Exercises 15 to 20

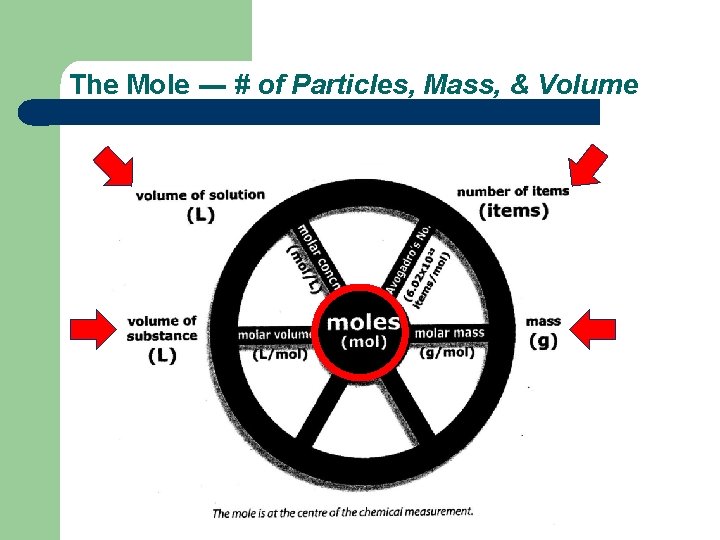
The Mole --- # of Particles, Mass, & Volume Multiple Conversions Between Moles, Mass, Volume and #of particles Try it. . . Homework. . . Exercises 21

The Mole --- # of Particles, Mass, & Volume #21 How many atoms are contained in 1 molecule of each of the following? – – – A – CH 3 CO 2 H B – NH 4 Cl C – (CH 3)2 CO D – (NH 4)2 SO 4 E – C 15 H 22 O 6 N 2 S F – Ni(H 2 O)4(NH 3)2 Cl 2 8 6 10 15 46 23

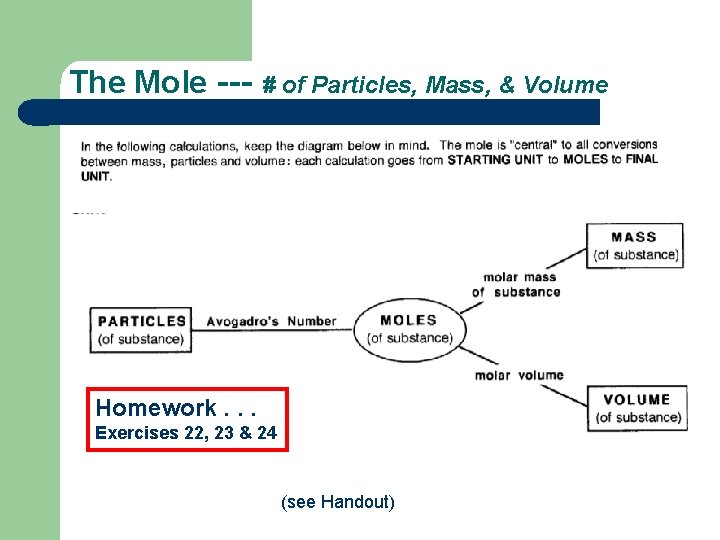
The Mole --- # of Particles, Mass, & Volume (see Handout)

The Mole --- # of Particles, Mass, & Volume

The Mole --- # of Particles, Mass, & Volume

The Mole --- # of Particles, Mass, & Volume Homework. . . Exercises 22, 23 & 24 (see Handout)

Homework. . . The Mole --- Try it. . . 1. . 2. . Exercises 22, 23 & 24 # of Particles, Mass, & Volume

The Mole --- # of Particles, Mass, & Volume Show video: The Mole – 10 min -- library

The Mole --- # of Particles, Mass, & Volume Try it. . . 1. . Clues . . . 2. . density = mass of 1 mol = 32. 0 g = 1. 43 g/L V vol. of 1 mol 22. 4 L

Homework. . . Exercises 25, to 34 The Mole --Try it. . . 3. . 4. . Go to Molarity # of Particles, Mass, & Volume

The Mole --- Percentage Composition

The Mole --- Percentage Composition

Homework. . . Exercises 44 & 45 The Mole --- Percentage Composition Try it. . . 1 mol = 30 g; 2 mol C = 24 g 24/30 = 80% 1 mol = 126. 8 g; 1 mol Fe = 55. 8 g 55. 8/126. 8 = 44% 1 mol = 162. 3 g; 1 mol Fe = 55. 8 g 55. 8/162. 3 = 34. 4% 6 mol H = 6 g 6/30 = 20% 2 mol Cl = 71 g 71/126. 8 = 56 % 3 mol Cl = 106. 5 g 106. 5/162. 3 = 65. 6%

The Mole --- Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula 1 more point to make. .

The Mole --- Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula Reminder: Try it. . . Hence the empirical formula is C 2 H 3 N Divide by the smallest Number To get unit comparison

The Mole --Try it. . . Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula Try it. . . Homework. . . Exercises #46

The Mole --- Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula

The Mole --Try it. . . Empirical & Molecular Formula

The Mole --Try it. . . Empirical & Molecular Formula

The Mole --- Empirical & Molecular Formula Try it. . . Homework. . . Exercises #47 to #55

The Mole --- Molar Concentration Knowing the concentration of a solution provides a way to find how much of a particular substance exists in a given volume of the solution.

The Mole --- Molar Concentration

The Mole --- Molar Concentration Try it. . . 1 -2 -3 -Equation for Molar Concentration

The Mole --- Molar Concentration Try it. . .

The Mole --- Molar Concentration Try it. . . Homework. . . Exercises #59 to #77

The Mole --- Molar Concentration Dilution Calculations: Or. . .

The Mole --- Molar Concentration

Handout: Dilution Calculations The Mole --- Molar Concentration

Homework. . . Exercises #78 to #94 The Mole --- Molar Concentration

The Mole --- Molar Concentration of Ions Go to Dissolved Ions

Quiz: Concentration Review The Mole --- Summary Handout: Molarity Summary Handout: Practice Problems ALSO: Mole Test Review Homework. . . Exercises #95 to #102

The Mole --- The End