Unit 4 Review Chemical Reactions Compounds The image


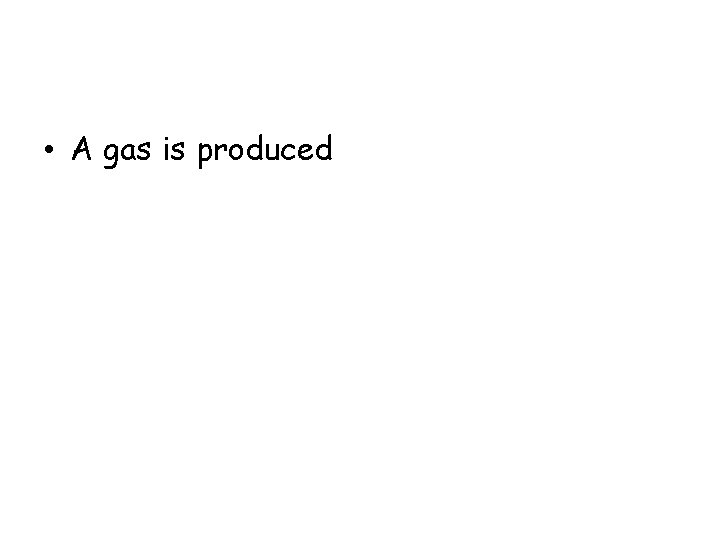

















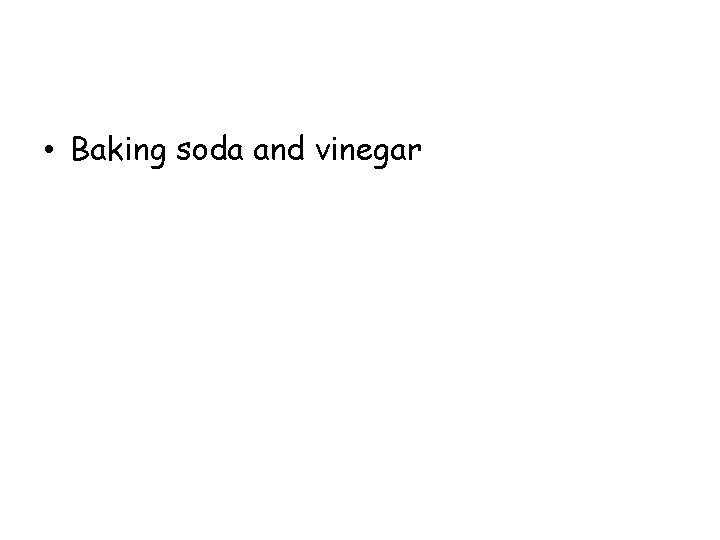






















































- Slides: 83

Unit 4 Review Chemical Reactions & Compounds

The image shows what happens when cold medicine tablets are placed in room-temperature water. What evidence would indicate that a new substance is produced?
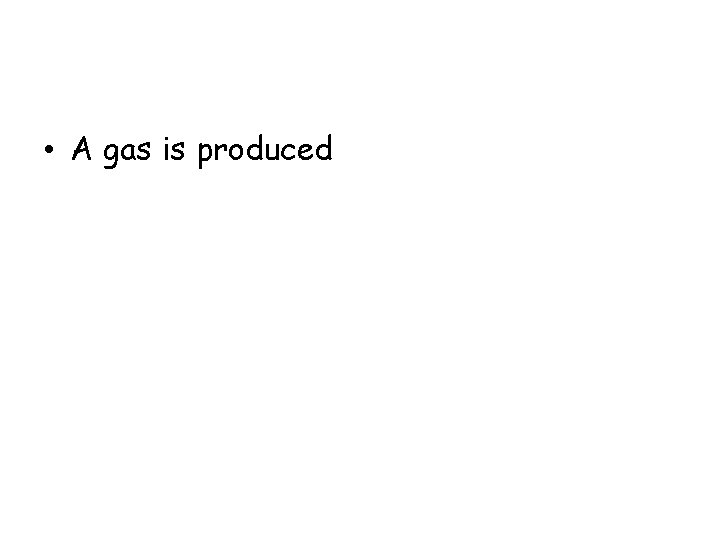
• A gas is produced

A student measures out 100 m. L of two different clear liquids and then pours one into the other. The mixture, which has a volume of 200 m. L, turns bright red, then begins to become quite cold. In this process, the first evidence that a new substance had formed was observed when the -
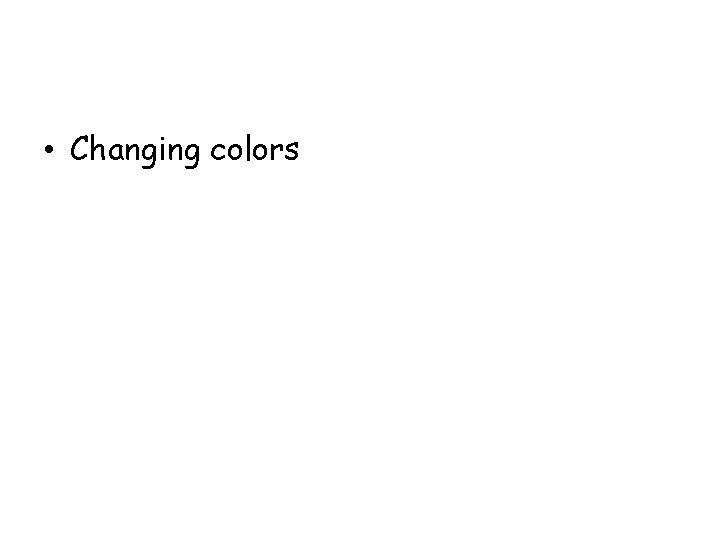
• Changing colors

Two clear liquids are weighed and then combined. Observations are made. 125 g 148 g The combined liquids have a greater mass than either of the starting liquids Does the result indicate that a new substance has formed?

• Physical, there should be more mass when two substances combine – nothing was produced

Two clear liquids are combined and observations are made. Does the following result indicate that a new substance has formed? + = The liquids, when mixed, produce a white solid which settles to the bottom

• Chemical, precipitate

A mixture is made by combining two clear liquids and observations are made. Does the following result indicate that a new substance has formed? The mixture of the two liquids takes the shape of the container they are in + =
• Characteristics of a liquid – not a chemical reaction

A mixture is made by combining four clear liquids and observations are made. Does the following result indicate that a new substance has formed? The denser of the two liquids lies below the other less dense liquid

• No new substance, evidence of density

Does this indicate a new substance has been formed? A solid has become a liquid

• Change in state of matter, no new substance

Does this indicate a new substance has been formed? A substance has permanently changed color

• Chemical change – permanent change in color

Does this indicate a new substance has been formed? A solid has dissolved in a liquid

• No, can be undone by evaporation, or distilling

Does this indicate a new substance has been formed? The volume of a substance increased.

• Yes, a permanent change has occurred

Which mixture in the following table shows evidence that a new substance was formed? Mixture Observations Baking soda and vinegar Bubbles form Salt and water Crystals are no longer visible when dissolved Oil and water Oil rises to the top Iron fillings and sand Iron fillings are attracted to a magnet
• Baking soda and vinegar

Students combine several chemicals and record the observations in their science notebooks. A list of the observed results is given here. How many of the observations are evidence that a new substance was formed? • • • Production of a gas (bubbles) Phase change (solid to liquid) A precipitate forms Change in shape Crystals dissolving Heat produced

• Production of a gas (bubbles) • A precipitate forms • Heat produced

Bubbles are evidence of _______

• A gas is being produced

Chemical or Physical? Gives off heat

• chemical

Chemical or Physical? Granules dissolve

• physical

Chemical or Physical? Volume increases

• Chemical

Does volume of a substance increasing indicate a new substance has been formed?

• Yes, chemical

Chemical or Physical? Granules seem to get smaller

• No, dissolving

Chemical or Physical? Two substances are mixed, would a decrease in temperature indicate a new substance is formed?

• Yes, endothermic reaction

Chemical or Physical? Two substances are mixed, would an increase in mass indicate a new substance is formed?
• No, would be expected when two masses are combined

Chemical or Physical? Two substances are mixed, if one of the substances dissolved indicate a new substance is formed?

• No, can be undone by evaporation, or distilling

Chemical or Physical? Two clear liquid substances are mixed, a white powder can be seen settling on the bottom of the test tube. The white powder is called?

• precipitate

What is always a characteristic of a chemical reaction?

• A new substance is formed

Physical or Chemical Frying an egg

Physical or Chemical

Physical or Chemical Toast

Physical or Chemical Toast Chemical

Physical or Chemical Bread Rising

Physical or Chemical

Physical or Chemical Cracking an Egg

Physical or Chemical Cracking an Egg Physical

Physical or Chemical Slicing Bread

Physical or Chemical Slicing Bread Physical

Physical or Chemical Lighting a Match

Physical or Chemical Lighting a Match Chemical

Physical or Chemical Candle wax melting

Physical or Chemical Physical

Physical or Chemical Candle wick burning

Physical or Chemical

Physical or Chemical Roasting marshmallows

Physical or Chemical Roasting marshmallows Chemical

Physical or Chemical Rusty Cans

Physical or Chemical Rusty Cans Chemical

Physical or Chemical Ice Melting

Physical or Chemical Ice Melting Physical

Physical or Chemical Glass Breaking

Physical or Chemical Glass Breaking Physical

Physical or Chemical Boiling Water

Physical or Chemical Boiling Water Physical

Physical or Chemical Fresh Lemonade

Physical or Chemical Fresh Lemonade Physical

Physical or Chemical Baking a Cake

Physical or Chemical Baking a Cake Chemical

Physical or Chemical Mowing the Lawn

Physical or Chemical Mowing the Lawn Physical

Physical or Chemical Fireworks

Physical or Chemical Fireworks Chemical

Physical or Chemical Digesting food

Physical or Chemical Digesting food Chemical