Ionic Bonding New Unit Chemical Bonding Essential Questions

Ionic Bonding

New Unit: Chemical Bonding Essential Questions: What are the processes by which different atoms come together to form new compounds and what forces hold solutions of these compounds/molecules together?


Ions �An atom with a charge Positive: cation Negative: anion How do these atoms become charged?

Valence Electrons �Electrons in the highest occupied energy level �The number of valence electrons greatly determines chemical properties of an element and how it will react How can we find the number of valence electrons in an element?

By using electron configurations Electron configuration for chlorine: 1 s 22 p 63 s 23 p 5 What is the highest energy level reached? 3 How many electrons are there? 7

Because of trends, we can also use group numbers! Looking at the electron configuration for elements in the same group, they will all have the same number of valence electrons (just the A groups though! Not the B groups. )

Guided Practice on Valence Electrons How many valence electrons are there in the following elements? Chlorine Calcium Aluminum Carbon Neon

Electron Dot Structures �Also known as Lewis Structures �Diagrams that represent the number of valence electrons in the atom 1. 2. 3. Find number of valence electrons Write element symbol Fill valence electrons around symbol, one on each side first then double up

Guided Practice on Lewis Structures Find the Lewis Dot Structures for the following elements: Chlorine Calcium Aluminum Carbon Neon

Octet Rule �Noble gases are the most stable elements on the Periodic Table due to the fact that they have 8 valence electrons (a complete outer shell) �Atoms will either lose or gain electrons through bonding in order to attain those complete 8 configurations Metal elements tend to lose electrons while nonmetals tend to gain

Forming ions �How do cations form? Losing valence electrons ▪ Who tends to form cations? �How do anions form? Gaining valence electrons ▪ Who tends to form anions?


Forming cations �Metals �Lose valence electrons Example: Calcium Normal calcium ion 20 protons 20 electrons no charge 20 protons 18 electrons +2 charge

Forming anions �Nonmetals �Gain electrons to get to an octet Example: Chlorine normal chlorine 17 protons 17 electrons no charge chlorine ion 17 protons 18 electrons -1 charge

Independent Practice

Formation of Ionic Compounds �Composed of a cation and an anion �Held together by ionic bonds �Represented by a chemical formula �A formula unit is the lowest whole-number ratio of ions in an ionic compound

Formation of Ionic Compounds �When writing the formulas, use the criss- cross method Mg+2 Cl-1 Mg. Cl 2

So then, What’s an ionic bond? �A chemical electrostatic force between a cation and an anion. �Can you have two cations come together? Two anions?
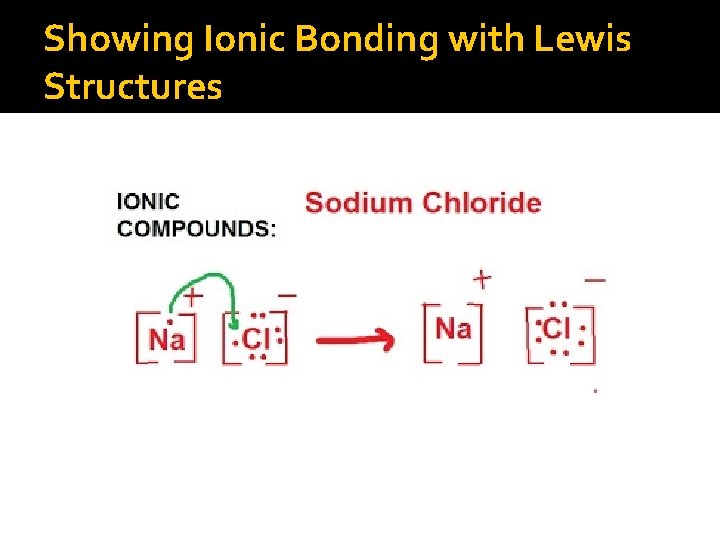
Showing Ionic Bonding with Lewis Structures
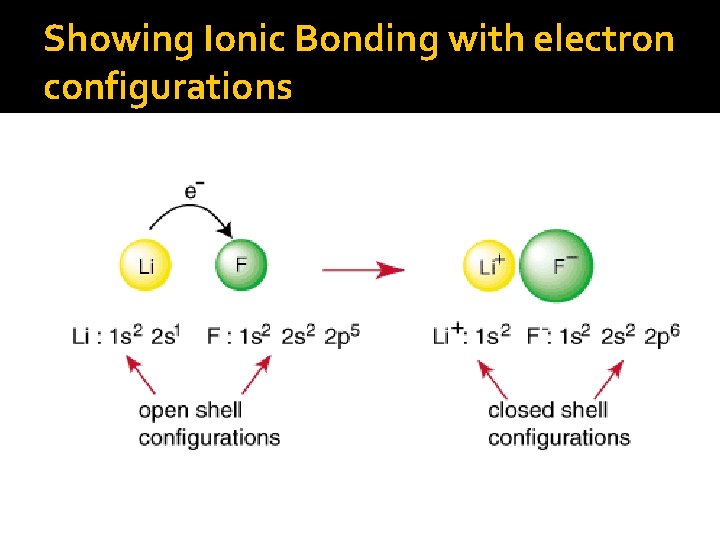
Showing Ionic Bonding with electron configurations

Guided Practice on Ionic Compound Formulas �For the following pairs of elements, write the formula for the ionic compound formed Aluminum and Chlorine Sodium and Sulfur Nitrogen and fluorine

Question �Why are ionic compounds composed of ions but have no charge overall?

Properties of Ionic compounds �Most are crystalline solids at room temperature �Generally have high melting points �Can conduct electric current when melted or dissolved in water
Ions Discussion and Demo

Graphic Organizer Review


Independent Practice �Finish the two worksheets and complete the ionic review questions on a left page of your notebook.
- Slides: 28