Matter Matter Anything that has mass and volume

















![• Starting substances [reactants] have different compositions and properties from the new substances • Starting substances [reactants] have different compositions and properties from the new substances](https://slidetodoc.com/presentation_image_h2/ae44dd16e2e9b3bf8b2972933a70ade4/image-41.jpg)




















- Slides: 72

Matter

Matter

• Anything that has mass and volume (occupied space) MATTER Defined

Matter Pure Substances

• Uniform and unchanging composition • Chemical formula can be written for a substance Pure Substances

PROPERTIES OF MATTER Characteristics and behavior

Characteristics that can be observed through the five senses or measured without changing the sample’s composition or identity Physical Properties

Physical properties of pure substances are consistent and unchanging due to their uniform and unchanging compositions Physical Properties

Extensive Properties Intensive Properties • Depend upon the amount of substance present • Examples: • Independent of amount of substance present • Examples: • Mass • Length • Volume Two Types • Color, odor, taste, hardness • Density, melting point, boiling point • Malleability, ductility • Electrical and thermal conductivity

What are some physical properties of water?

The ability of a substance to combine with or change into one or more other substances Includes the inability to change into other substances Chemical Properties

Evident when substance comes in contact with another substance or when thermal or electrical energy is applied Chemical Properties

• The ability to: rust corrode burn explode rot change color react or the inability to do these things Chemical Properties

Theability water to The ability ofofiron ofto rust decompose when magnesium combined to into burn within hydrogen andofoxygen the presence oxygen Examples

Substances have unique sets of physical and chemical properties Properties are helpful in identifying unknown substances Properties

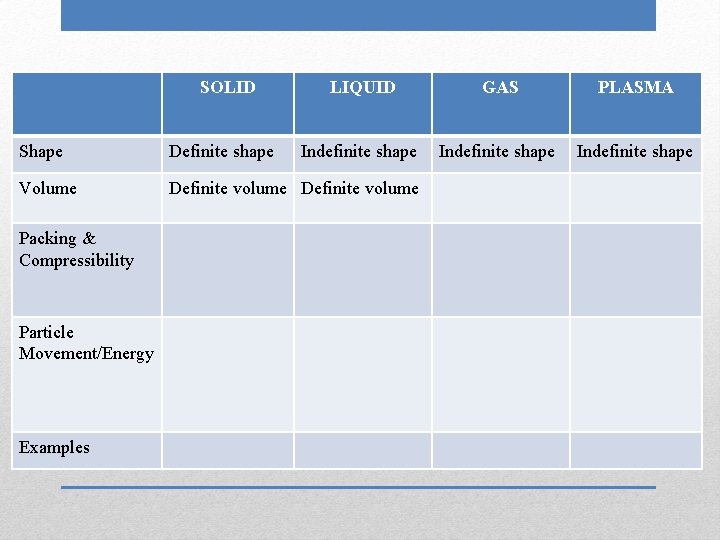
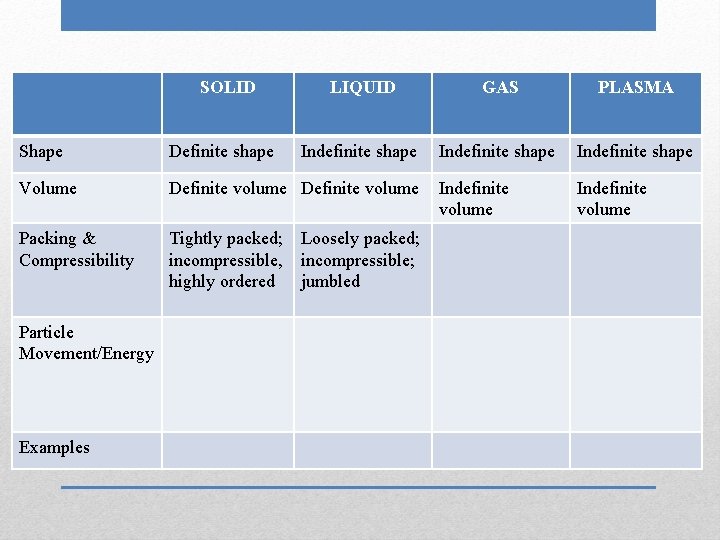
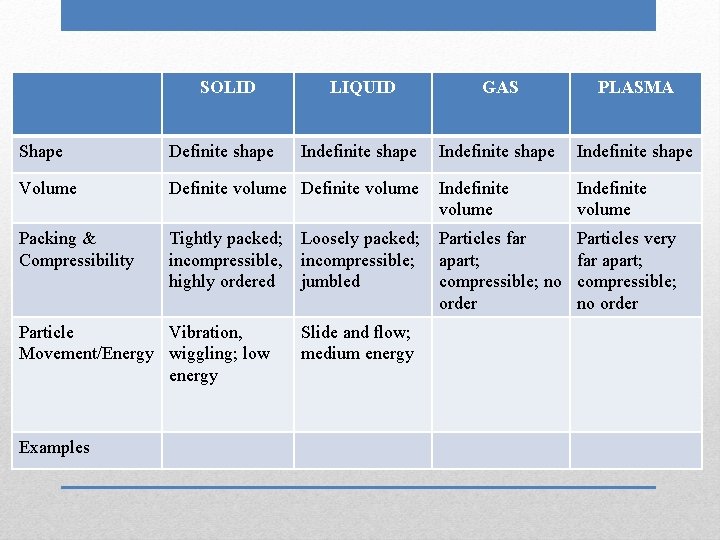
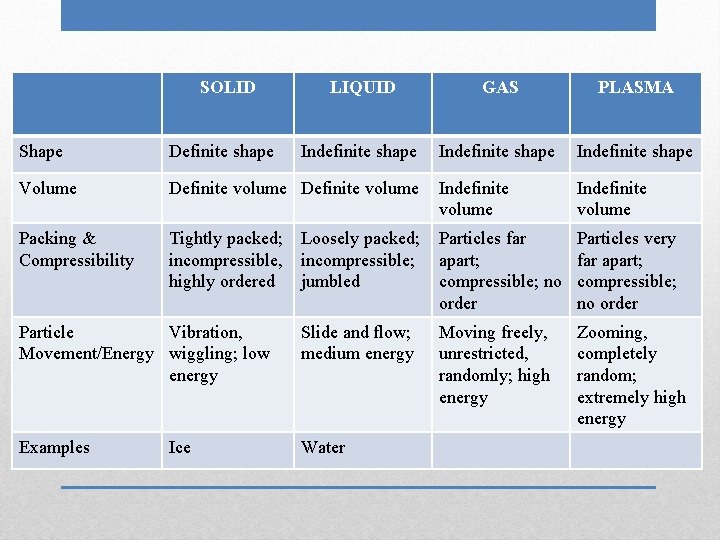
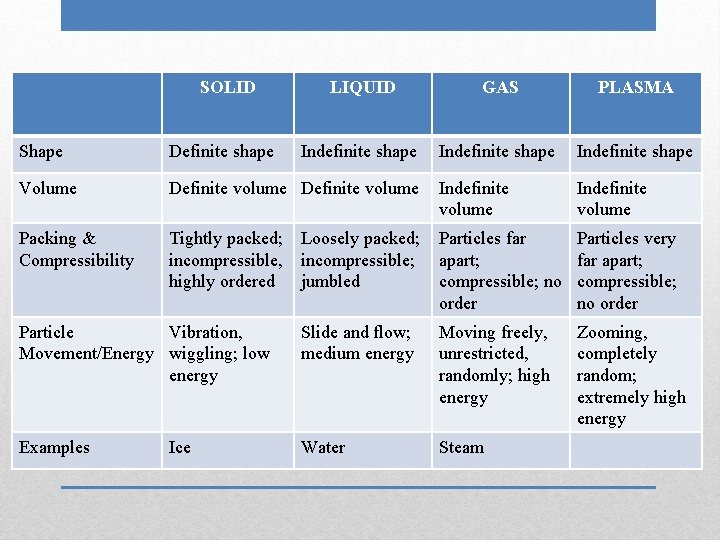
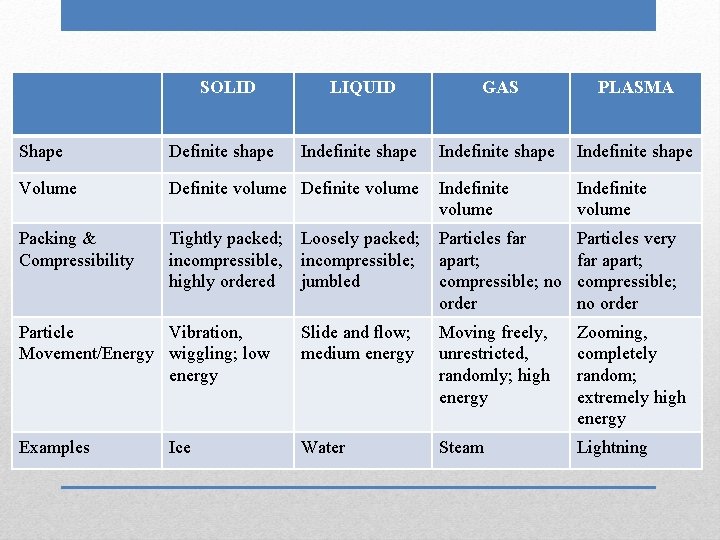
SOLID LIQUID GAS Shape Volume Packing & Compressibility Particle Movement/Energy Examples Phases (States) of Matter PLASMA

SOLID Shape Volume Packing & Compressibility Particle Movement/Energy Examples Definite shape LIQUID GAS PLASMA

SOLID Shape Volume Packing & Compressibility Particle Movement/Energy Examples Definite shape LIQUID Indefinite shape GAS PLASMA

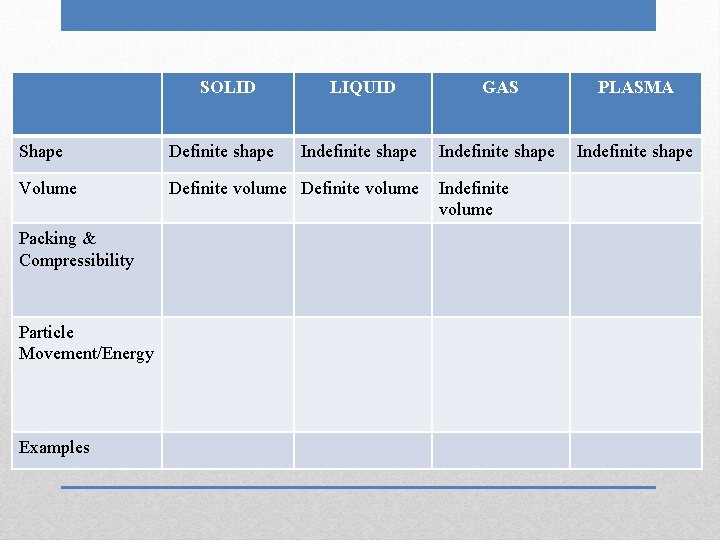
SOLID Shape Volume Packing & Compressibility Particle Movement/Energy Examples Definite shape LIQUID GAS Indefinite shape PLASMA

SOLID Shape Volume Packing & Compressibility Particle Movement/Energy Examples Definite shape LIQUID GAS PLASMA Indefinite shape

SOLID Shape Definite shape Volume Definite volume Packing & Compressibility Particle Movement/Energy Examples LIQUID GAS PLASMA Indefinite shape

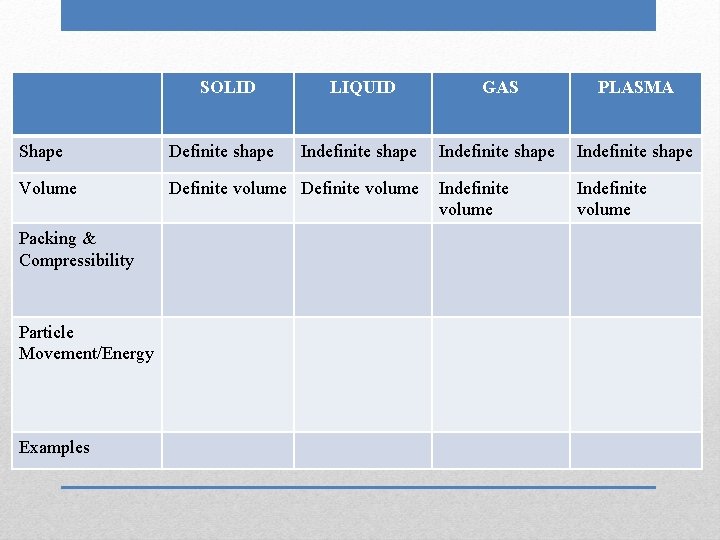
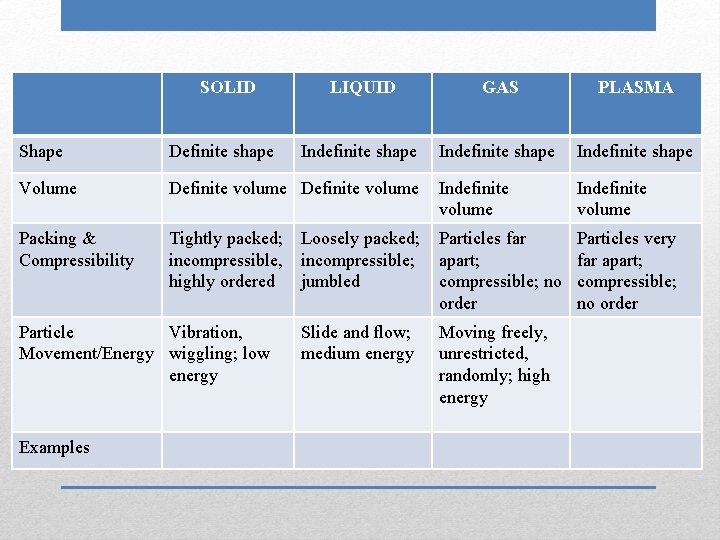
SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Packing & Compressibility Particle Movement/Energy Examples

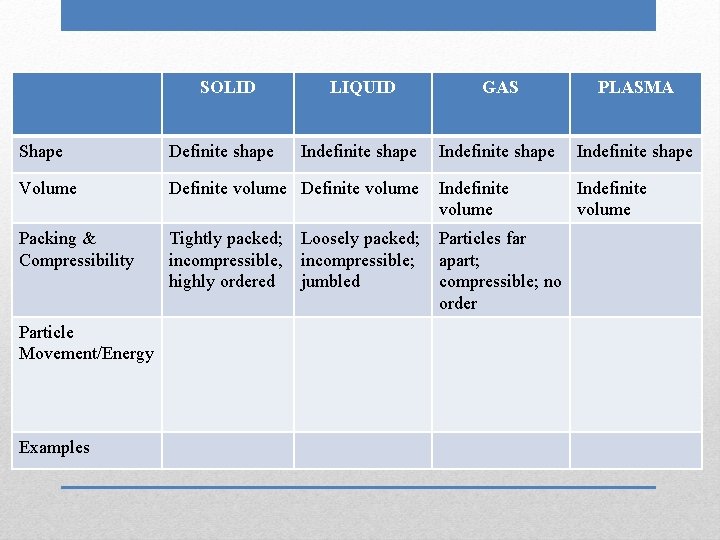
SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Packing & Compressibility Particle Movement/Energy Examples Indefinite volume

SOLID LIQUID GAS PLASMA Indefinite shape Indefinite volume Shape Definite shape Volume Definite volume Packing & Compressibility Particle Movement/Energy Examples

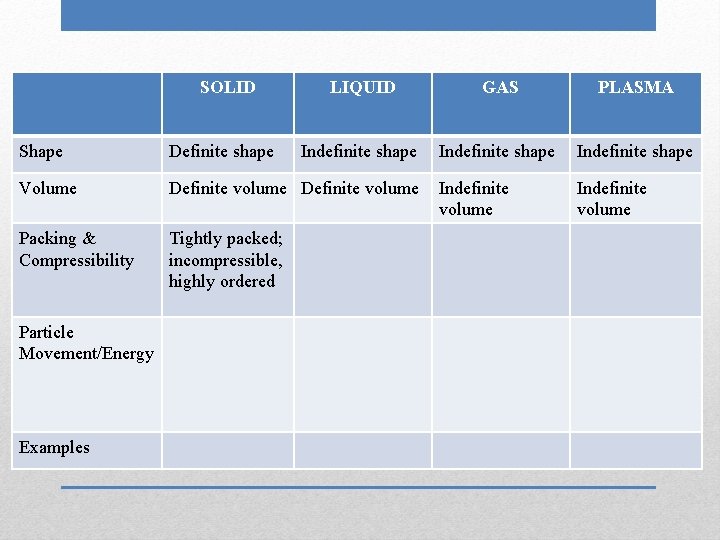
SOLID LIQUID GAS PLASMA Indefinite shape Indefinite volume Shape Definite shape Volume Definite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particle Movement/Energy Examples

SOLID LIQUID GAS PLASMA Indefinite shape Indefinite volume Shape Definite shape Volume Definite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particle Movement/Energy Examples Loosely packed; incompressible; jumbled

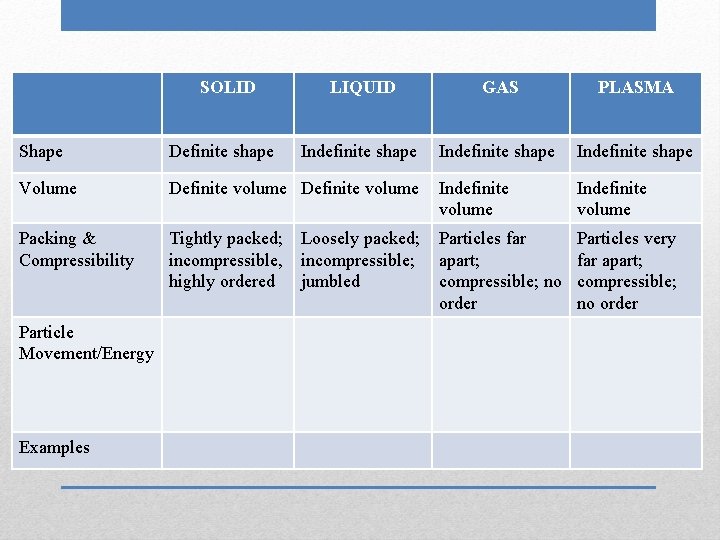
SOLID LIQUID GAS PLASMA Indefinite shape Indefinite volume Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particles far apart; compressible; no order Particle Movement/Energy Examples Loosely packed; incompressible; jumbled

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Movement/Energy Examples Loosely packed; incompressible; jumbled

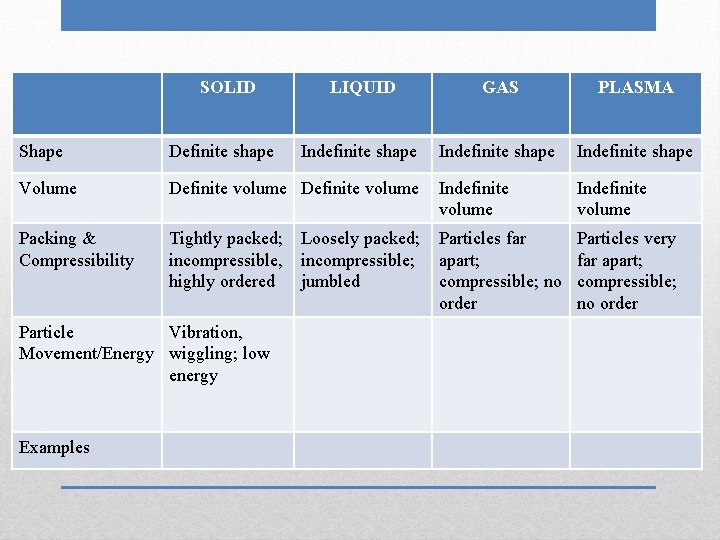
SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Vibration, Movement/Energy wiggling; low energy Examples Loosely packed; incompressible; jumbled

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Vibration, Movement/Energy wiggling; low energy Examples Loosely packed; incompressible; jumbled Slide and flow; medium energy

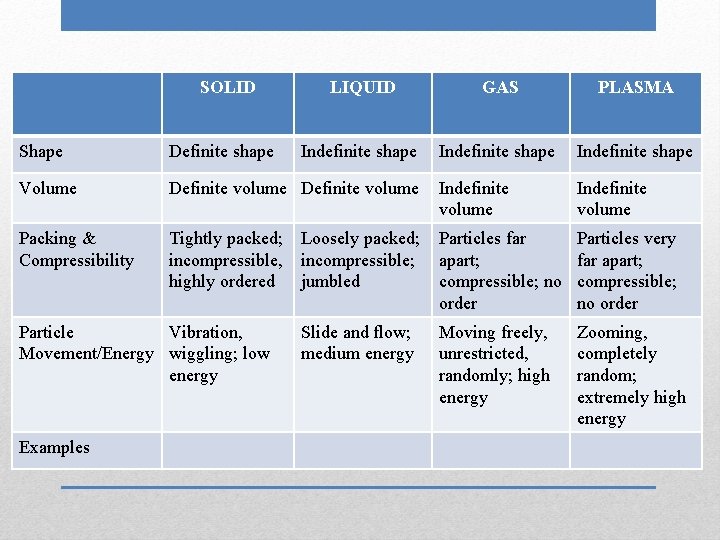
SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Particle Vibration, Movement/Energy wiggling; low energy Examples

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Zooming, completely random; extremely high energy Particle Vibration, Movement/Energy wiggling; low energy Examples

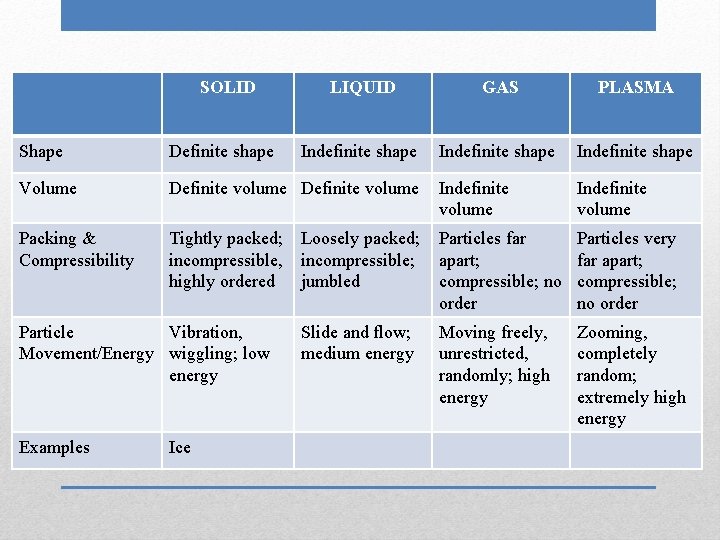
SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Zooming, completely random; extremely high energy Particle Vibration, Movement/Energy wiggling; low energy Examples Ice

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Vibration, Movement/Energy wiggling; low energy Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Zooming, completely random; extremely high energy Examples Water Ice

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Vibration, Movement/Energy wiggling; low energy Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Zooming, completely random; extremely high energy Examples Water Steam Ice

SOLID LIQUID GAS PLASMA Indefinite shape Shape Definite shape Volume Definite volume Indefinite volume Packing & Compressibility Tightly packed; incompressible, highly ordered Loosely packed; incompressible; jumbled Particles far apart; compressible; no order Particles very far apart; compressible; no order Particle Vibration, Movement/Energy wiggling; low energy Slide and flow; medium energy Moving freely, unrestricted, randomly; high energy Zooming, completely random; extremely high energy Examples Water Steam Lightning Ice

VAPOR VS. GAS Gas: substance that is naturally in gaseous state at room temperature Vapor: gaseous state of substance that is normally solid or liquid at room temperature

Changes which alter a substance without changing its composition or identity Physical Changes

• Cut, break, bend, grind, crumple, split, crush, dissolve, fold • Include phase changes: most substances undergo a change from one state of matter to another as the temperature and pressure conditions change • Melt, freeze, boil, vaporize, condense Physical Changes

Processes involving one or more substances changing into new substances Also referred to as chemical reactions Chemical Changes
![Starting substances reactants have different compositions and properties from the new substances • Starting substances [reactants] have different compositions and properties from the new substances](https://slidetodoc.com/presentation_image_h2/ae44dd16e2e9b3bf8b2972933a70ade4/image-41.jpg)
• Starting substances [reactants] have different compositions and properties from the new substances formed [products] • Represented by chemical equations Reactants → Products Chemical Changes

• • Explode Rust Oxidize Corrode Tarnish Ferment Burn Rot Chemical Changes

• • • Formation of a gas Formation of a solid (precipitate) Change in temperature or energy Change in smell or production of odor Change in color Change in magnetism Evidence/Indicators of Chemical Reaction

LAW OF CONSERVATION OF MATTER Mass is neither created nor destroyed in any process; it is conserved.

LAW OF CONSERVATION OF MATTER Massreactants = Massproducts

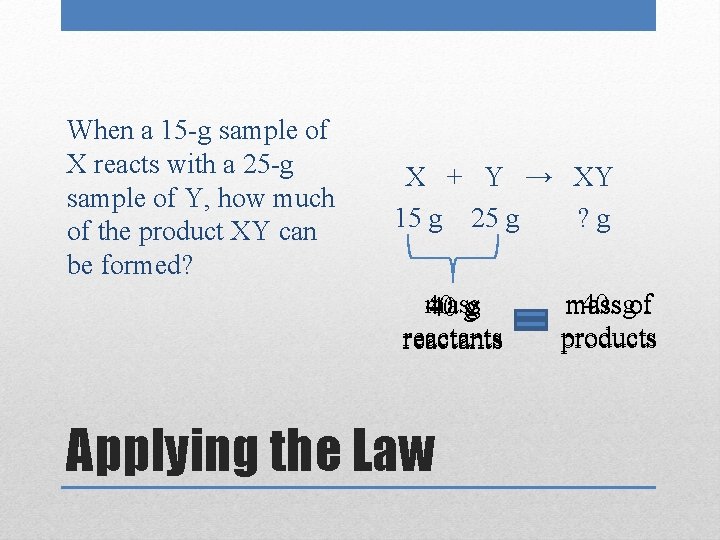
When a 15 -g sample of X reacts with a 25 -g sample of Y, how much of the product XY can be formed? X + Y → XY 15 g 25 g ? g mass 40. 40 gg reactants Applying the Law 40. gof mass products

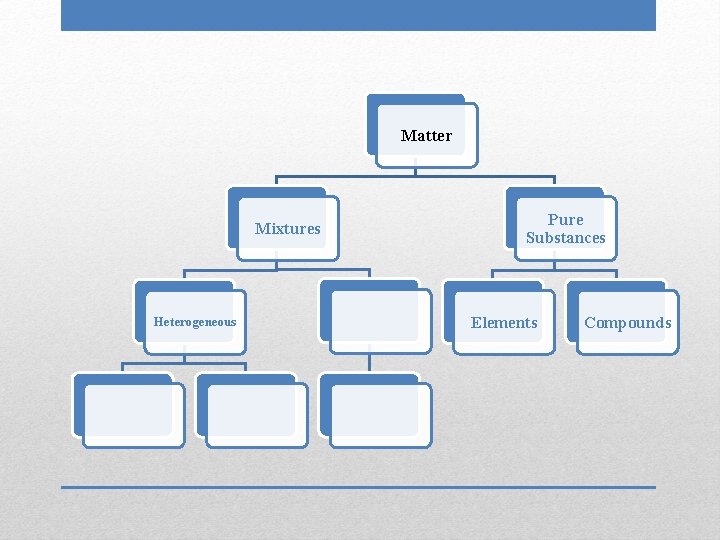
Matter Pure Substances

Matter Pure Substances Elements

• Contain only one type of atom: one name, one symbol • Found on Periodic Table • Cannot be separated into simpler substances through physical or chemical means Elements

Matter Pure Substances Elements Compounds

• Two or more substances chemically combined: two names/symbols • Can be broken down by chemical means, which requires energy • Properties of compounds are very different from properties of individual elements Compounds

Matter Mixtures Pure Substances Elements Compounds

• • Two or more substances physically combined Substances retain their individual properties Variable composition Can be separated by physical means Mixtures

Matter Mixtures Heterogeneous Pure Substances Elements Compounds

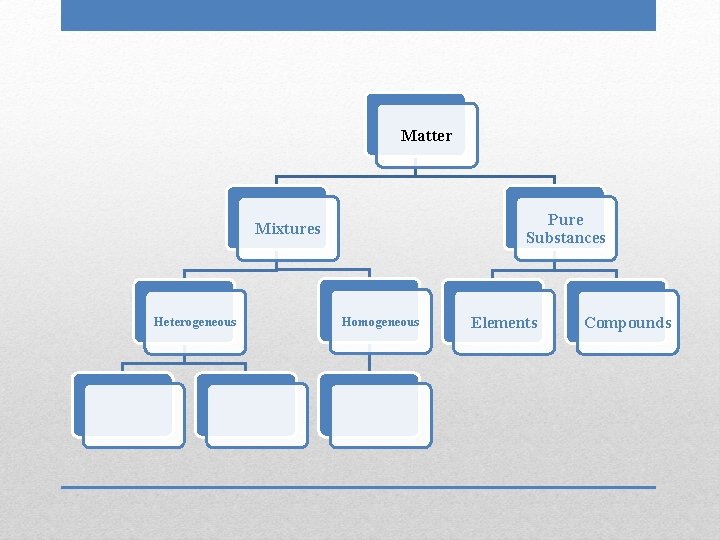
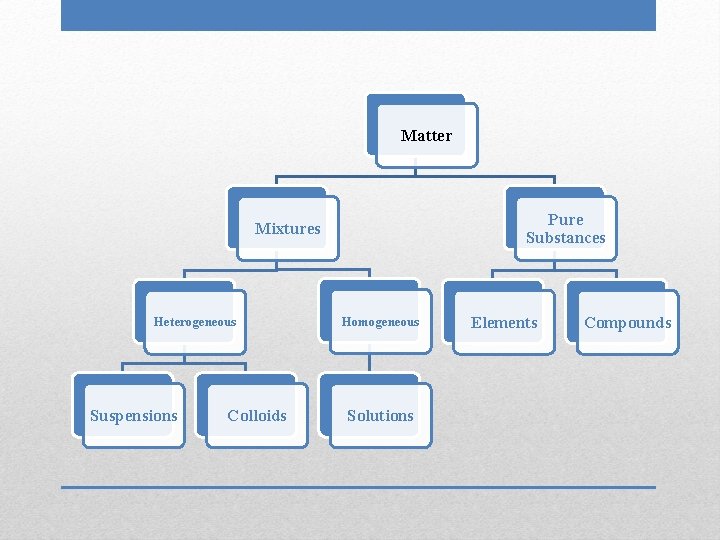
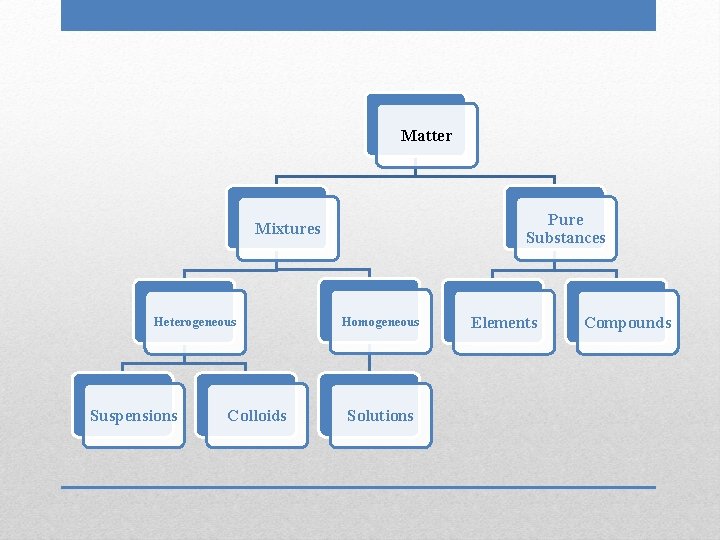
• Not uniform in composition • Different phases; individual substances remain distinct • Tend to have cloudy appearance Heterogeneous Mixtures

Matter Pure Substances Mixtures Heterogeneous Homogeneous Elements Compounds

• Uniform, constant composition • Single phase • Clear, transparent Homogeneous Mixtures

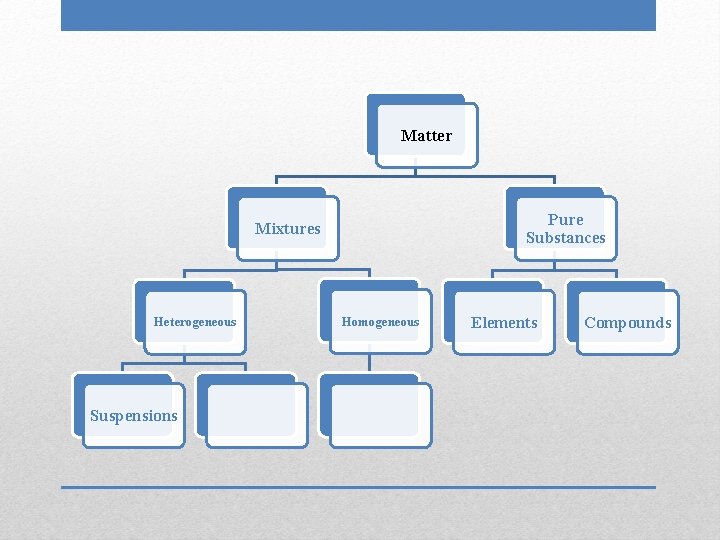
Matter Pure Substances Mixtures Heterogeneous Suspensions Homogeneous Elements Compounds

• Largest particle size • Particles can be filtered out of mixture • Positive Tyndall effect Suspensions

• Phenomenon in which particles of a mixture scatter light Tyndall Effect

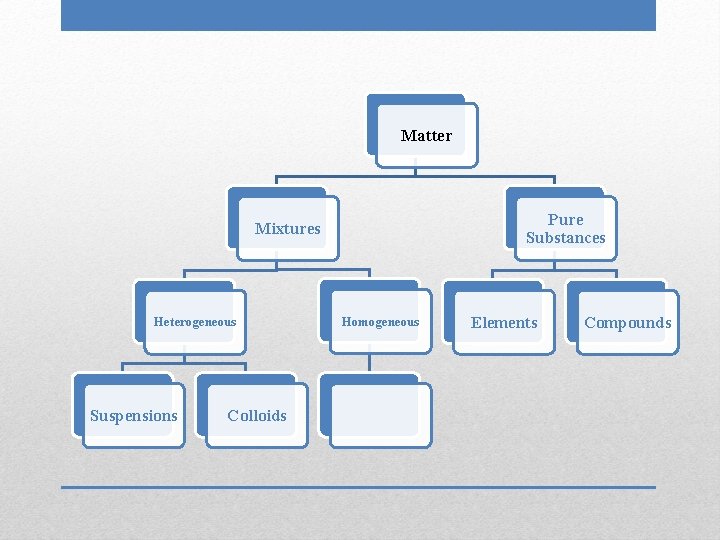
Matter Pure Substances Mixtures Heterogeneous Suspensions Colloids Homogeneous Elements Compounds

• Intermediate particle size • Particles do not filter out • Positive Tyndall effect Colloids

Matter Pure Substances Mixtures Heterogeneous Suspensions Colloids Homogeneous Solutions Elements Compounds

• Smallest particle size • Particles do not filter out • Negative Tyndall effect Solutions

Matter Pure Substances Mixtures Heterogeneous Suspensions Colloids Homogeneous Solutions Elements Compounds

Separation Techniques To separate mixtures into their component substances

• Manual means “by hand” • Use tool, such as tweezers, to remove and separate components of a mixture • Used with suspensions Manual Separation

• Uses a porous barrier to separate solids from liquids. • Example: Filter paper in a funnel to separate sand from water • Used with suspensions Filtration

• Based on the differences in boiling points of substances • Example: When boiling salt water solution, water vaporizes first • Used with solutions, colloids Distillation

• Results in formation of pure solid particles of a substance from a solution • Example: When making rock candy, sugar forms solid crystals from liquid solution • Used with solutions Crystallization

• Based on tendency of components of mixture to travel across surface of another material • Example: Separating ink dyes • Used with solutions Chromatography

• Allows a liquid to be separated quickly from a heavier solid • Example: Pouring water off a mixture with solid materials • Used with suspensions Decantation