Matter Anything that has mass substance and volume

Matter ■ Anything that has mass (substance) and volume (takes up space). ■ Matter is made of atoms. ■ Atoms function as building blocks for all substances. ■ Everything around us is either a pure substance, a mixture, or a combination of both.
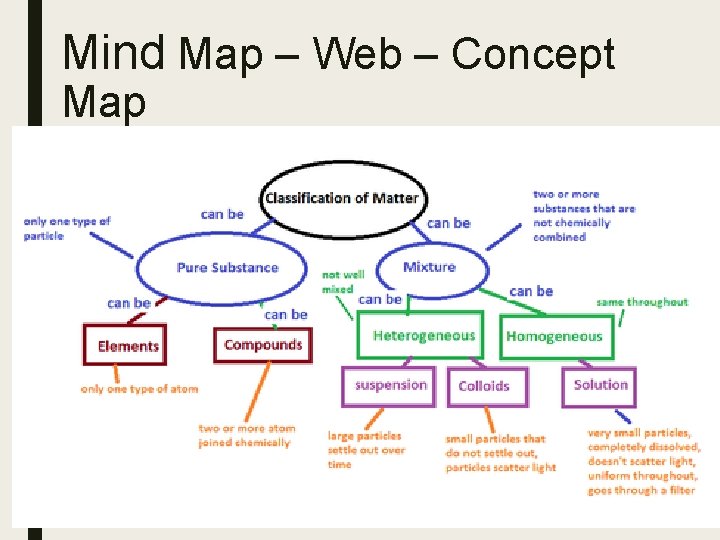
Mind Map – Web – Concept Map

Pure Substances ■ Pure substances cannot be broken down by physical changes. ■ This means you can’t take it apart unless you do a chemical reaction (chemical change). ■ Include elements and compounds.

Pure Substances - Elements ■ Elements are pure substances. ■ Elements are made of the same type of atoms. ■ H = one atom of hydrogen ■ O = one atom of oxygen ■ C = one atom of carbon

Molecule ■ A molecule is a pure substance made up of at least two atoms bonded together. ■ A molecule may be two of the same element (the diatomic elements like O 2 , etc. ) ■ Or, a molecule may be a compound like H 2 O. ■ All compounds are molecules; however not all molecules are compounds.

Pure Substances - Compounds § A compound is made when two or more different elements are chemically bonded together. § Compounds cannot be broken down except by a chemical process. § Compounds have formulas that show the specific proportions of the elements that make them. § A compound has different properties than the elements that make it up.

Examples of Compounds ■ ■ ■ Na. Cl H 2 O 2 CO CO 2 C 6 H 12 O 6 § § § Table salt Hydrogen peroxide Carbon monoxide Carbon dioxide Glucose

Mixtures ■ Mixtures are a result of a physical change. ■ A mixture is a combination of substances that are NOT chemically bonded. ■ Mixtures are different than pure substances because they can be taken apart by physical processes like boiling, filtering, magnets, using a dropper, distillation, using a centrifuge, and by hand. ■ Mixtures do not have a chemical formula. ■ Mixtures are either heterogeneous or homogeneous. ■ Mixtures can be solid, liquid, or gas.

Heterogeneous Mixtures § Heterogeneous means different. § These mixtures are not the same throughout. § One section may have bigger chunks than other sections. § You can usually see that the parts are different. § Examples: § Salad § Chili § Dirt § A toy box § A pizza

Homogeneous Mixtures ■ Homogeneous mixtures are the same throughout. ■ You cannot usually see the parts. ■ Examples: – Homogenized milk – Ice cream – Soft drinks (Coke, Dr. Pepper) – Air – Brass

What is a Solution? ■ A mixture of two or more substances that is identical throughout –a homogeneous mixture. ■ Almost anything can be a solution: – Solids dissolved in liquids (sugar in tea) – Gases dissolved in liquids (ex. carbonated drinks) – Liquids in liquids; gases in gases; solids in solids ■ If you mix things up and they stay in an even distribution, it is a solution.

Solutions ■ Made by dissolving. ■ In this example a solid is dissolved into a liquid. ■ The solute (sugar) is the substance or matter being dissolved or is soluble (able to be dissolved) in the solvent. ■ The solvent (water) is the substance into which the solute is dissolved.


How it Works (In Dot Diagrams)

Important to Remember! ■ Solutions are the same no matter where they are located in a mixture ■ For example, if you mixed a solution of water and sugar and took a sample from the top of the glass, it would have the same amount of sugar as if you took it from the bottom or middle of the glass.

Special Types of Mixtures ■ Alloys: are basically a mixture of two or more metals. – Example: brass is a mixture of copper and zinc ■ Colloid: a mixture with small undissolved particles that do not settle out, but the particles are large enough to scatter a light beam. – Example: Jello, cream ■ Suspension: a mixture in which particles are large enough to be seen and easily separated by settling or filtration. – Example: mixing pepper and water; a muddy pond
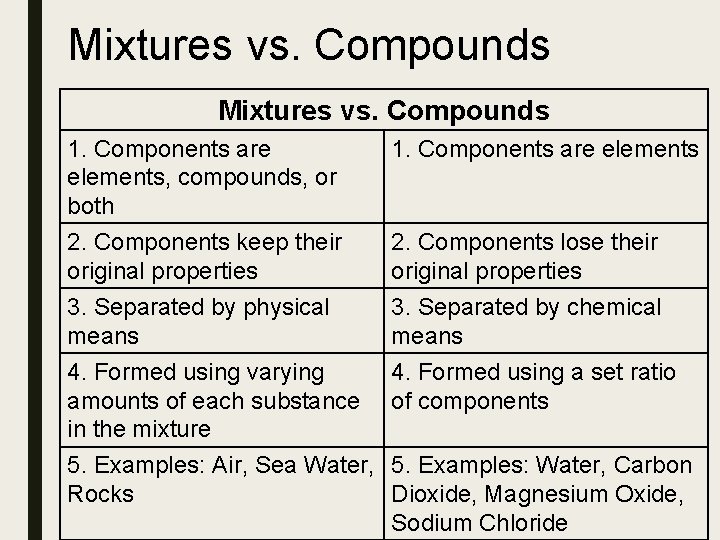
Mixtures vs. Compounds 1. Components are elements, compounds, or both 1. Components are elements 2. Components keep their original properties 2. Components lose their original properties 3. Separated by physical means 3. Separated by chemical means 4. Formed using varying amounts of each substance in the mixture 4. Formed using a set ratio of components 5. Examples: Air, Sea Water, 5. Examples: Water, Carbon Rocks Dioxide, Magnesium Oxide, Sodium Chloride

- Slides: 18