Solutions Solution Solvent Solute Solvent a substance that

Solutions

Solution = Solvent + Solute • Solvent: a substance that dissolves another substance – Or, the substance present in greater amount • Solute: a substance which is dissolved by another substance – Or, the substance present in lesser amount
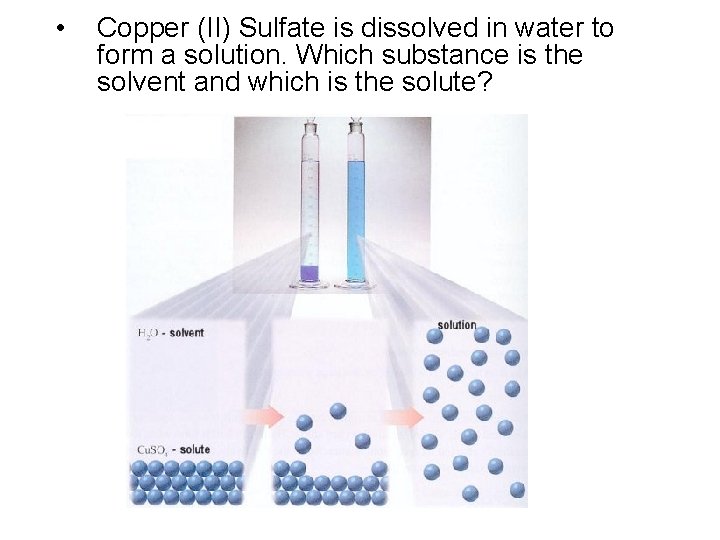
• Copper (II) Sulfate is dissolved in water to form a solution. Which substance is the solvent and which is the solute?

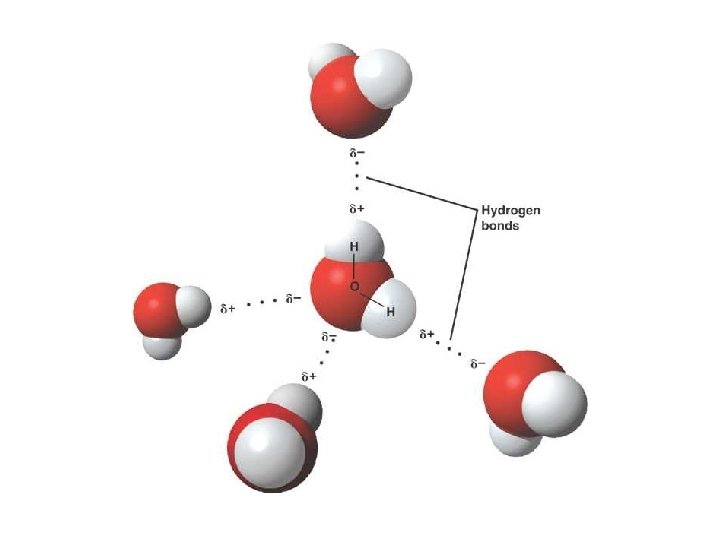
• Water is sometimes called the “Universal Solvent” – It is the most common solvent in nature/biological systems • Why do you think water is such a good solvent? – Polarity – Hydrogen bonding
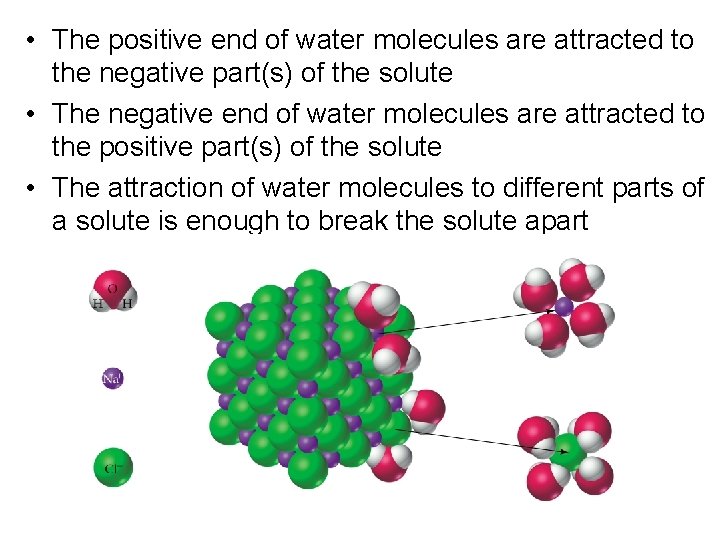
• The positive end of water molecules are attracted to the negative part(s) of the solute • The negative end of water molecules are attracted to the positive part(s) of the solute • The attraction of water molecules to different parts of a solute is enough to break the solute apart
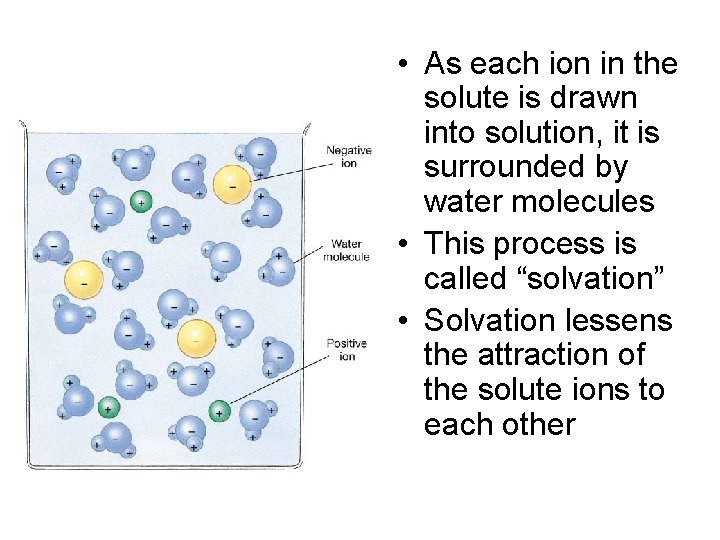
• As each ion in the solute is drawn into solution, it is surrounded by water molecules • This process is called “solvation” • Solvation lessens the attraction of the solute ions to each other

• If water is the universal solvent, then why does it not dissolve oil? • For a solution to form, the solvent and solute molecules must be attracted to each other • “Like Dissolves Like” • Polar solvents dissolve polar solutes • Nonpolar substances are attracted to other nonpolar substances, therefore nonpolar solvents will dissolve nonpolar solutes – Examples: I 2, Hexane, Cooking Oils, Dichloromethane (CH 2 Cl 2)

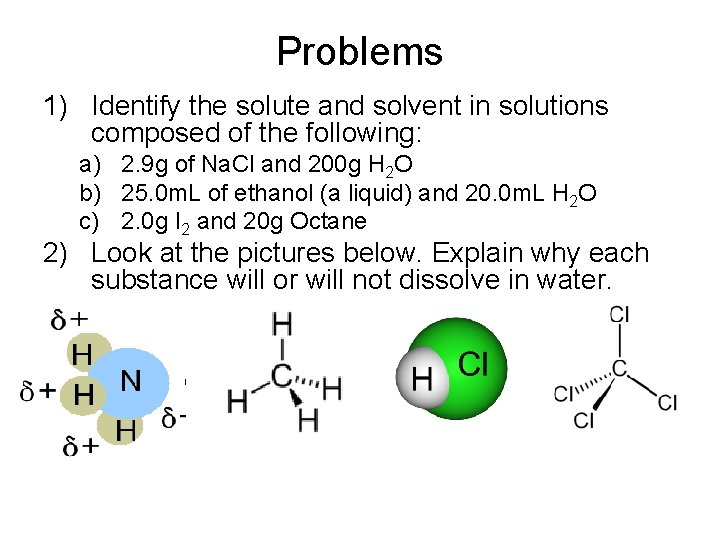
Problems 1) Identify the solute and solvent in solutions composed of the following: a) 2. 9 g of Na. Cl and 200 g H 2 O b) 25. 0 m. L of ethanol (a liquid) and 20. 0 m. L H 2 O c) 2. 0 g I 2 and 20 g Octane 2) Look at the pictures below. Explain why each substance will or will not dissolve in water.

Solubility • Solubility: the amount of solute that will dissolve in a specific solvent under given conditions – Polar solutes will be more soluble in polar solvents – Non-polar solutes will be more soluble in non-polar solvents – Amphiphilic solutes will be soluble in both polar and non-polar solvents • Have hydrophobic and hydrophilic regions – Example: Phospholipids
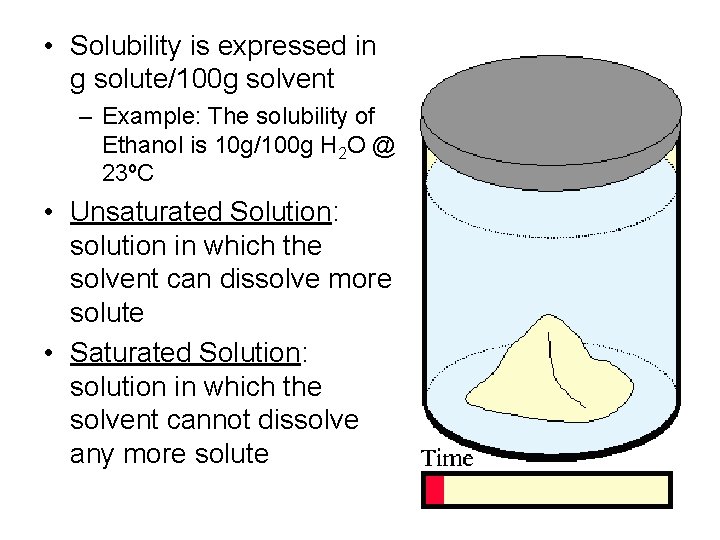
• Solubility is expressed in g solute/100 g solvent – Example: The solubility of Ethanol is 10 g/100 g H 2 O @ 23ºC • Unsaturated Solution: solution in which the solvent can dissolve more solute • Saturated Solution: solution in which the solvent cannot dissolve any more solute

• When a solution is saturated, the rate of dissolution is the same as the rate of recrystallization/precipitation
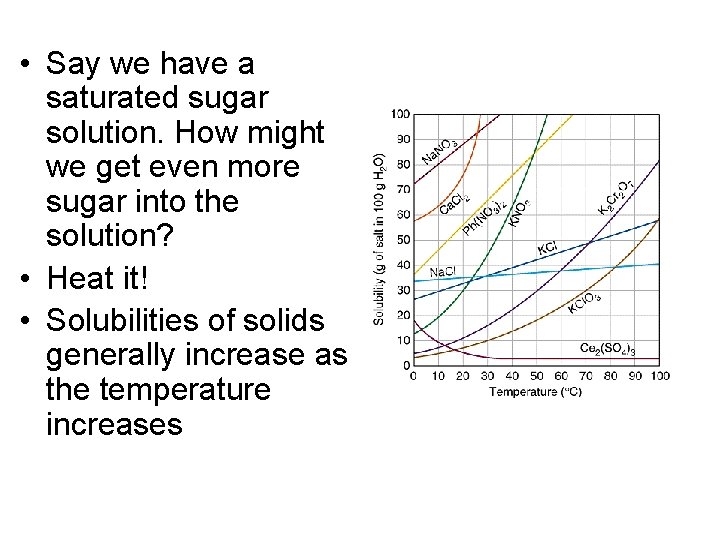
• Say we have a saturated sugar solution. How might we get even more sugar into the solution? • Heat it! • Solubilities of solids generally increase as the temperature increases

• Supersaturated solution: an unstable solution which temporarily contains more dissolved solute than a normal saturated solution would at that specified temp
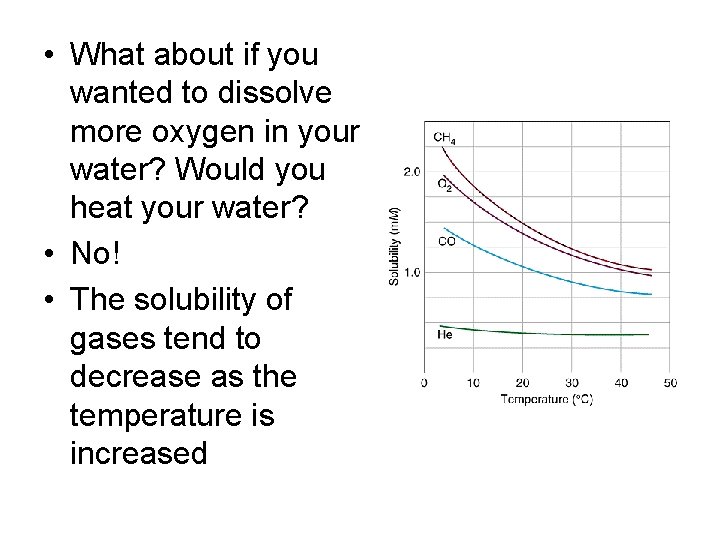
• What about if you wanted to dissolve more oxygen in your water? Would you heat your water? • No! • The solubility of gases tend to decrease as the temperature is increased

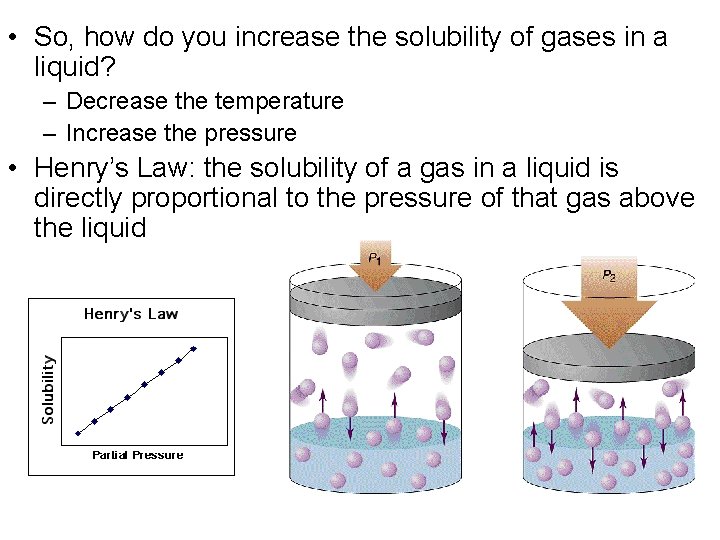
• So, how do you increase the solubility of gases in a liquid? – Decrease the temperature – Increase the pressure • Henry’s Law: the solubility of a gas in a liquid is directly proportional to the pressure of that gas above the liquid

Soluble and Insoluble Salts • How do you know that Na. Cl is soluble in water? • What about KBr? Is it soluble? • What about Ag. Cl? • You can tell if a salt will be soluble or insoluble in water by looking up the cation and the anion on a solubility chart
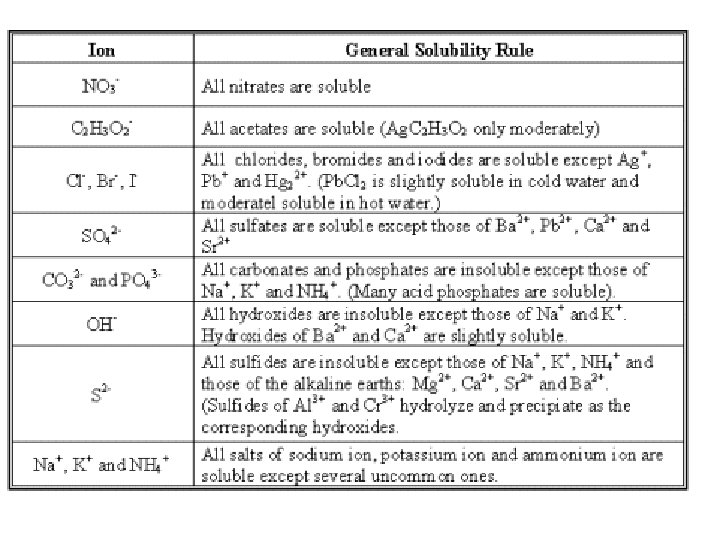

Problems 1) Is the CO 2, which is dissolved in soda, more or less soluble when you open the can? Why? 2) Which of the following is probably Ni(OH)2 in water? 3) Which is most likely Na 2 SO 4 in water?

Problems Which of the following salts is soluble in water? Which is insoluble? 1) Na. Cl 2) KBr 3) Ag. Cl

KBr Na. Cl Ag. Cl

Problems Which of the following salts will be soluble in water? Which will be insoluble? 1) Ag 2 O 2) Cd. S 3) Fe(OH)2 4) Na 2 SO 4 5) KCl

Cd. S Ag 2 O Fe(OH)2 Na 2 SO 4 KCl

Concentrations • Concentration = amount solute/amount solution • There are various ways to express concentration • The most common way to express concentration is Molarity (M) • Molarity = moles solute/liters solution • Units: mol/L

Problems 1) You dissolve 5. 66 g KBr in enough H 2 O to make 27 m. L of solution. What is the molarity of your solution? 2) You dissolve 2. 45 mg of Cu. SO 4 in enough water to make 55. 5 m. L of solution, what is your concentration? 3) If you have 60. 0 m. L of a 0. 988 M Na. Cl solution, how much Na. Cl did you start with?

• Mass-volume percent (% m/v): mass of the solute divided by the volume of solution and multiplied by 100 • % m/v =(mass solute)/(volume solution) x 100 • Also defined as mass of solute per 100 ml of solution • Usually expressed in g/ml Problems 1) You dissolve 5. 66 g KBr in enough H 2 O to make 27 m. L of solution. What is the mass/volume percent for the previous solution? 2) You dissolve 4. 44 g Na. Cl in enough water to make 0. 075 L of solution. What is the massvolume percent? 3) You have a % m/v of 3. 98 Na. Br in 350 ml of solution. How much Na. Br was used to make this solution?

• Percent by Mass (%m/m): mass of solute in mass of solution multiplied by 100 • Mass % = mass solute/mass solution x 100 • Remember: mass solution=mass solute + mass solvent • Also defined as the number of grams of solute per 100 g of solution Problems 1) You make a solution by dissolving 7. 55 g Na. Cl in 52. 4 g H 2 O. What is the mass % of solute in this solution? 2) You make a solution by dissolving 2. 45 g KCl in 20. 6 m. L of water. What is the mass % of solute in this solution? 3) You have a %m/m of 10. 0% sucrose. If you used 25. 0 g sucrose to make the solution, how much solvent (water) did you use?

• Percent by volume (% v/v): volume of solute divided by volume of solution multiplied by 100 • VP = volume solute/volume solution x 100 • Units of volume must be the same for both solute and solvent Problems 1) You dissolve a 5 cm 3 cube of sugar in 20 m. L of water. What is your volume percent of solute in this solution? 2) Your bottle of Jack Daniels says that the alcohol content is 14%. What does this mean?

More Problems 1) Your child has an ear infection and is given a suspension of 5. 0 % (m/v) amoxicillin. How many grams of the antibiotic is she given in 10 m. L? 2) How many milliliters of a 4. 0% (m/v) drug are needed if the patient is to receive 0. 250 g?

Dilutions • Dilution: the process by which more solvent is added to a solution in order to lower the concentration • C 1 V 1 = C 2 V 2 • C = Concentration • V = Volume

Problems 1) Joan has 50 m. L of a 0. 498 M glucose solution. She’d like to dilute this to a 0. 250 M glucose solution. What will her final volume be? 2) A nurse wants to prepare a 1. 0% (m/v) silver nitrate solution from 24 m. L or a 3. 0% stock solution of silver nitrate. How much water should be added to the 24 m. L? 3) Mary has a 2. 5 L bottle of a 0. 989 M Na. OH solution. She would like to make 525 m. L of a 0. 755 M Na. OH solution. How will she do this?

Mixtures • Heterogeneous Mixture: a mixture in which the particles of each component remain separate and can be observed as individual substances – See distinct phases – See interface – Solute particle size above 200 nm


• Homogeneous Mixture: a mixture in which the composition is the same throughout – Only one phase present – Cannot see interface – Particle size less than 1 nm – Often called “solutions”


Colloids • Mixtures with properties intermediate between heterogeneous and homogeneous mixtures • Particles tend to be small enough to pass through filters, but too large to pass through semipermeable membranes (1 nm-200 nm) • Particles are large enough to scatter light, producing a phenomenon called the Tyndall effect • Examples: Jell-O, Fog, Dust in air, Mayo

The Tyndall Effect • Named after the Irish scientist John Tyndall • Describes the light scattering effect cause by particles in a colloid – Can see the beam of light – Can see the particles in the colloid • Used to distinguish colloids from other types of mixtures

Suspensions • Heterogeneous mixtures which contain particles which are large enough to be trapped by filters and semipermeable membranes, but small enough to stay suspended for a while before settling out. • Examples: Muddy water, liquid medications, paint – Most things that direct you to shake or stir before using

Osmosis • The movement of water, through a semipermeable membrane, from regions of low solute concentration to regions of higher solute concentration • Spontaneous • The membrane must be permeable to the solvent, but not the solute
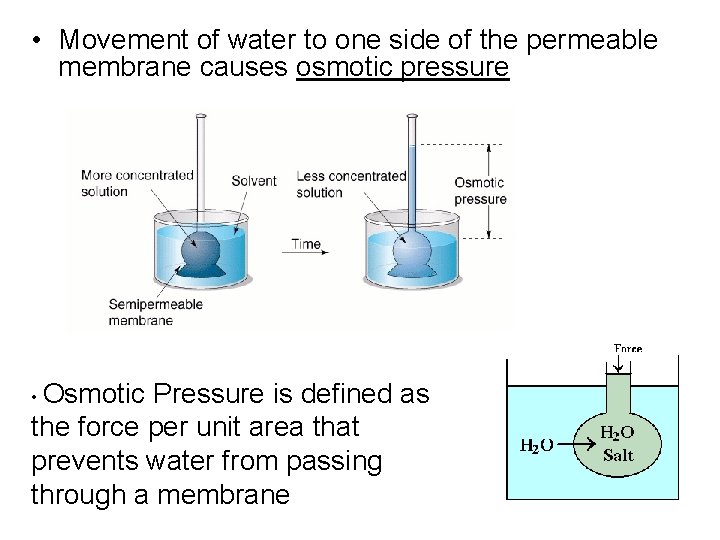
• Movement of water to one side of the permeable membrane causes osmotic pressure • Osmotic Pressure is defined as the force per unit area that prevents water from passing through a membrane
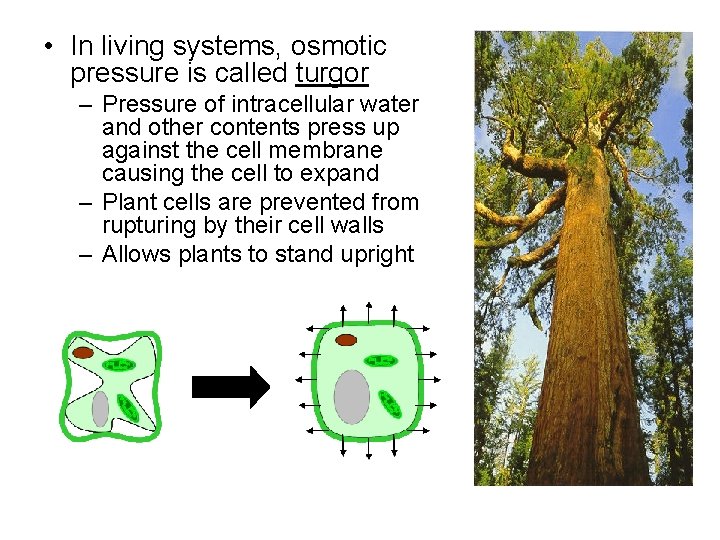
• In living systems, osmotic pressure is called turgor – Pressure of intracellular water and other contents press up against the cell membrane causing the cell to expand – Plant cells are prevented from rupturing by their cell walls – Allows plants to stand upright
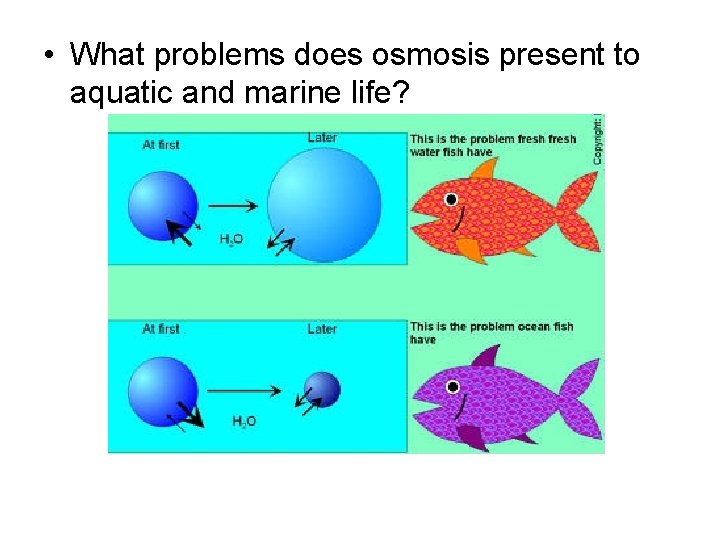
• What problems does osmosis present to aquatic and marine life?
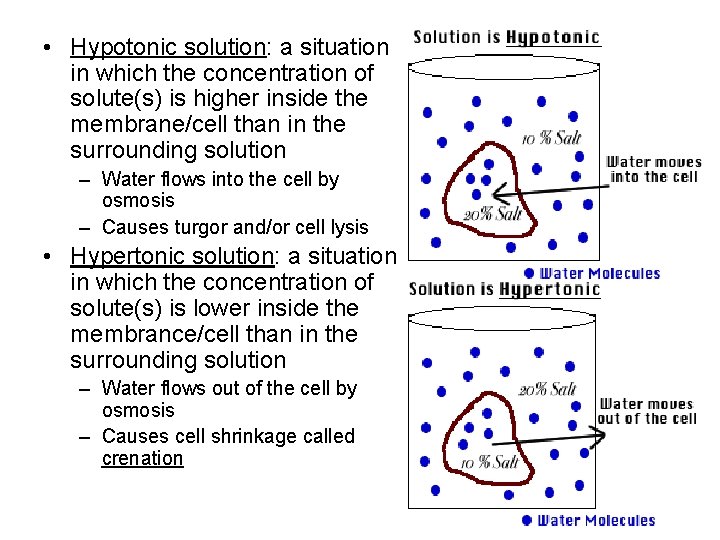
• Hypotonic solution: a situation in which the concentration of solute(s) is higher inside the membrane/cell than in the surrounding solution – Water flows into the cell by osmosis – Causes turgor and/or cell lysis • Hypertonic solution: a situation in which the concentration of solute(s) is lower inside the membrance/cell than in the surrounding solution – Water flows out of the cell by osmosis – Causes cell shrinkage called crenation
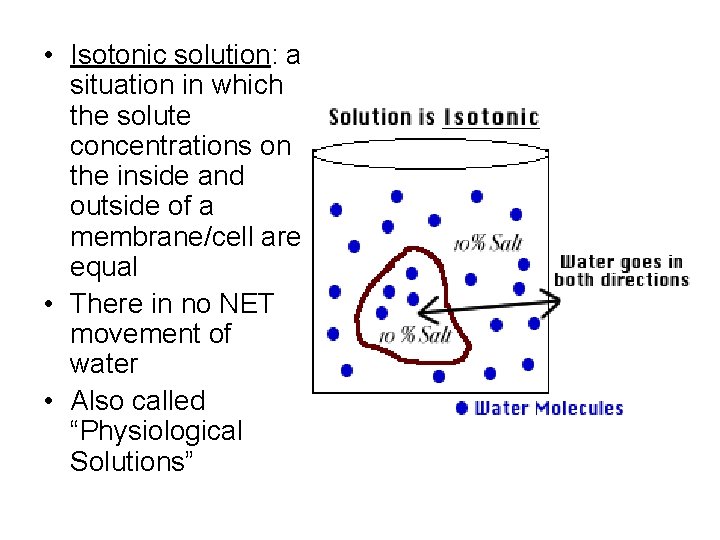
• Isotonic solution: a situation in which the solute concentrations on the inside and outside of a membrane/cell are equal • There in no NET movement of water • Also called “Physiological Solutions”
- Slides: 47