Unit 2 4 Rearrangement of Atoms in Chemical











































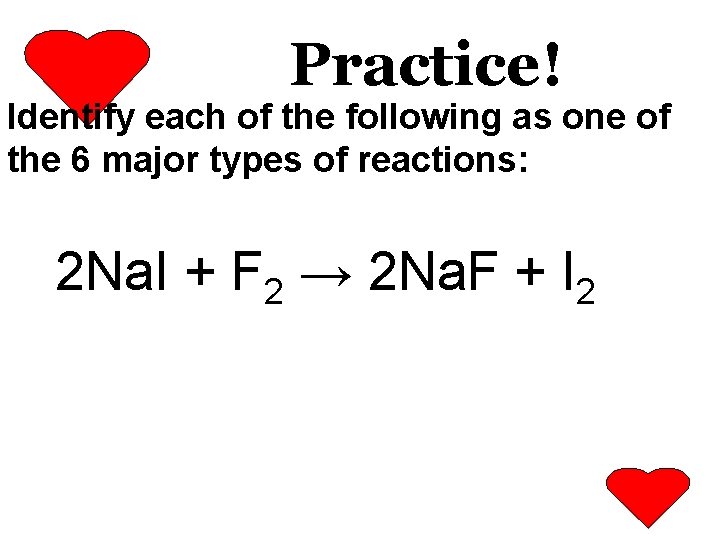














- Slides: 64

Unit 2 -4: Rearrangement of Atoms in Chemical Reactions

Types of Chemical Reactions • All chemical reactions follow the same basic formula: Reactants Products

In grade 9 you learned that: • If energy is required for a reaction to occur: Reactants + energy Products - Then the reaction is classified as ENDOTHERMIC • If energy is released or produced during a reaction: Reactants Products + energy - Then the reaction is classified as EXOTHERMIC

• Reactions can be further classified based on HOW the reactants combine and HOW the products form • There are 6 common types of reactions that you need to be able to identify!

Classification of Chemical Reactions: The Soap-Opera of Chemistry

6 Main types of reactions • Synthesis- “Love at first sight” • Decomposition- “The big break-up” • Single Replacement- “My best friends girlfriend” • Double Replacement- “Revenge” Also, Neutralization • Combustion- “The player”

Synthesis- “Love at first sight” I Ilike Can I get your Hey your braces hat Harry number Hey Sally Basic equation: A+B AB

Synthesis- “Love at first sight” Two individual elements combine to produce a compound. • Example: 2 Na + Cl 2 2 Na. Cl

Practice: Mg + N 2

Practice: Al + F 2

Practice: K + O 2

Practice: Cd + I 2

Practice: Cs + P 4

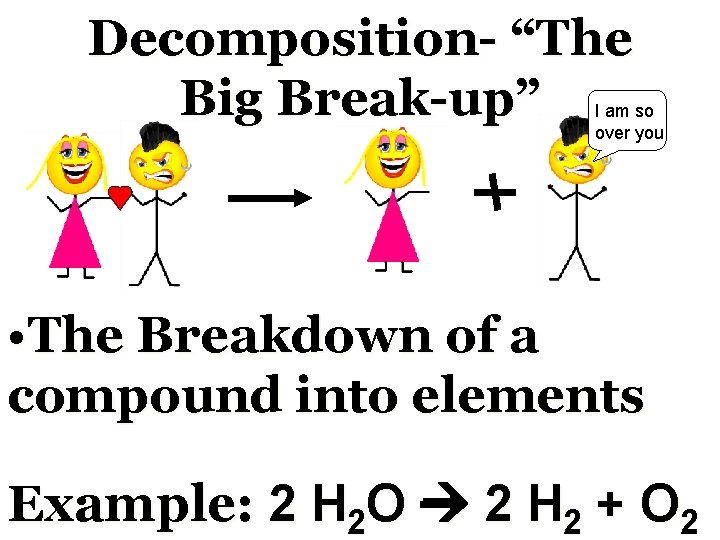
Decomposition- “The Big Whatever Break-up” We are I was Im leaving washing my hair Where were you last night But, baby I love you Through I don’t believe you Basic Equation: AB A+B

Decomposition- “The Big Break-up” I am so over you • The Breakdown of a compound into elements Example: 2 H 2 O 2 H 2 + O 2

Practice: Au. Cl 3

Practice: K 2 O

Practice: Mg. F 2

Practice: Ca 3 N 2

Practice: Cs. I

Single Replacement- “My Best Friends Girlfriend” Hey, Baby. This Hey Mark, here comes How you Oh Pierre! Mark your friend Pierre doing? Im leaving you Man, Marks Girl is pretty Hot! Stinks! Basic Eqn: AB + C AC + B Or AB + C CB + A

Single Replacement- “My Best Friends Girlfriend” • “Free” element must be the more reactive element for a reaction to occur ( Pierre is more suave then Mark) • Basically, a compound and element react to produce a different compound and element • Example: 2 K + 2 HCl 2 KCl + H 2

Practice: Pb. Cl 4 + Al

Practice: Na + Cu 2 O

Practice: Cu. F 2 + Mg

Practice: Cl 2 + Cs. Br

Practice: Be + Fe(NO 3)2

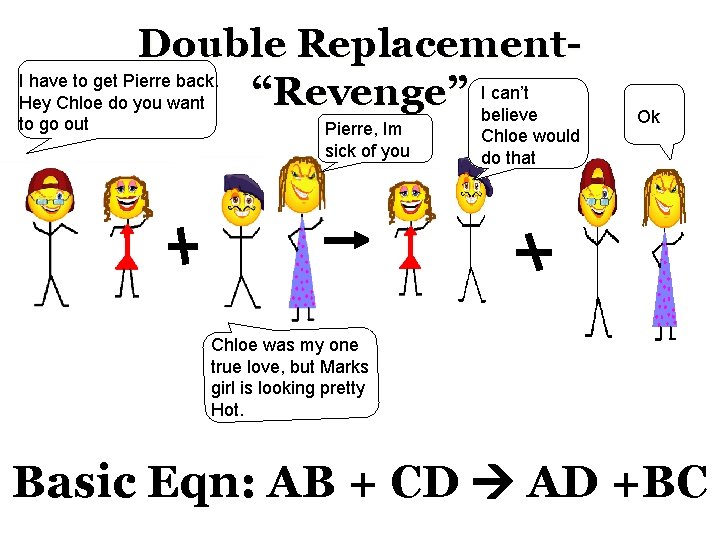
Double Replacement. I have to get Pierre back. can’t “Revenge” Ibelieve Hey Chloe do you want to go out Pierre, Im sick of you Chloe would do that Ok Chloe was my one true love, but Marks girl is looking pretty Hot. Basic Eqn: AB + CD AD +BC

Double Replacement“Revenge” • Two compounds react to produce two different compounds. Ex: 2 KI(aq) + Pb(NO 3)2(aq) Pb. I 2(s) + 2 KNO 3(aq)

Practice: Ca. S + Na. OH

Practice: K 3 PO 4 + Mg. I 2

Practice: Sr. Cl 2 + Pb(NO 3)2

Practice: Al. Cl 3 + Cu. NO 3

Practice: Ag. NO 3 + Na 2 Cr. O 4

Psst…. • Neutralization Reactions are a specific type of Double Replacement Reaction!! ACID + BASE SALT + WATER • H 2 SO 4 + Ca(OH)2 → Ca. SO 4 + 2 H 2 O Acid Base Salt Water

Practice: HBr + Na. OH

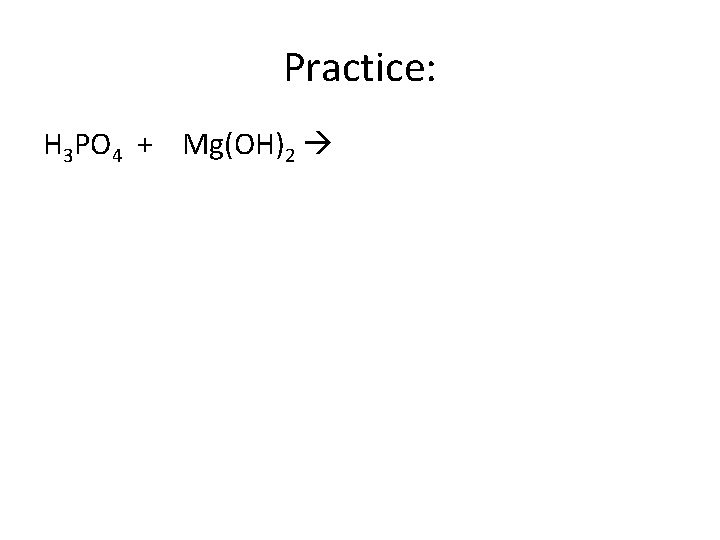
Practice: H 3 PO 4 + Mg(OH)2

Practice: HCl + Pb(OH)2

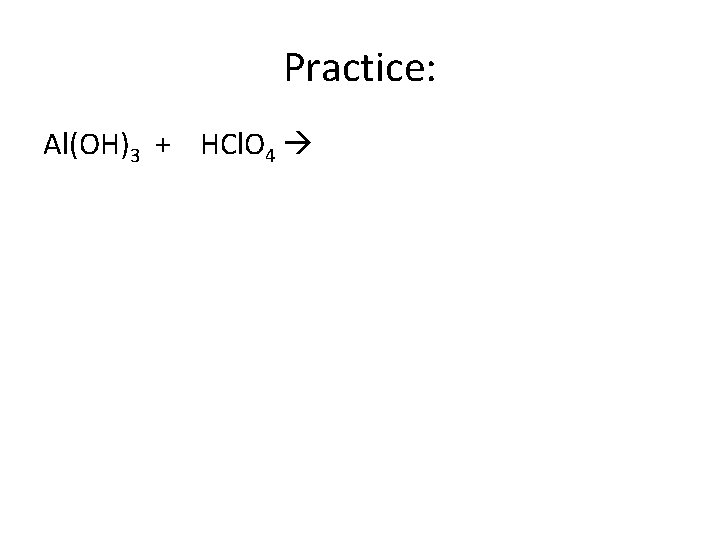
Practice: Al(OH)3 + HCl. O 4

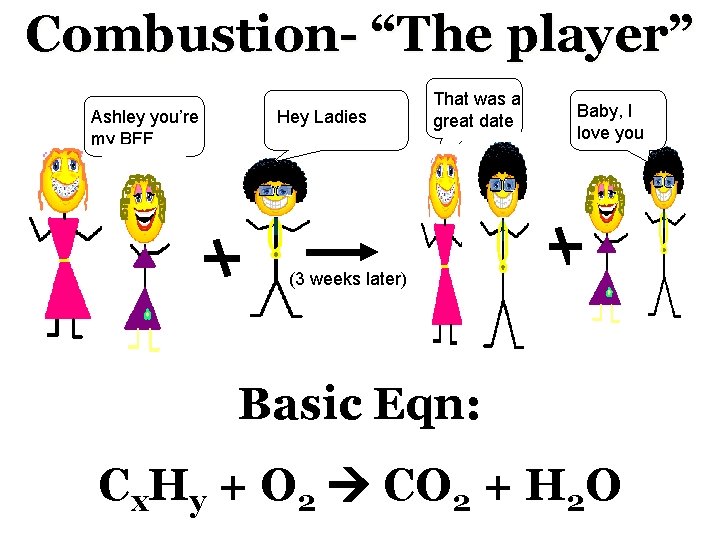
Combustion- “The player” Ashley you’re my BFF Hey Ladies That was a great date Baby, I love you (3 weeks later) Basic Eqn: Cx. Hy + O 2 CO 2 + H 2 O

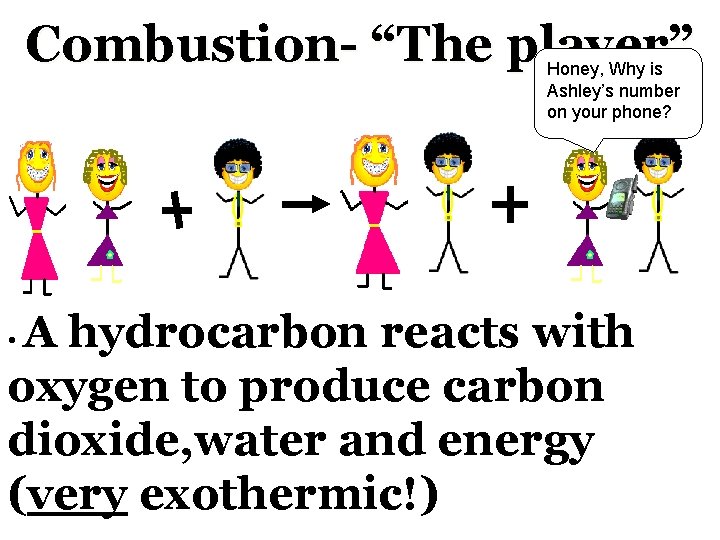
Combustion- “The player” Honey, Why is Ashley’s number on your phone? A hydrocarbon reacts with oxygen to produce carbon dioxide, water and energy (very exothermic!) •

Practice: C 3 H 8 + O 2

Practice: C 4 H 10 + O 2

Practice: C 2 H 4 + O 2

Practice: C 6 H 12 O 6 + O 2

Practice: C 12 H 22 O 11 + O 2

Practice! Identify each of the following as one of the 6 major types of reactions: Pb. O 2 → Pb + O 2

Practice! Identify each of the following as one of the 6 major types of reactions: Mg + Ag. NO 3 → Mg(NO 3)2 + Ag

Practice! Identify each of the following as one of the 6 major types of reactions: H 2 + Cl 2 → 2 HCl
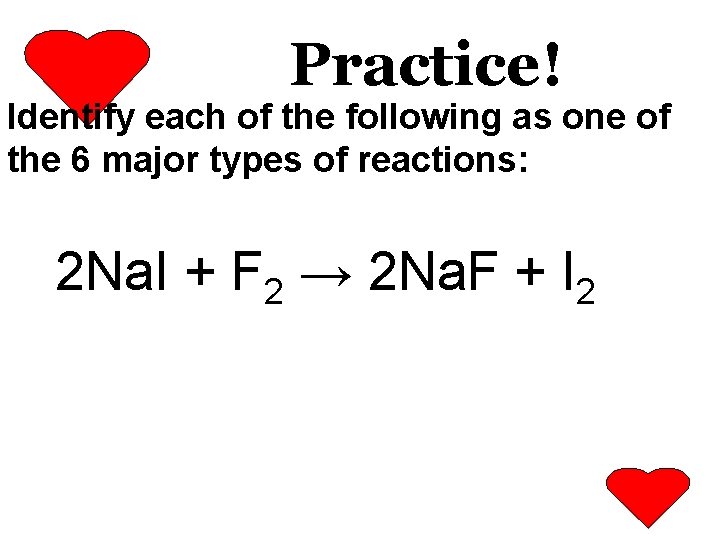
Practice! Identify each of the following as one of the 6 major types of reactions: 2 Na. I + F 2 → 2 Na. F + I 2

Practice! Identify each of the following as one of the 6 major types of reactions: C 12 H 22 O 11 + 12 O 2 → 12 CO 2 + 11 H 2 O

Practice! Identify each of the following as one of the 6 major types of reactions: 2 HI → H 2 + I 2

Practice! Identify each of the following as one of the 6 major types of reactions: 2 Li. Cl + Br 2 → 2 Li. Br + Cl 2

Practice! Identify each of the following as one of the 6 major types of reactions: H 2 SO 4 + 2 Na. OH → Na 2 SO 4 + 2 H 2 O

Practice! Identify each of the following as one of the 6 major types of reactions: Silver + chlorine → silver chloride

Practice! Identify each of the following as one of the 6 major types of reactions: Sulfur dioxide → sulfur + oxygen

Practice Problems Page 265 Check Your Understanding Page 271 #1 -6

Answers to CYU pg. 271 1. a) Neutralization b) Synthesis c) Double replacement d) Synthesis e) Combustion f) Double replacement g) Single replacement (metal) h) Neutralization i) Decomposition j) Single replacement (metal)

Answers to CYU pg. 271 2. Combustion reactions will always have O 2 as one of the reactants. 3. 1 a of these questions is both a neutralization and a double replacement reaction. 4. a) Synthesis: 4 Na + O 2 2 Na 2 O b) Double Replacement: Na 2 SO 4 + Ca. Cl 2 2 Na. Cl + Ca. SO 4

Answers to CYU pg. 271 4. c) Combustion: C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O d) Neutralization: H 2 SO 4 + 2 KOH K 2 SO 4 + 2 H 2 O e) Decomposition: 2 Al. Cl 3 2 Al + 3 Cl 2 f) Single Replacement: 3 Cd + 2 Au(NO 3)3 3 Cd(NO 3)2 + 2 Au

Answers to CYU pg. 271 4. g) Double replacement: Sr(OH)2 + Pb. Br 2 Sr. Br 2 + Pb(OH)2 h) Combustion: C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O i) Synthesis: 2 N 2 + 3 O 2 2 N 2 O 3 j) Single replacement: 2 HNO 3 + Zn Zn(NO 3)2 + H 2

Answers to CYU pg. 271 5. a) Synthesis: 6 Na + N 2 2 Na 3 N b) Decomposition: 2 Al. F 3 2 Al + 3 F 2 c) Single Replacement: 3 Cu. SO 4 + 2 Al Al 2(SO 4)3 + 3 Cu d) Double Replacement: Ca. I 2 + Pb(NO 3)2 Ca(NO 3)2 + Pb. I 2

Answers to CYU pg. 271 5. e) Combustion: 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O f) Double replacement: Ag. NO 3 + Na. Br Ag. Br + Na. NO 3 g) Single replacement (non-metal): 2 Cs. I + Cl 2 2 Cs. Cl + I 2 h) Neutralization: HCl + Na. OH Na. Cl + H 2 O

Answers to CYU pg. 271 5. i) Double replacement: K 2 Cr 2 O 7 + 2 Ag. NO 3 Ag 2 Cr 2 O 7 + 2 KNO 3 j) Combustion: C 5 H 10 O 5 + 5 O 2 5 CO 2 + 5 H 2 O 6. 2 Fe + 3 Cl 2 2 Fe. Cl 3