Packet 4 The Periodic Table Reference Table Periodic

Packet #4 The Periodic Table Reference Table: Periodic Table & Table S “The Elements”

Development of Periodic Table n Dmitri Mendeleev, a Russian chemist is given credit for being the first scientist to arrange elements in a usable manner n Mendeleev observed that when the elements were arranged in order of increasing atomic mass, similar chemical and physical properties appeared at regular intervals n Mendeleev developed the first periodic table according to increasing atomic mass

Dmitri Mendeleev

Modern Periodic Table n The modern periodic table is not arranged by increasing atomic mass, but rather increasing atomic number n Henry Mosely an English scientist used X-rays to identify the atomic number of the elements n He arranged elements according to increasing atomic number which led to the modern atomic law

Periodic Law n States that the properties of elements are periodic functions of their atomic numbers n This means that when elements are arranged by increasing atomic #, those with similar properties will be located at regular intervals (in the same group) n The placement of an element on the Periodic Table gives an indication of the chemical and physical properties of that element
Periods

Periods n Periods are horizontal rows on the periodic table n Period 1 contains H and He n The number at the beginning of the period indicates the energy level where valence electrons are located for atoms in that period
Groups or Families

Groups or Families n Vertical Columns n Elements in a group have the same number of valence electrons. n Elements in a group have similar properties and react similarly because they have the same number of valence electrons (this is known as Periodic Law)

Common Group Names Group 1: Alkali Metals Group 2: Alkaline Earth Metals Groups 3 -12: Transition Elements (metals) Group 17: Halogen Group 18: Noble Gases

Classifying Elements • Elements are classified as either: • Metals • Non-metals • Metalloids • Noble Gases • All elements to the left of it are metals. • All elements to the right are non-metals except for Group 18 that are known as Noble Gases, and • All elements touching the zigzag are called metalloids. • Aluminum an Polonium are exceptions, it is a metal although it touches the zig zag
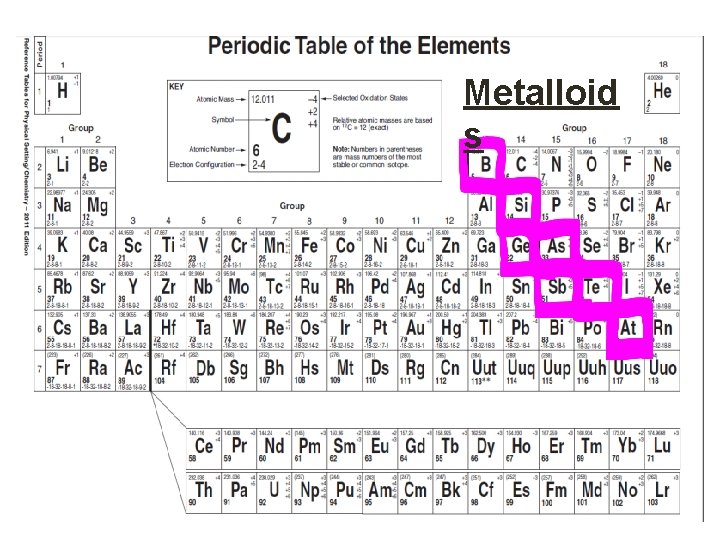
Metalloid s
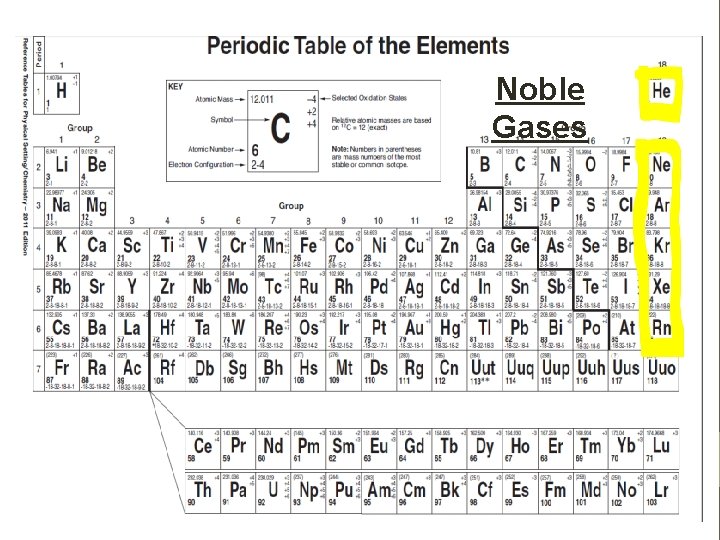
Noble Gases
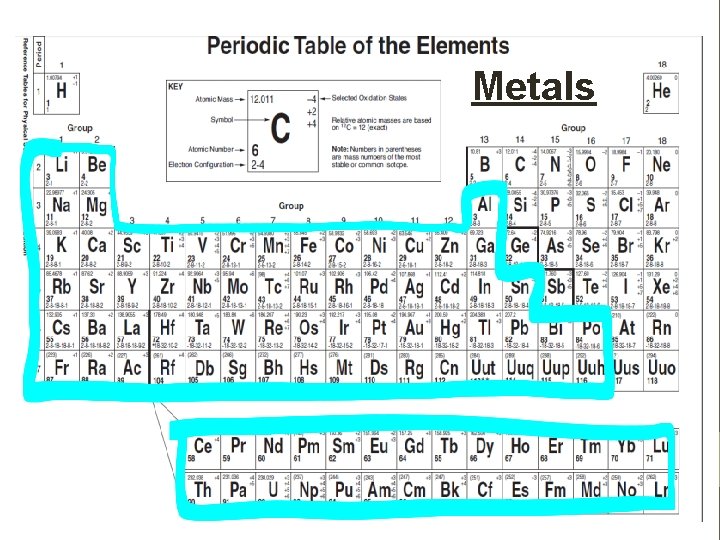
Metals
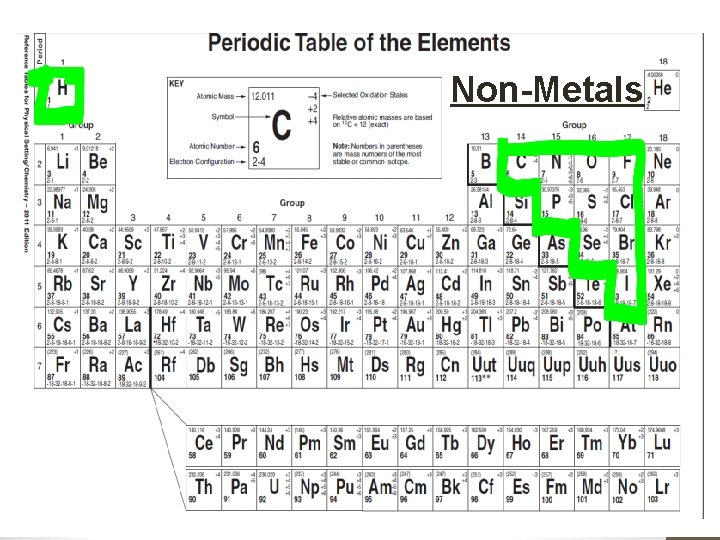
Non-Metals

Classifying Elements • The most reactive metal on the PT is francium. • The most reactive non-metal on the PT is fluorine. • Representative Elements: Groups 1, 2, and 13 -17 – these groups follow a trend we will soon discuss. • Transition Elements: Groups 3 -12 – these elements do NOT follow the general trends – more than 1 charge produce a COLOR!!
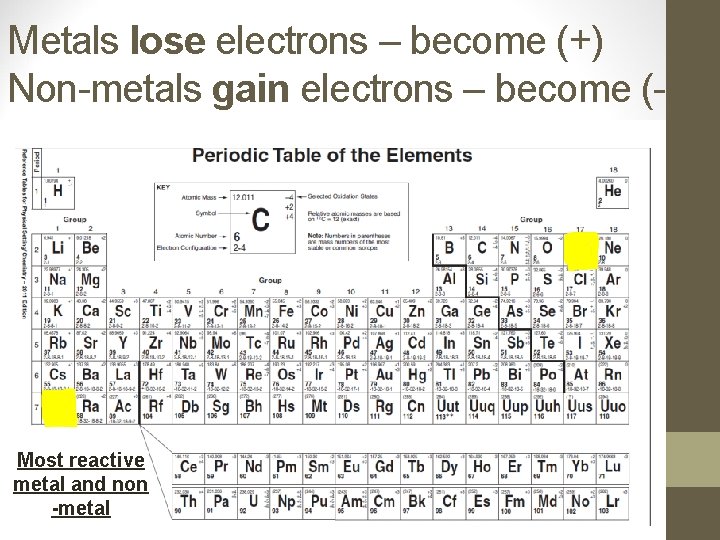
Metals lose electrons – become (+) Non-metals gain electrons – become (-) Most reactive metal and non -metal
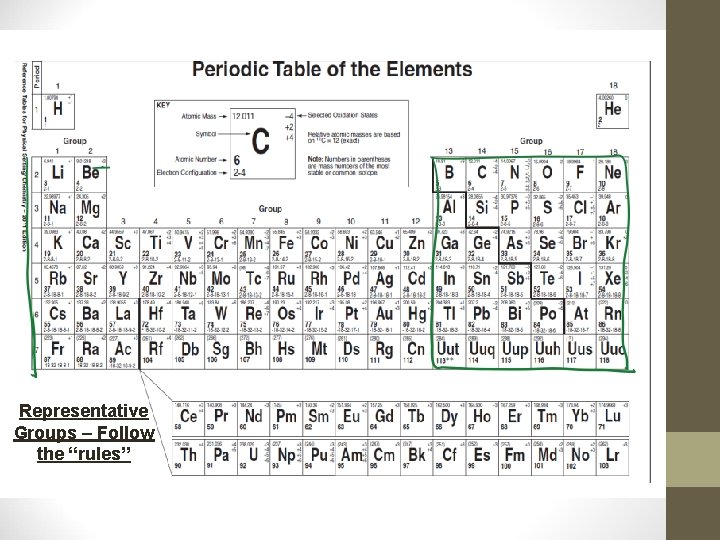
Representative Groups – Follow the “rules”
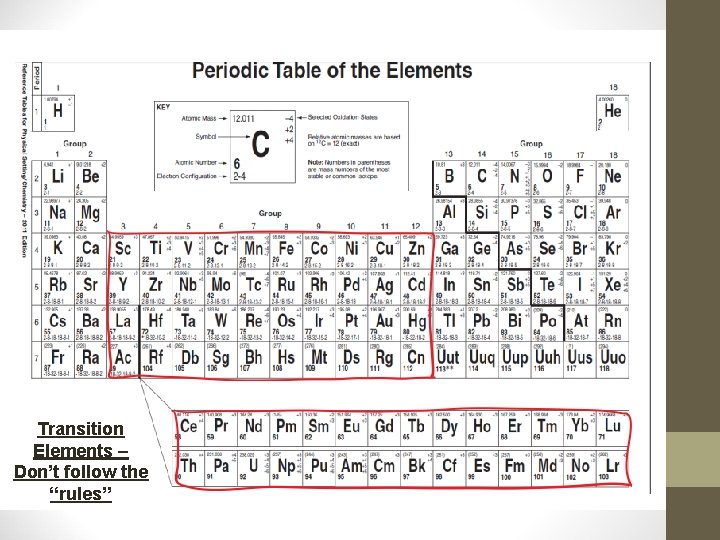
Transition Elements – Don’t follow the “rules”

• SOLIDS: Most of the elements of the PT • LIQUIDS: Hg & Br • GASES: 11 TOTAL (always reactive)H 2, N 2, O 2, F 2, Cl 2 (typically non-reactive)Noble Gases
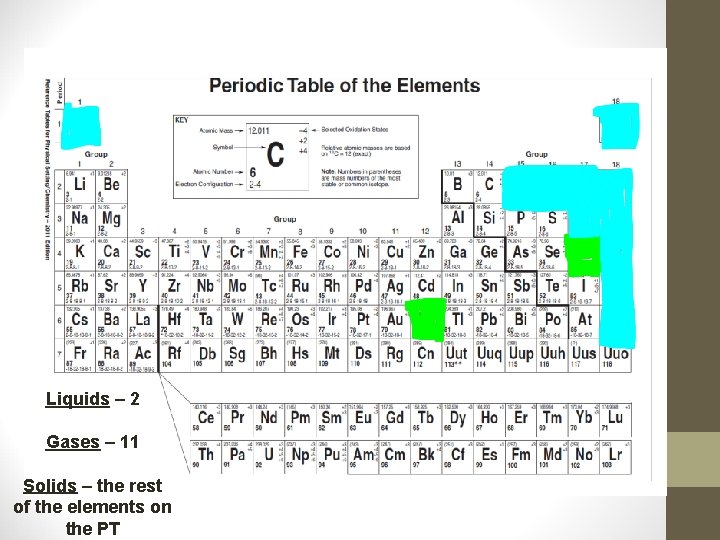
Liquids – 2 Gases – 11 Solids – the rest of the elements on the PT

Properties of Elements Physical Properties of Metals • Metallic luster (shine) • Malleable (can be made into sheets) • Ductile (can be made into wires) • Metals are good conductors of heat and electricity • Most metals are solids at room temperature or STP which stands for standard temperature and pressure (exception is mercury which is liquid at room temperature).

Properties of Elements • Elements in Group 1 form compounds that are soluble like sodium chloride, potassium sulfide and lithium sulfate. • Elements in Group 2 form compounds that some of which are soluble and some are not. • Density of Group 2 is greater that Group 1 (see table S). • Group one can be cut with a knife, elements in Group 2 are harder, and elements in Group 3 -12 (transition elements) are much harder.

Properties of Elements Physical Properties: Non-metals, Metalloids, and Noble Gases • Non-metals: Lack metallic luster and are brittle in the solid phase; they are poor conductors of heat and electricity; non-metals are usually gases, molecular solids or network solids (exception is bromine which is a liquid at room temperature). • Metalloids: Have some properties of both metals and nonmetals. • Noble Gases: Are all gases. Have 8 valence electrons (helium is the exception with 2).

Properties of Elements Chemical Properties • Group 1 & Group 2: Elements in Group 1 react faster than elements in group 2 because Group 1 only has one valence electron and Group 2 • Non-metal Groups (13 -17): Elements in Group 17 react very fast and occur in compounds only; they need to gain only 1 electron to become stable. • Noble Gases: He, Ne, and Ar do not react (these are known as small noble gases) while Kr, Xe, and Rn can react with fluorine and oxygen (these are known as heavier noble gases).

• Reactivity of Metals in Water

Periodic Trends (Table S) Ionization Energy (IE): The energy required to lose an electron. • The lower the IE, the easier it is to lose an electron • Remember, metals want to lose electrons, and non-metals want to gain


Ionization energy down a group As you go down a group ionization energy decreases because the number of PELS increase, and so the valence electrons are further away from the positive nucleus; there is less attraction which results in less energy needed to remove electrons n Look at the ionization energies on Table S Li 520 Na 496 K 419 Rb 403 n

Ionization Energy across a Period n As you go from left to right ionization energy increases because as you go across a period, the number of valence electrons increase by 1, and therefore the less likely an element wants to lose an electron (on the contrary, they want to gain them). n Look at ionization energy values on Table S Li (520) Ne (2081)

Periodic Trends (Table S) Electronegativity (EN): The ability to attract an electron. • The larger the EN the more the atom attracts electrons • Remember, metals want to lose electrons, and non-metals want to gain


Electronegativity energy down a group • As you go down a group EN decreases because as you go down a group, the number of PELS increase, and therefore the outermost shell of electrons is further from the positively charged nucleus making the attraction for electrons decrease. • Look at the electronegativity values on Table S Li Na K Rb 1. 0. 9. 8. 8

Electronegativity across a Period • As you go from left to right EN increases because as you go across a period, the number of valence electrons increase by 1, and therefore the more likely an element wants to gain an electron (metal non-metal) • Look at ionization energies on Table S Li (1. 0) F (4. 0)

Periodic Trends (Table S) Atomic Radius (AR): Is the distance from the nucleus to the outer valence electrons.
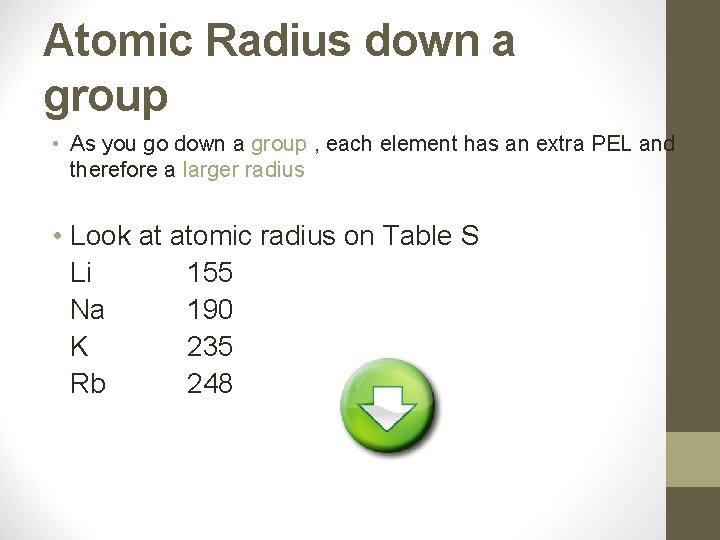
Atomic Radius down a group • As you go down a group , each element has an extra PEL and therefore a larger radius • Look at atomic radius on Table S Li 155 Na 190 K 235 Rb 248

Atomic Radius Across a Period • As you go across a period the radius decreases because as you go across a period the # of protons in the nucleus increases which increases the nucleus’ pull on the electrons. • Look at atomic radius on Table S Li (155) F (57)

Ionic Radius (IR) • The distance of the nucleus to the outermost valence electron in an ion. • Remember that an ion is a charged particle. Metals lose electrons and therefore are positively charged ions, and non-metals gain electrons and therefore are negatively charged ions.

Ionic Radius • RULE: Positive Ion (cation) LOSING AN ELECTRON MEANS THAT THE IONIC RADIUS IS LESS THAN THE ATOMIC RADIUS (LOSE = LESS) AR > IR • RULE: Negative Ion (anion) GAINING AN ELECTRON MEANS THAT THE IONIC RADIUS IS GREATER THAN THE ATOMIC RADIUS (GAIN = GREATER)

Allotropes Two or more forms of the same element that differ in their molecules. Allotropes have different properties. Oxygen has 2 allotropes: O 2 is the oxygen we breathe, and O 3 makes up the ozone layer. Carbon has many different allotropes which differ in arrangement of atoms • Diamond: every carbon bonded to 4 other carbons = very hard • Graphite: arranged in sheets or layers = lead pencils. • Coal: no definite pattern. • Buckminsterfullerence: rings of 5 and 6 carbon atoms, looks like the outside of a soccer ball






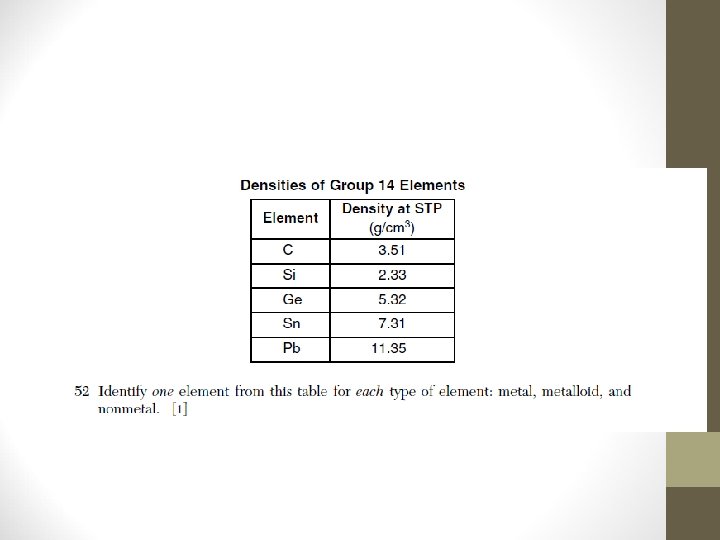




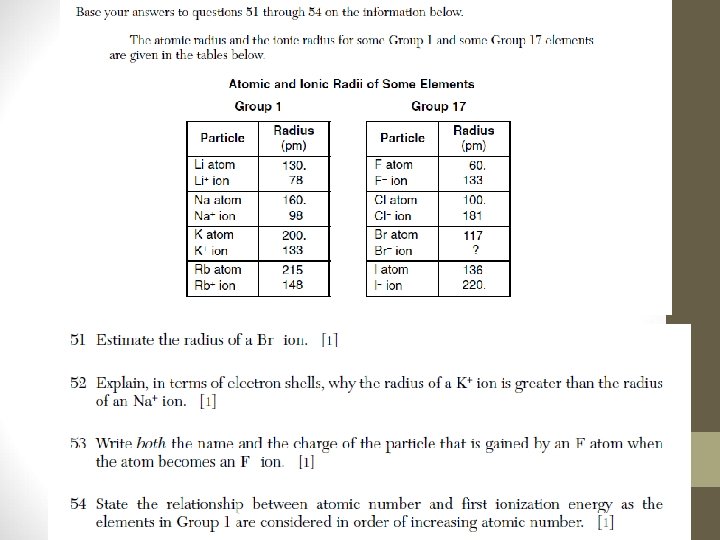

Review Questions 1) Which element is in Group 2 and Period 7 of the Periodic Table? A) radium B) manganese C) radon D) magnesium 2) On the Periodic Table, an element classified as a semimetal (metalloid) can be found in A) Period 4, Group 15 B) Period 3, Group 16 C) Period 2, Group 14 D) Period 6, Group 15

3) Which compound contains an alkali metal and a halogen? A) Rb. Cl B) Ca. Cl 2 C) Ca. S D) Rb 2 S 4) The pair of elements with the most similar chemical properties are A) S and Ar B) Mg and Ca C) Ca and Br D) Mg and S

5) As the atoms of the metals of Group 1 in the ground state are considered in order from top to bottom, the number of occupied principal energy levels A) remains the same B) decreases C) increases 6) The elements of Period 2 have the same A) atomic mass B) atomic number C) number of occupied sublevels D) number of occupied principal energy levels

7) Which atom has the largest atomic radius? A) Ca B) Mg C) Na D) K 8) As the elements of Group 16 are considered from top to bottom on the Periodic Table, the atomic radii A) decrease and the ionization energies decrease B) increase and the ionization energies decrease C) decrease and the ionization energies increase D) increase and the ionization energies increase

9) As a chlorine atom becomes a negative ion, the atom A) gains an electron and its radius increases B) loses an electron and its radius increases C) gains an electron and its radius decreases D) loses an electron and its radius decreases 10) Which of the following elements has the highest electronegativity? A) H B) K C) Ca D) Al

11) As a neutral sulfur atom gains two electrons, what happens to the radius of the atom? 12) Potassium ions are essential to human health. The movement of dissolved potassium ions, K+(aq), in and out of a nerve cell allows that cell to transmit an electrical impulse. What is the total number of electrons in a potassium ion?

13) The table below shows the EN of selected elements of Period 2 of the Periodic Table. (a) On the grid below, set up a scale for electronegativity on the y-axis. (b) Plot the data by drawing a best-fit line. (c) Using the graph, predict the electronegativity of nitrogen.
- Slides: 60