Section 2 Molecular Geometry Unit 10 Chemical Bonding

Section 2: Molecular Geometry Unit 10: Chemical Bonding
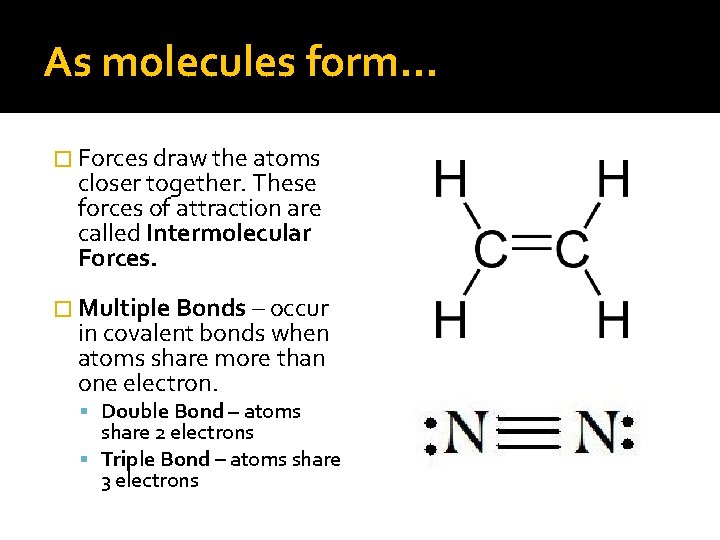
As molecules form… � Forces draw the atoms closer together. These forces of attraction are called Intermolecular Forces. � Multiple Bonds – occur in covalent bonds when atoms share more than one electron. Double Bond – atoms share 2 electrons Triple Bond – atoms share 3 electrons

Bond Strength vs Bond Length The more bonds between 2 atoms, the stronger the bond. The more bonds between atoms, the shorter the length of the bonds become Bond Strength Single Bond < Double Bond < Triple Bond Length Single Bond > Double Bond > Triple Bond

VSEPR Theory – “valence-shell electron-pair repulsion” States that repulsion between the sets of valence electrons causes sets to be placed as far apart as possible. Electrons have the same charge, which causes them to repel each other as far as possible Leads to unique angles between atoms in the molecule. ▪ Bond Angle – Angle made between two adjacent atoms in a molecule

VSEPR Theory Bending of molecules leads to one of these molecular shapes… Linear Trigonal-planar Tetrahedral Bent Trigonal-pyramidal Trigonal-bipyramidal Octahedral
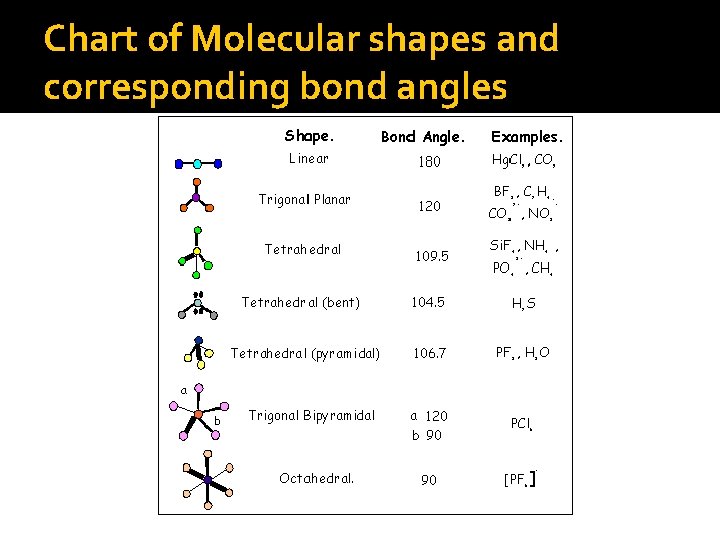
Chart of Molecular shapes and corresponding bond angles

Determining Polarity of a Molecule A molecule may be polar or non-polar, just like the individual covalent bonds You must consider 2 things when determining polarity of a molecule ▪ 1) The polarity of the individual bonds in the molecule ▪ 2) The shape or geometry of the molecule

Non-Polar Molecules There are 3 possible outcomes If all bonds are non-polar, then the whole molecule is non-polar regardless of its shape If there is symmetry in the molecule so that the polarity of the bonds cancels out, then the molecule is non-polar ▪ O=C=O The carbon-oxygen bond is a polar bond, but because they are exactly opposed to each other, the molecule is overall non-polar

Polar Molecules If there are polar bonds but there is no symmetry such that they cancel each other out, the overall molecule is polar ▪ Water is a typical example of this. The two O-H bonds are oriented in a V-shape, and so the don't cancel out ▪ Similarly, CH 3 Cl is also polar. It is the same shape as CCl 4 (see above), but now it doesn't have the same symmetry because there is only one C-Cl bond and the bonds don't cancel out anymore
- Slides: 9