The Periodic Table Periodic Table Dmitri Mendeleev 1834
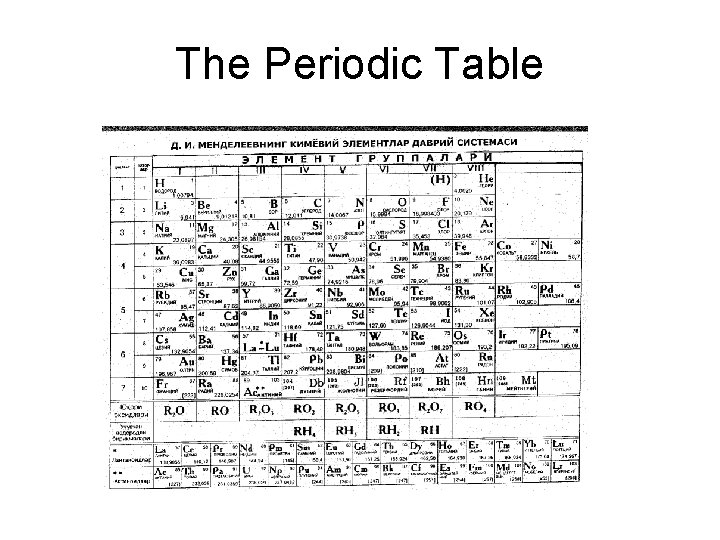
The Periodic Table
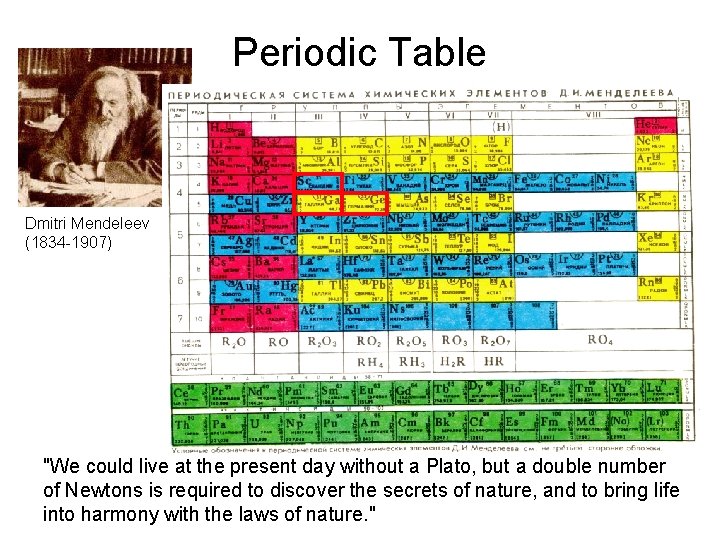
Periodic Table Dmitri Mendeleev (1834 -1907) "We could live at the present day without a Plato, but a double number of Newtons is required to discover the secrets of nature, and to bring life into harmony with the laws of nature. "
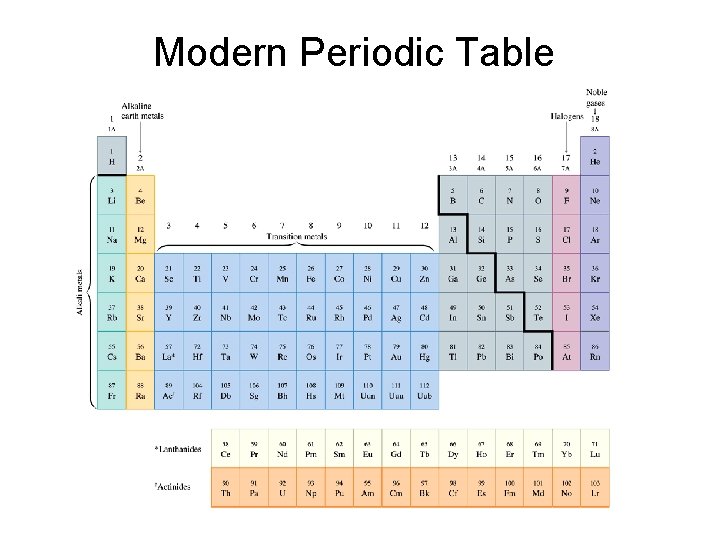
Modern Periodic Table
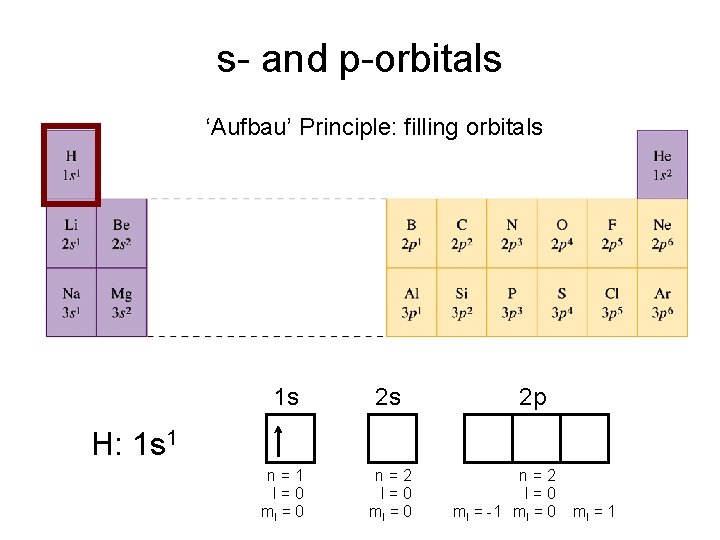
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s n=1 l=0 ml = 0 n=2 l=0 ml = 0 2 p H: 1 s 1 n=2 l=0 ml = -1 ml = 0 ml = 1
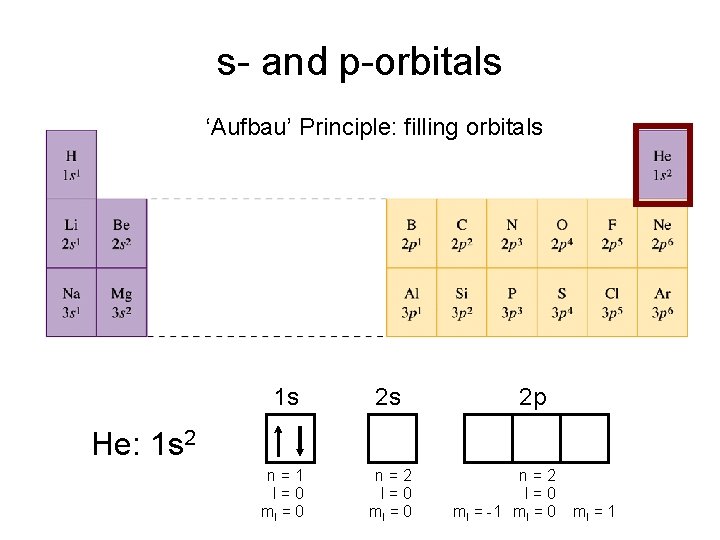
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s n=1 l=0 ml = 0 n=2 l=0 ml = 0 2 p He: 1 s 2 n=2 l=0 ml = -1 ml = 0 ml = 1
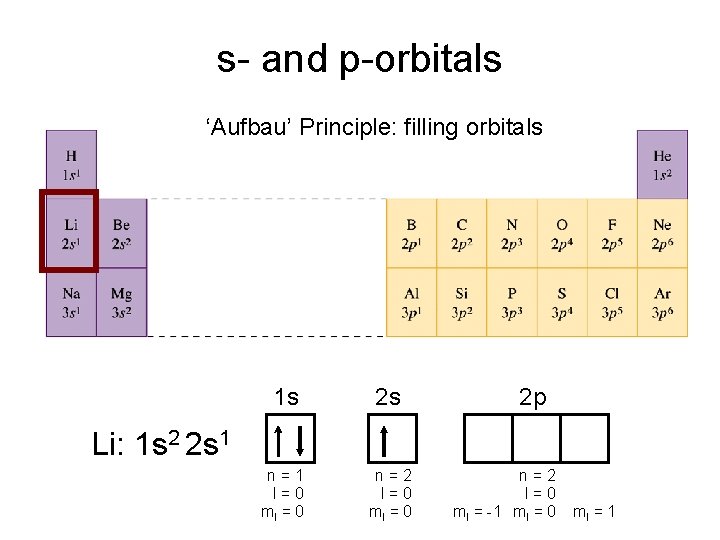
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s n=1 l=0 ml = 0 n=2 l=0 ml = 0 2 p Li: 1 s 2 2 s 1 n=2 l=0 ml = -1 ml = 0 ml = 1
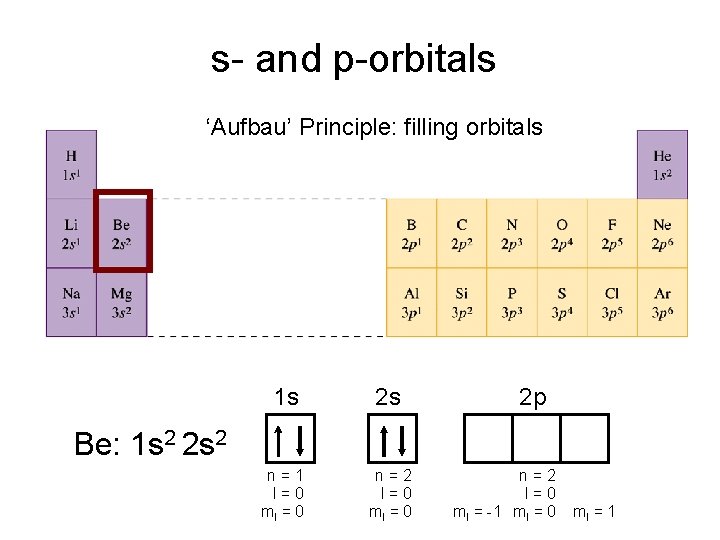
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s n=1 l=0 ml = 0 n=2 l=0 ml = 0 2 p Be: 1 s 2 2 s 2 n=2 l=0 ml = -1 ml = 0 ml = 1
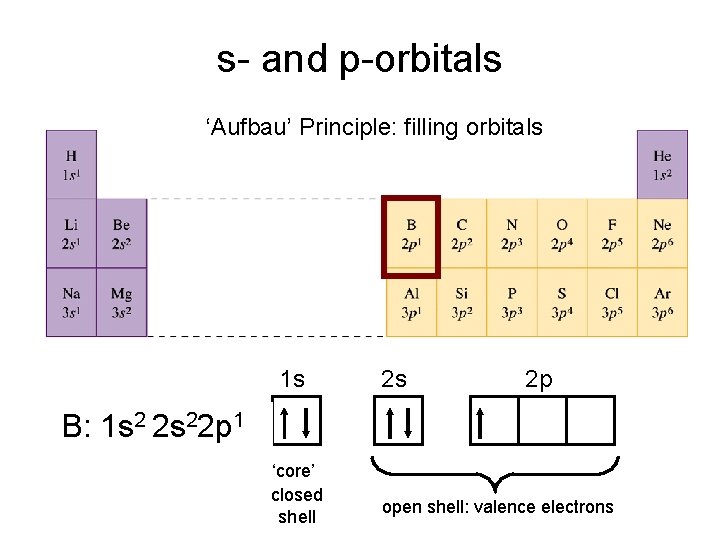
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s 2 p B: 1 s 2 2 s 22 p 1 ‘core’ closed shell open shell: valence electrons
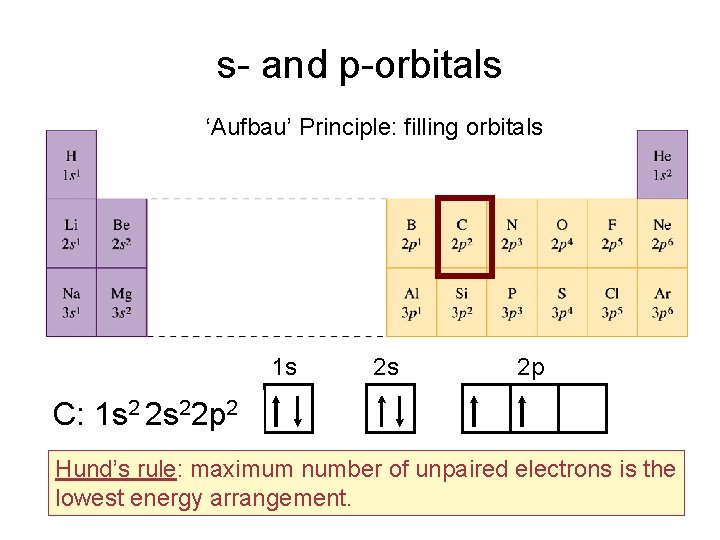
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s 2 s 2 p C: 1 s 2 2 s 22 p 2 Hund’s rule: maximum number of unpaired electrons is the lowest energy arrangement.
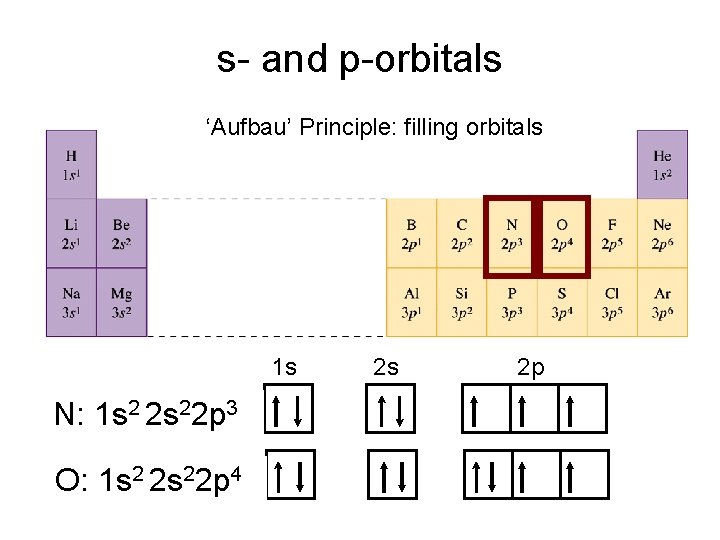
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s N: 1 s 2 2 s 22 p 3 O: 1 s 2 2 s 22 p 4 2 s 2 p
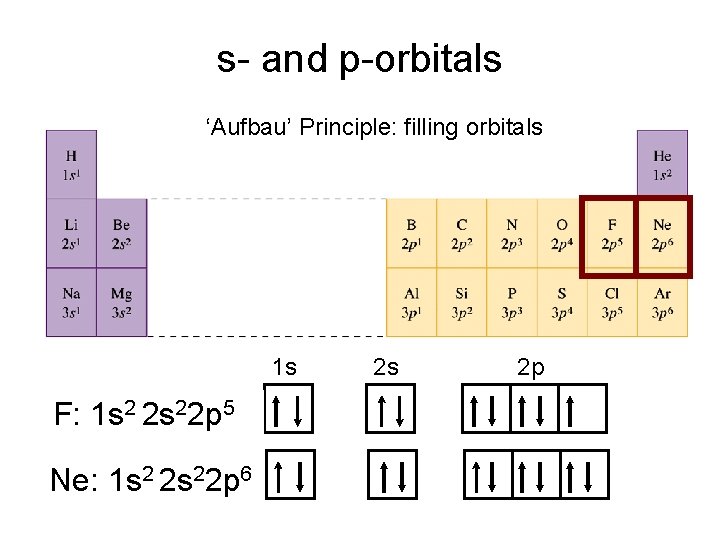
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals 1 s F: 1 s 2 2 s 22 p 5 Ne: 1 s 2 2 s 22 p 6 2 s 2 p
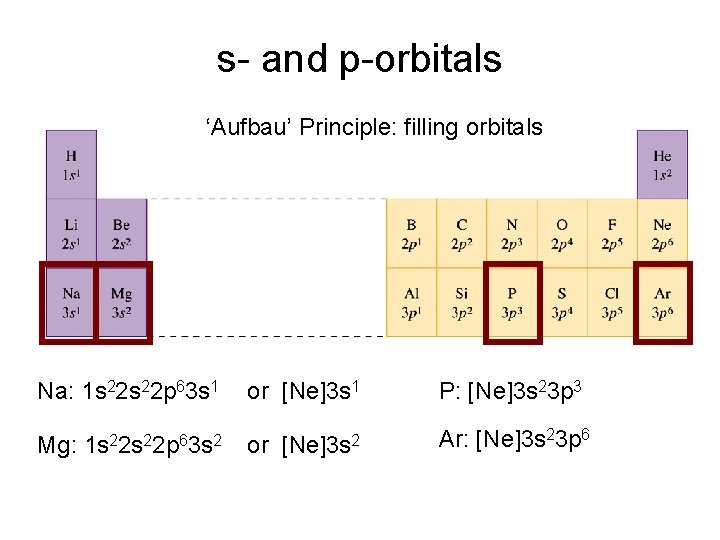
s- and p-orbitals ‘Aufbau’ Principle: filling orbitals Na: 1 s 22 p 63 s 1 or [Ne]3 s 1 1 s 22 p 63 s 2 [Ne]3 s 2 Mg: or P: [Ne]3 s 23 p 3 Ar: [Ne]3 s 23 p 6
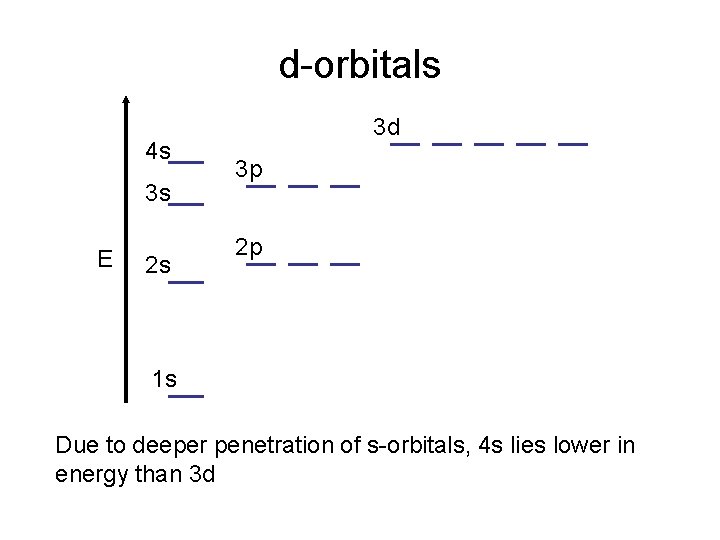
d-orbitals 4 s 3 s E 2 s 3 d 3 p 2 p 1 s Due to deeper penetration of s-orbitals, 4 s lies lower in energy than 3 d
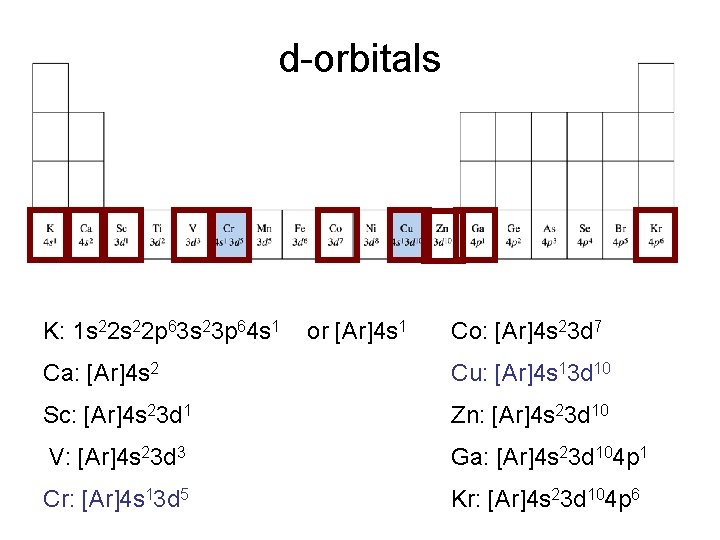
d-orbitals K: 1 s 22 p 63 s 23 p 64 s 1 or [Ar]4 s 1 Co: [Ar]4 s 23 d 7 Ca: [Ar]4 s 2 Cu: [Ar]4 s 13 d 10 Sc: [Ar]4 s 23 d 1 Zn: [Ar]4 s 23 d 10 V: [Ar]4 s 23 d 3 Ga: [Ar]4 s 23 d 104 p 1 Cr: [Ar]4 s 13 d 5 Kr: [Ar]4 s 23 d 104 p 6
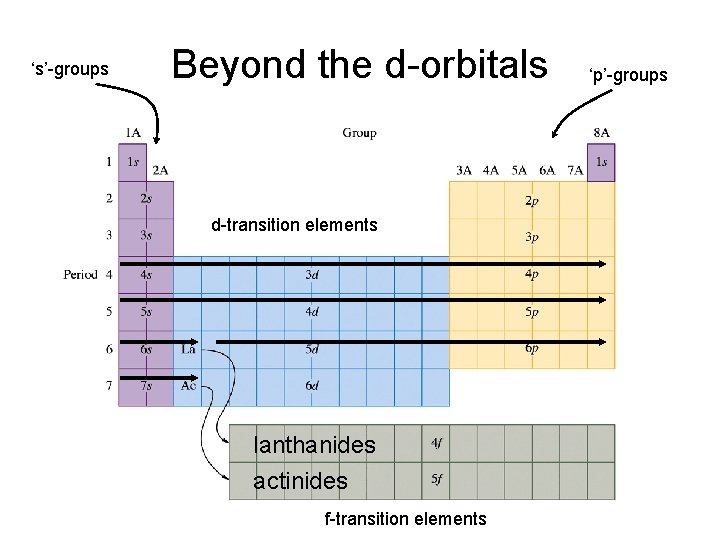
‘s’-groups Beyond the d-orbitals d-transition elements lanthanides actinides f-transition elements ‘p’-groups

Aufbau rules 1. Within a shell (n) the filling order is s>p>d>f 2. Within a subshell (l), lowest energy arrangement has the highest number of unpaired spin (Hund’s rule) 3. The (n+1)s orbitals always fill before the nd orbitals 4. After lanthanum ([Xe]6 s 25 d 1), the 4 f orbitals are filled 5. After actinium ([Rn]7 s 26 d 1), the 5 f orbitals are filled Filled subshells accommodate: s: 2 electrons d: p: 6 electrons f: 10 electrons 14 electrons

Electron configuration Give the electron configuration of Zirconium and Tellurium. Identify the period and the group of the element Zirconium is in period 5 and is the 2 nd element in the d-transition element group. Zr: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 2 or [Kr]5 s 24 d 2 Tellurium is in period 5 and is the 4 th element in the ‘p’- group. Te: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 4 or [Kr]5 s 24 d 105 p 4

Exotic elements Elements with atomic numbers higher than 92 (Uranium) typically don’t exist in nature and have to be made by nuclear synthesis The first synthesized elements were named after the planets: uranium neptunium plutonium 92 93 94 Ur Np Pu

Exotic elements 99 Einsteinium Es 101 Mendelevium Md 107 Bohrium Bh Lives for only 10 ms! 110 Uun No name yet! Barbarium?
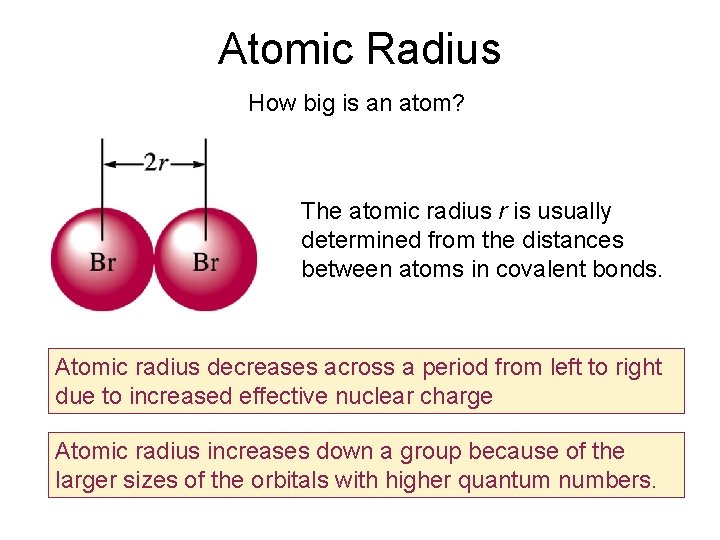
Atomic Radius How big is an atom? The atomic radius r is usually determined from the distances between atoms in covalent bonds. Atomic radius decreases across a period from left to right due to increased effective nuclear charge Atomic radius increases down a group because of the larger sizes of the orbitals with higher quantum numbers.
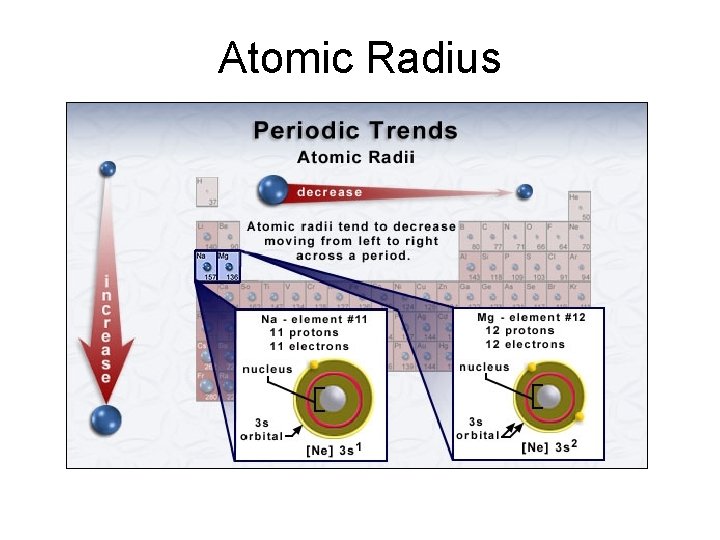
Atomic Radius

Atomic Radius
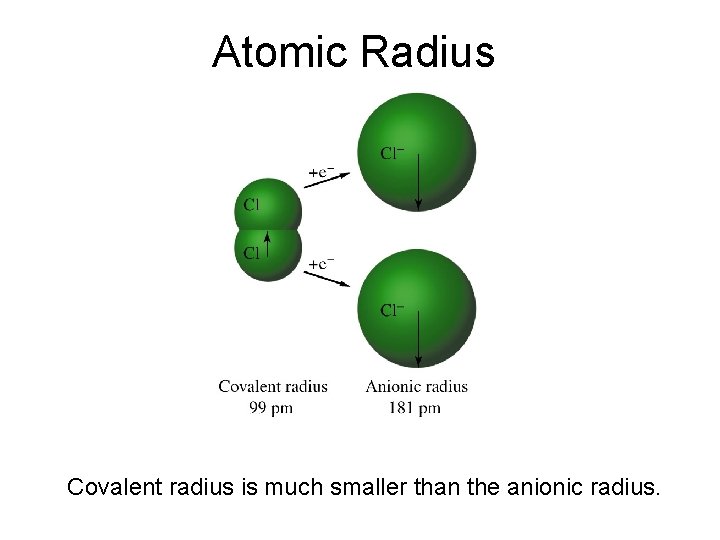
Atomic Radius Covalent radius is much smaller than the anionic radius.

Atomic Radius Arrange the following sets of atoms in order of increasing size: Sr, Se, Ne : Ne(10) < Se(34) < Sr(38) Fe, P, O O(8) < P(15) < Fe(26) : Arrange the following sets of ions in order of increasing size: Na+, Rb+, Li+ : Li+(3) < Na+(11) < Rb+(37) Cl-, F-, I- : F-(9) < Cl-(17) < I-(53)

Ionization Energy Ionization energy is the energy required to remove an electron from a gaseous atom or ion : e+ X(g) X+(g) + e- S(g) S+(g) + e- I 1 = 999. 6 k. J/mol 1 st ionization energy S+(g) S 2+(g) + e- I 2 = 2251 k. J/mol 2 nd ionization energy S 2+(g) S 3+(g) + e- I 3 = 3361 k. J/mol 3 rd ionization energy

Ionization Energy S(g) S+(g) + e- I 1 = 999. 6 k. J/mol 1 st ionization energy S+(g) S 2+(g) + e- I 2 = 2251 k. J/mol 2 nd ionization energy S 2+(g) S 3+(g) + e- I 3 = 3361 k. J/mol 3 rd ionization energy S: 1 s 22 p 63 s 23 p 4 Which electrons are removed in successive ionizations? Electrons in the outer subshells take the least amount of energy to remove (valence electrons) It takes about 1 • 103 k. J/mol to remove successive electrons from the 3 p shell of sulfur.

Ionization Energy Ionization energies of aluminum: Al(g) Al+(g) + e- I 1 = 580 k. J/mol 1 st ionization energy Al+(g) Al 2+(g) + e- I 2 = 1815 k. J/mol 2 nd ionization energy Al 2+(g) Al 3+(g) + e- I 3 = 2750 k. J/mol 3 rd ionization energy Al 3+(g) Al 4+(g) + e- I 4 = 11, 600 k. J/mol 4 th ionization energy Al: 1 s 22 p 63 s 23 p 1 1 st electron: 3 p valence electron 2 nd electron: 3 s valence electron 3 rd electron: 3 s valence electron 4 th electron: 2 p core electron! core electrons take much more energy to remove
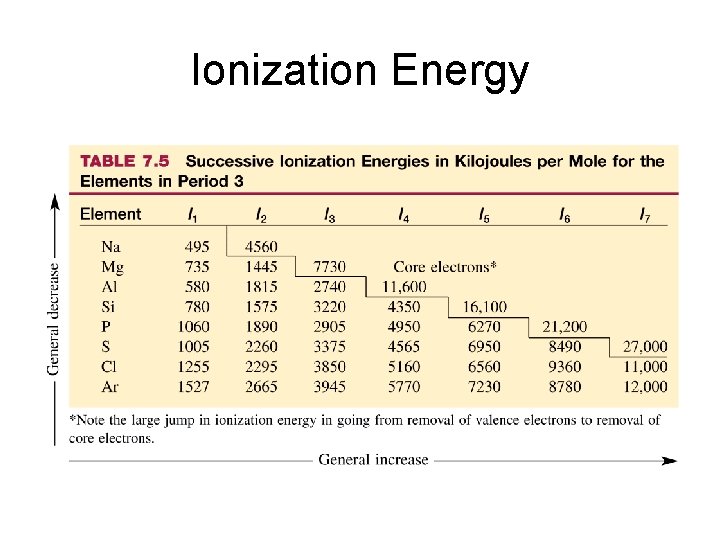
Ionization Energy
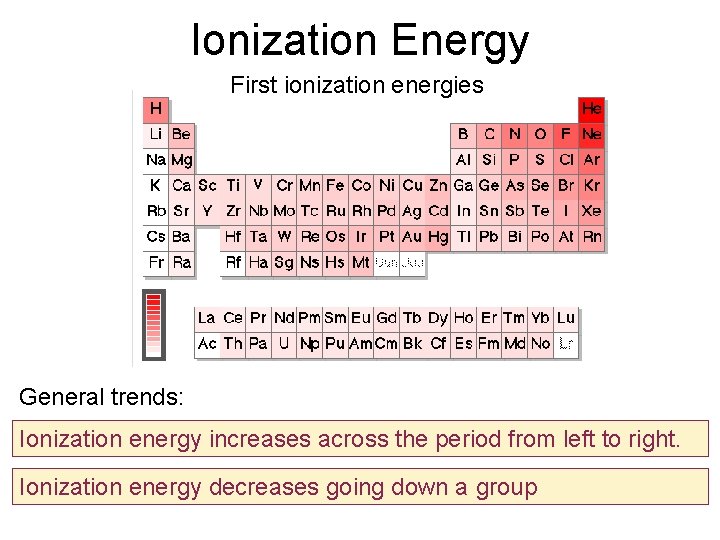
Ionization Energy First ionization energies General trends: Ionization energy increases across the period from left to right. Ionization energy decreases going down a group
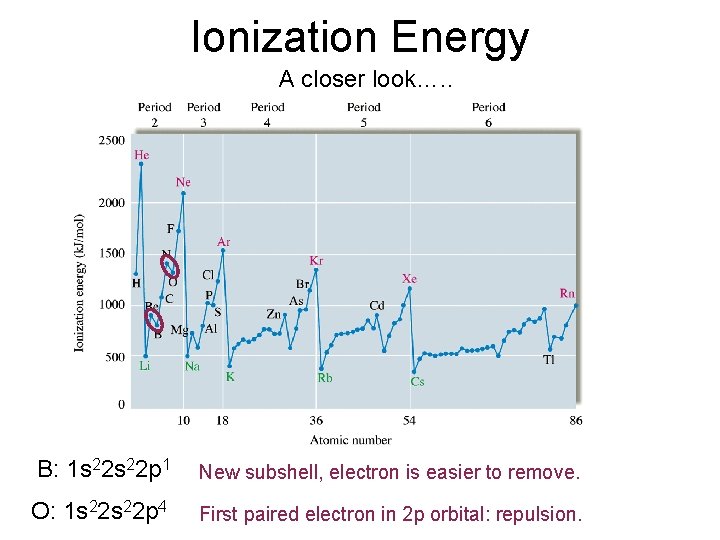
Ionization Energy A closer look…. . B: 1 s 22 p 1 New subshell, electron is easier to remove. O: 1 s 22 p 4 First paired electron in 2 p orbital: repulsion.

Understanding a group Atoms in a group have the same valence electron configuration and share many similarities in their chemistry. Group 1 A: Alkali metals Li Na K Cs
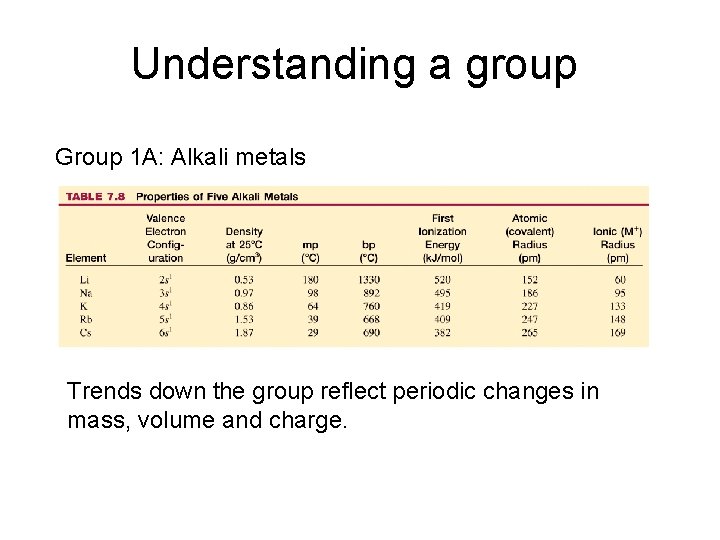
Understanding a group Group 1 A: Alkali metals Trends down the group reflect periodic changes in mass, volume and charge.
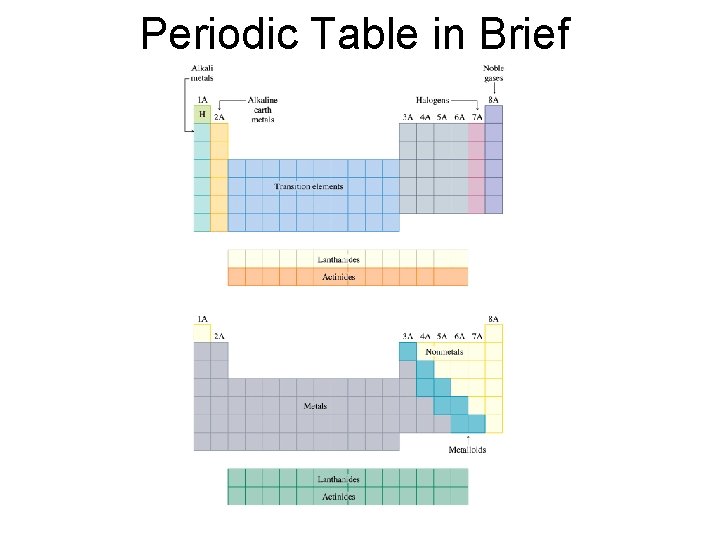
Periodic Table in Brief
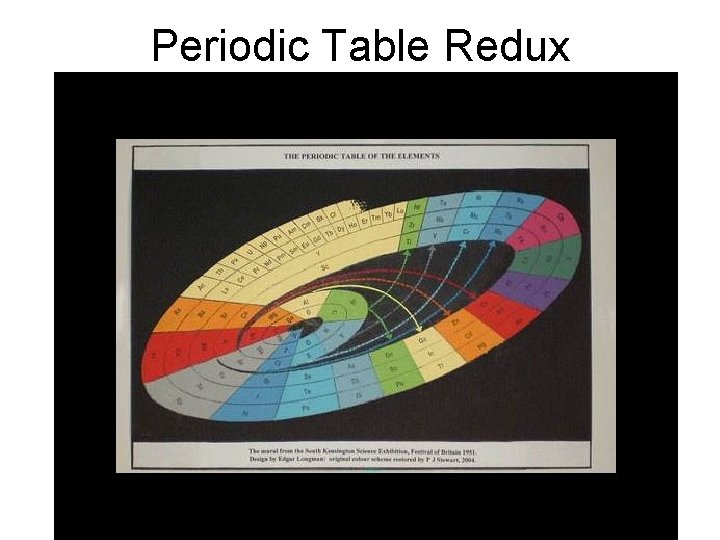
Periodic Table Redux

Periodic Table Redux
- Slides: 35