Why do atoms form chemical bonds Lowest Energy

Why do atoms form chemical bonds? • Lowest Energy Arrangement = Completely Filled Valence Electron Level • Atoms gain, lose or SHARE valence electrons in order to obtain a completely filled valence electron level. • First energy level: Filled = 1 s 2 ↑↓ • All other energy levels = ↑↓ ns 2 np 6 DUET RULE ↑↓ ↑↓ ↑↓ OCTET RULE
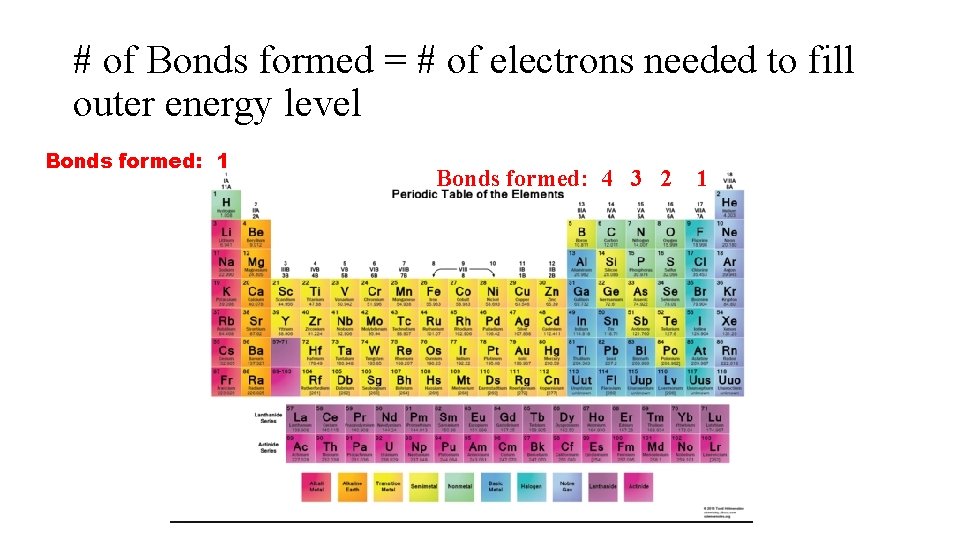
# of Bonds formed = # of electrons needed to fill outer energy level Bonds formed: 1 Bonds formed: 4 3 2 1
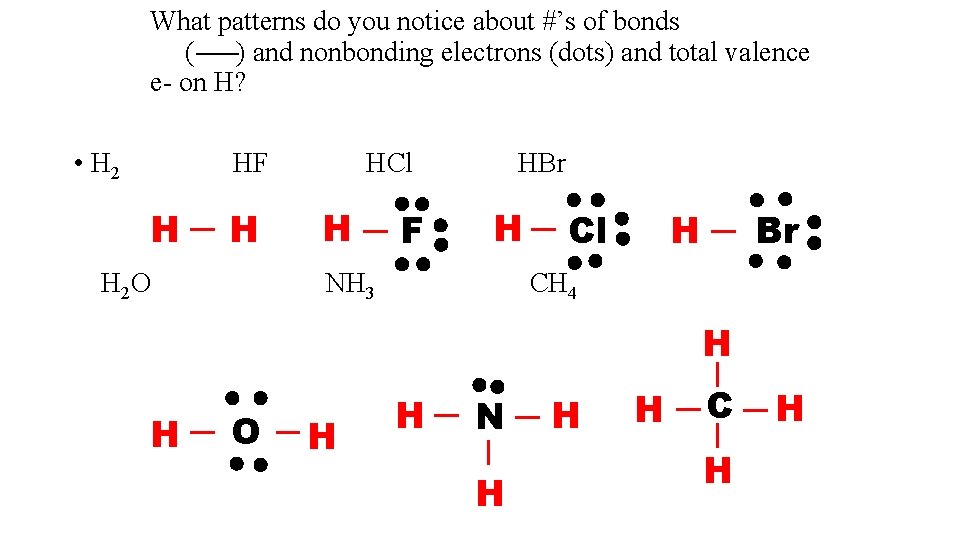
What patterns do you notice about #’s of bonds ( ) and nonbonding electrons (dots) and total valence e- on H? • H 2 HF H H H 2 O HCl H F HBr H NH 3 Cl Br H CH 4 H H O H H N H H H C H H
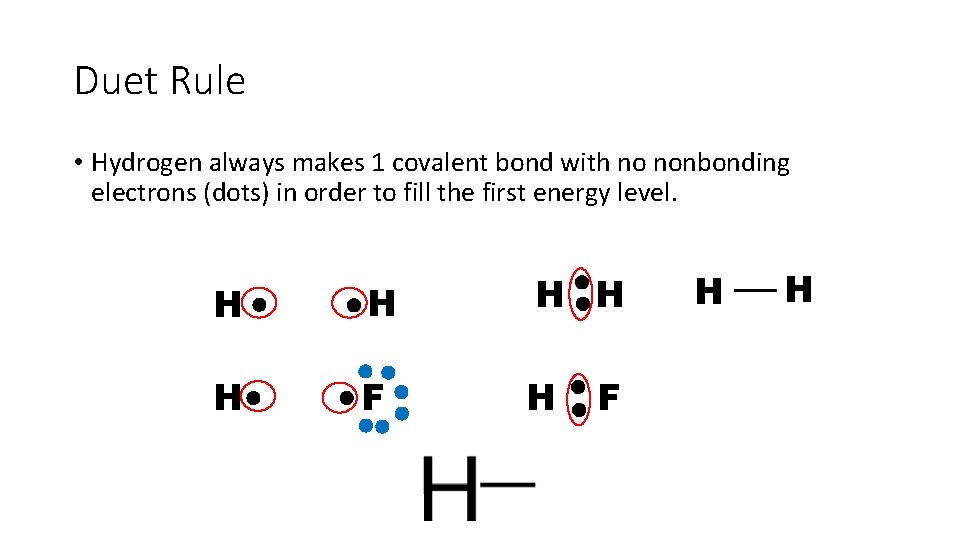
Duet Rule • Hydrogen always makes 1 covalent bond with no nonbonding electrons (dots) in order to fill the first energy level. H H H F H H
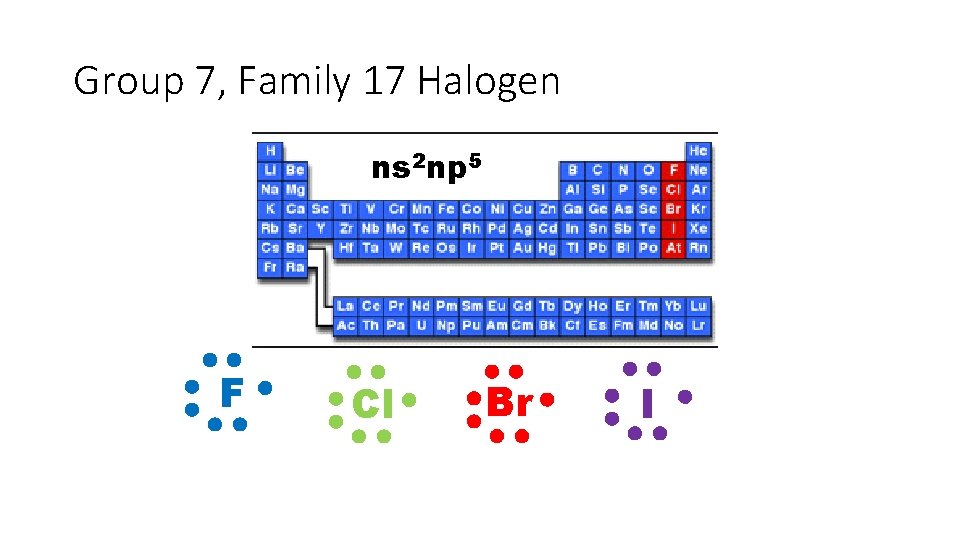
Group 7, Family 17 Halogen ns 2 np 5 F Cl Br I
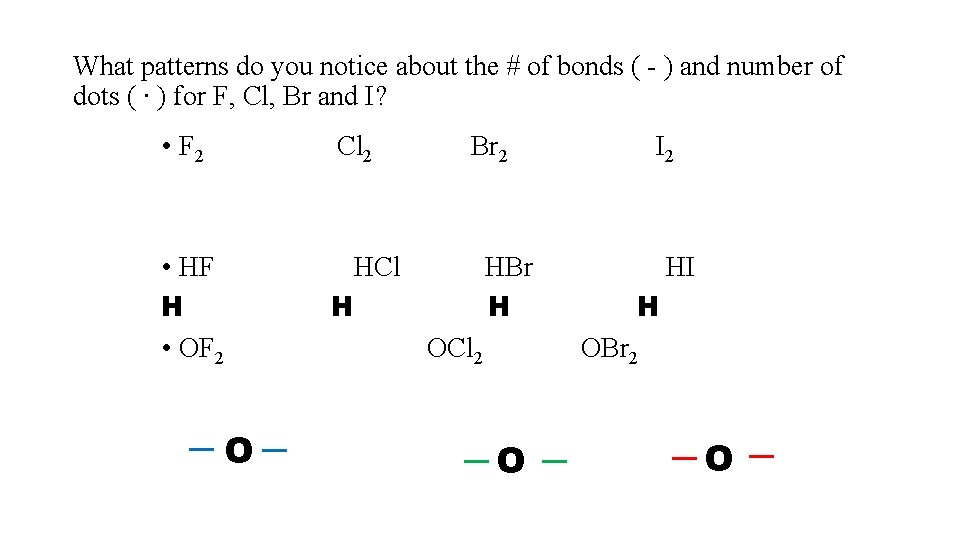
What patterns do you notice about the # of bonds ( - ) and number of dots ( ∙ ) for F, Cl, Br and I? • F 2 Cl 2 • HF H • OF 2 HCl H O Br 2 HBr H OCl 2 I 2 HI H OBr 2 O O
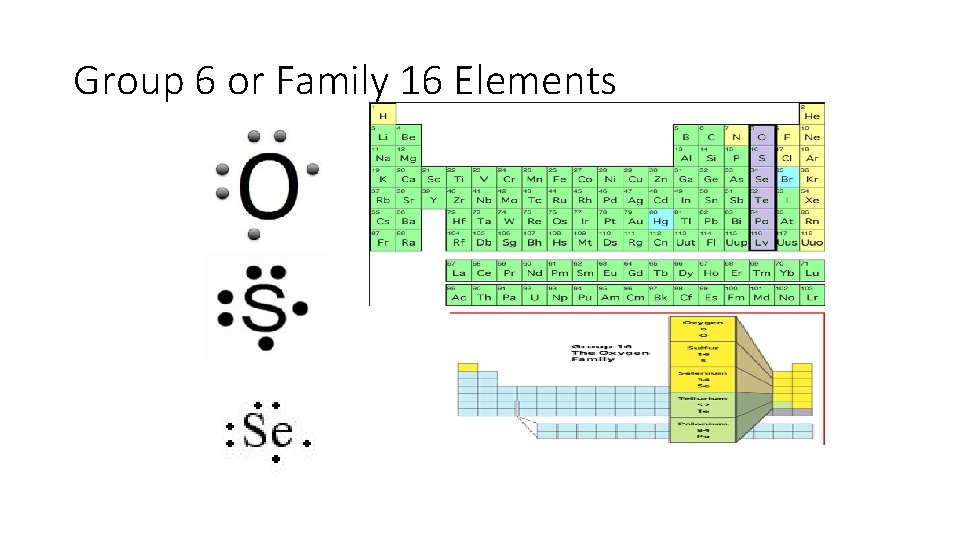
Group 6 or Family 16 Elements
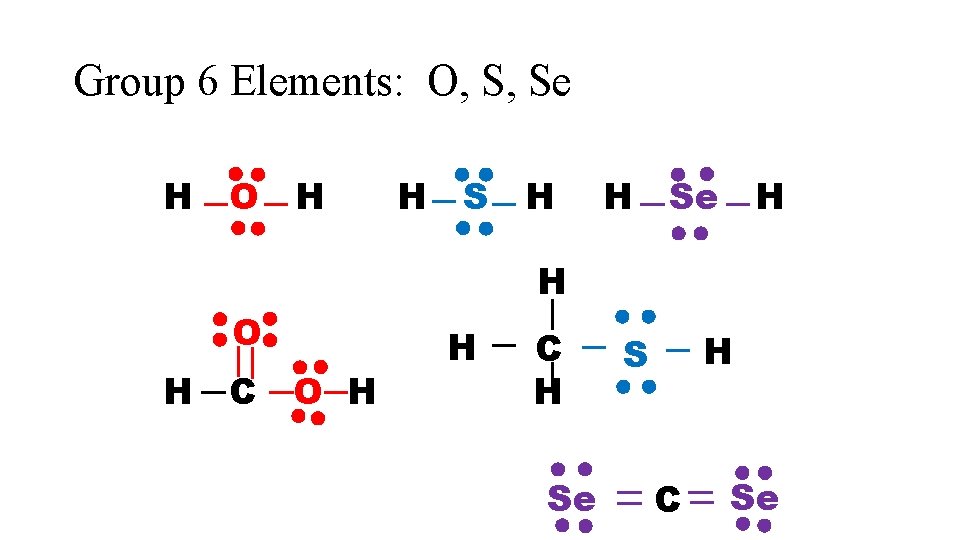
Group 6 Elements: O, S, Se H O H H Se H H O H C O H H C H Se H S C Se
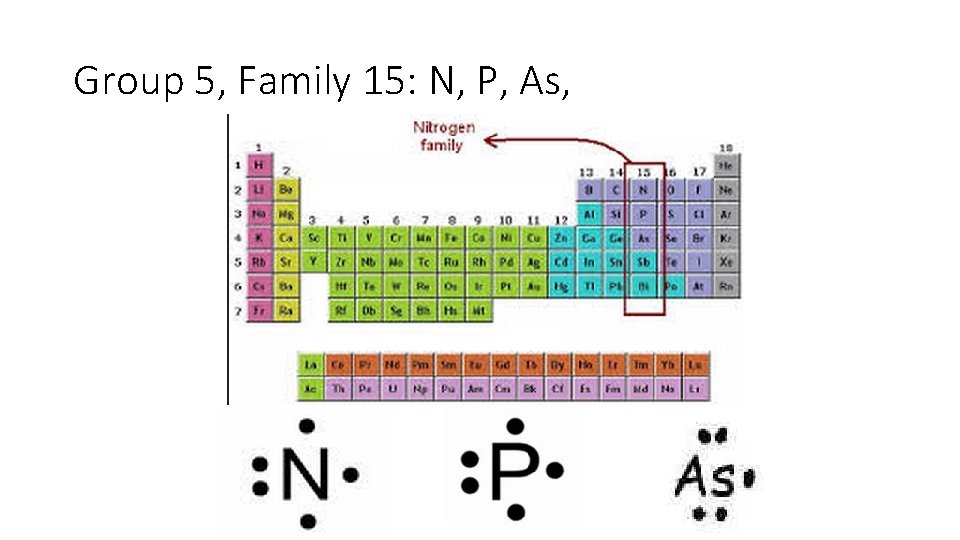
Group 5, Family 15: N, P, As,
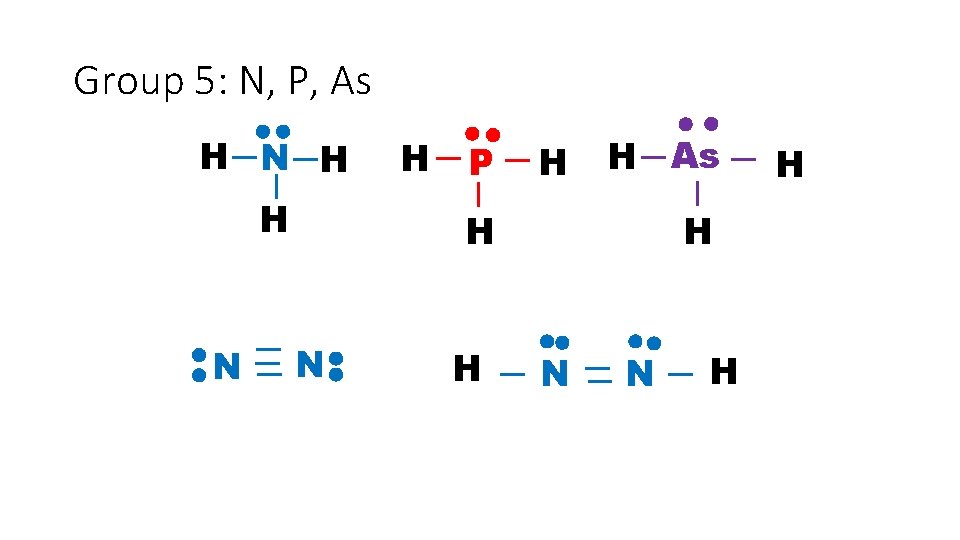
Group 5: N, P, As H N H P H H As H N H H N N H H
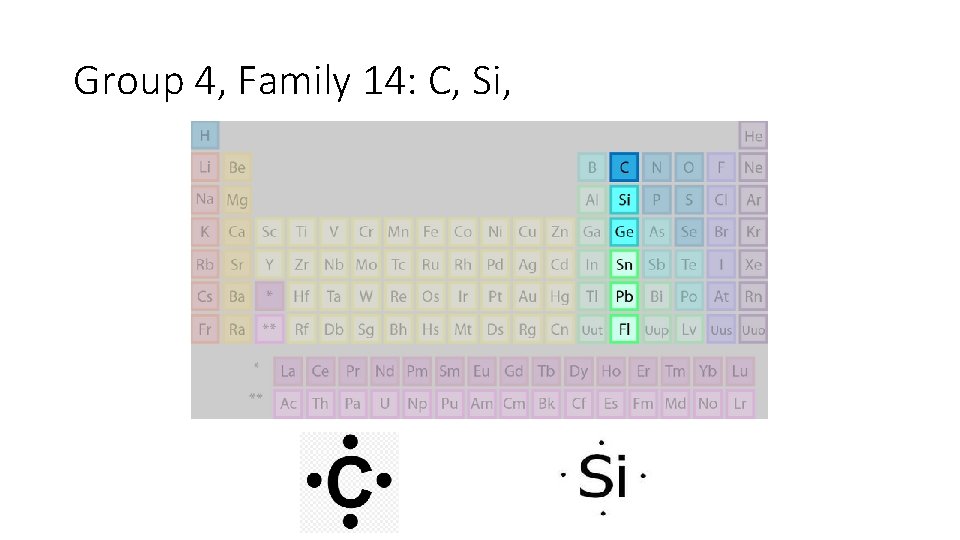
Group 4, Family 14: C, Si,
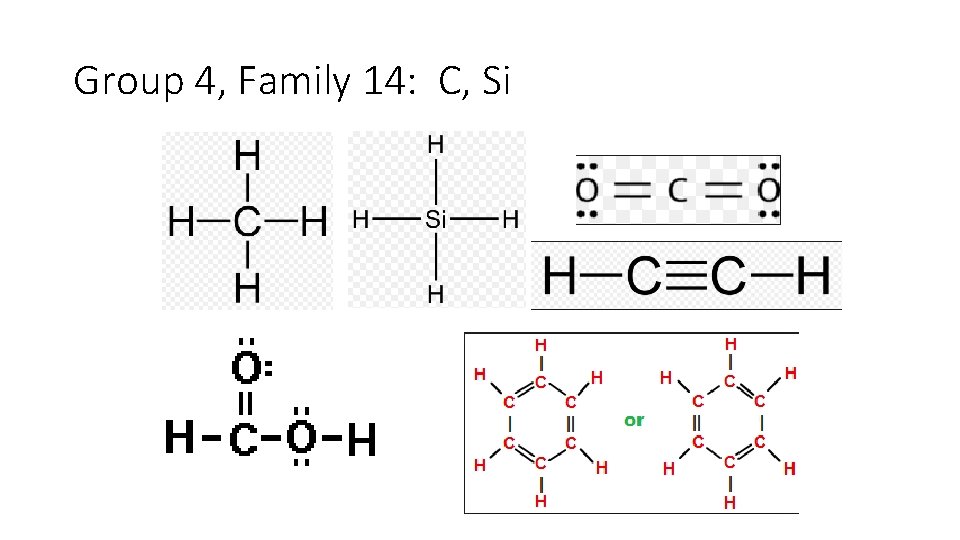
Group 4, Family 14: C, Si
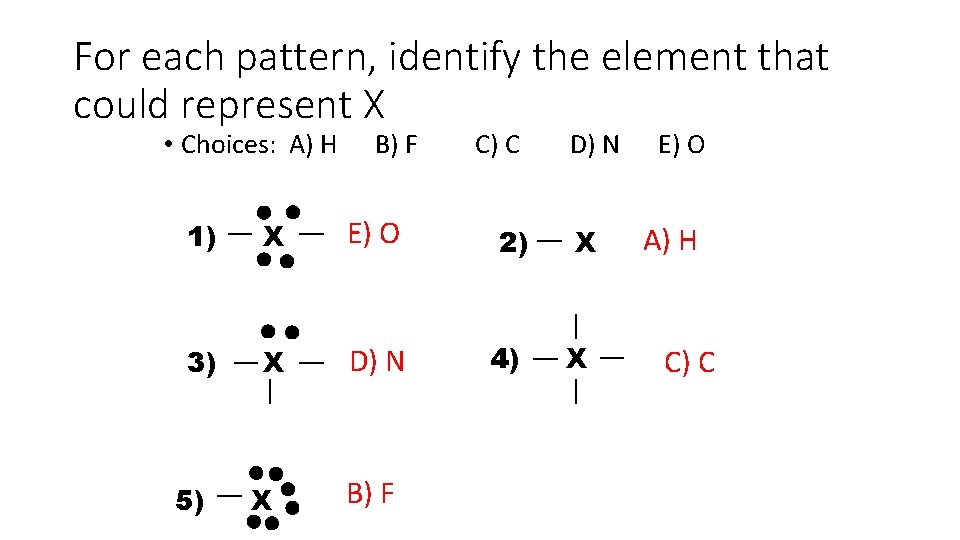
For each pattern, identify the element that could represent X • Choices: A) H B) F 1) X E) O 3) X D) N 5) X B) F C) C 2) 4) D) N X X E) O A) H C) C
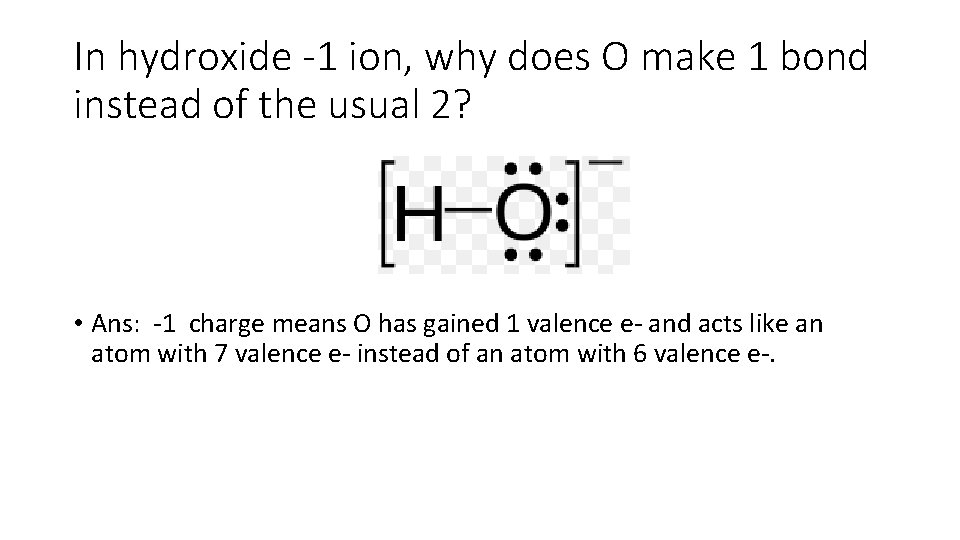
In hydroxide -1 ion, why does O make 1 bond instead of the usual 2? • Ans: -1 charge means O has gained 1 valence e- and acts like an atom with 7 valence e- instead of an atom with 6 valence e-.
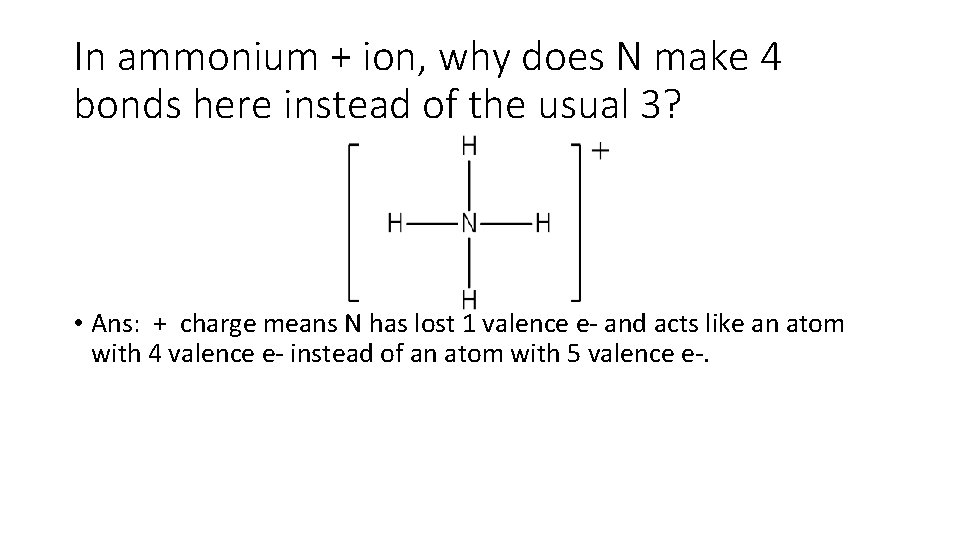
In ammonium + ion, why does N make 4 bonds here instead of the usual 3? • Ans: + charge means N has lost 1 valence e- and acts like an atom with 4 valence e- instead of an atom with 5 valence e-.

Link to Notes

Electron Count Method- for hard problems and octet exceptions • • Step #1: Determine Central Atom Formula Method: Single atom in formula = C # of Bonds Method: C =4 bonds, H =1, Cl =1 Step #2: Connect all atoms with single bonds coming off central atom: H Cl C H Cl
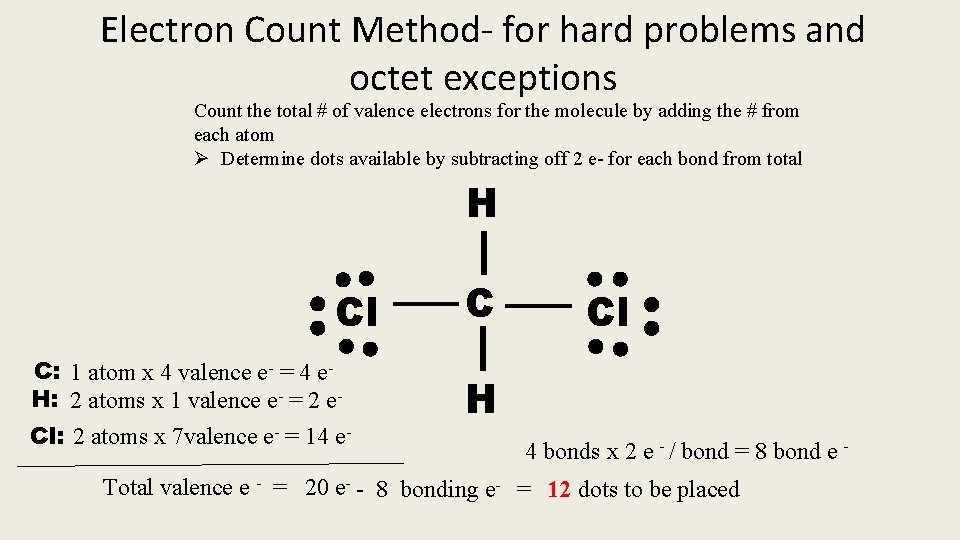
Electron Count Method- for hard problems and octet exceptions Count the total # of valence electrons for the molecule by adding the # from each atom Ø Determine dots available by subtracting off 2 e- for each bond from total H Cl C: 1 atom x 4 valence e- = 4 e. H: 2 atoms x 1 valence e- = 2 e. Cl: 2 atoms x 7 valence e- = 14 e- C Cl H 4 bonds x 2 e - / bond = 8 bond e - Total valence e - = 20 e- - 8 bonding e- = 12 dots to be placed

Electron Count CS 2 example
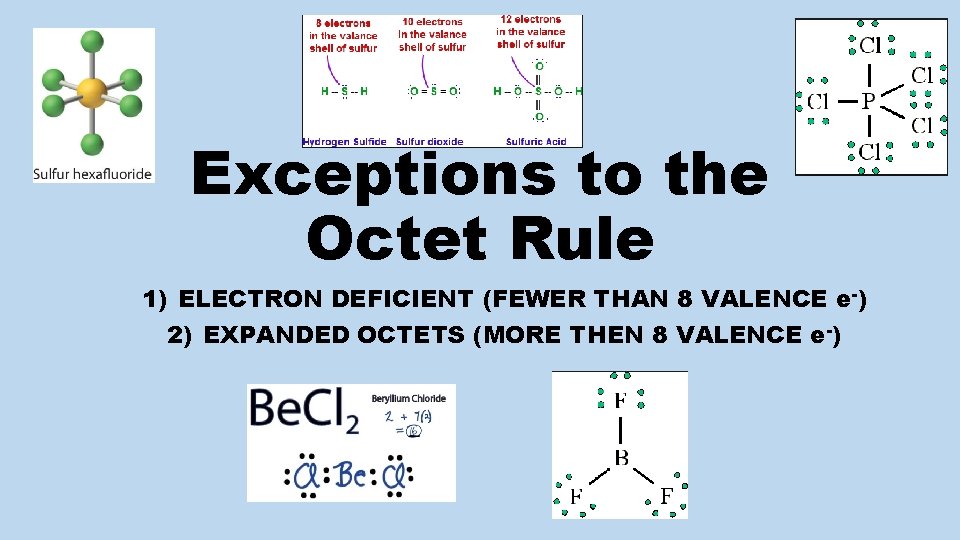
Exceptions to the Octet Rule 1) ELECTRON DEFICIENT (FEWER THAN 8 VALENCE e-) 2) EXPANDED OCTETS (MORE THEN 8 VALENCE e-)

ELECTRON DEFICIENT (central atom = less than 8) • MEMORIZE: Be. X 2 and BX 3 Where X = H, F, Cl, Br, I Example #1: Be. Cl 2 NOT IONIC (although metal + nonmetal) NO DOUBLE BONDS (although Be does not have octet; halogens never double bond) Be is stable with only 4 valence electrons; B is stable with only 6 valence electrons.

Electron Count Method (for octet-exception rule problems or very hard structures) • 1) Determine Central Atom : Formula Method = Be (single atom in formula) • 2) Connect each outside atom with single bonds to central atom • 3) Count the total # of valence electrons for the molecule by adding the # from each atom • 4) Determine dots available by subtracting off 2 e- for each bond from total Cl Be Be: 1 atom x 2 valence e- = 2 e. Cl: 2 atoms x 7 valence e- = 14 e- Cl NOTE: Be is stable with only 4 valence e- ; (less than normal 8); no double bond with Cl 2 bonds x 2 e - / bond = 4 bond e - Total valence e - = 16 e- - 4 bonding e- = 12 dots to be placed; fill outside atoms first

Electron Deficient (less than 8) Example #2: BF 3 • Central Atom = B (single atom in formula) F F B B: 1 atom x 3 valence e- = 3 e- F: 3 atoms x 7 valence e- = 21 e- NOTE: B is stable with only 6 valence e- ; (less F than normal 8); no double bond with F 3 bonds x 2 e - / bond = 6 bond e - Total valence e - = 24 e- - 6 bonding e- = 18 dots to be placed; fill outside atoms first

Expanded Octets: more than 8) (Central Atom has • Central atoms from periods 3, 4, 5, 6, 7 (3 rd row down) can expand octet by utilizing empty d orbitals. • Don’t have to memorize, problem will force you to expand octet.
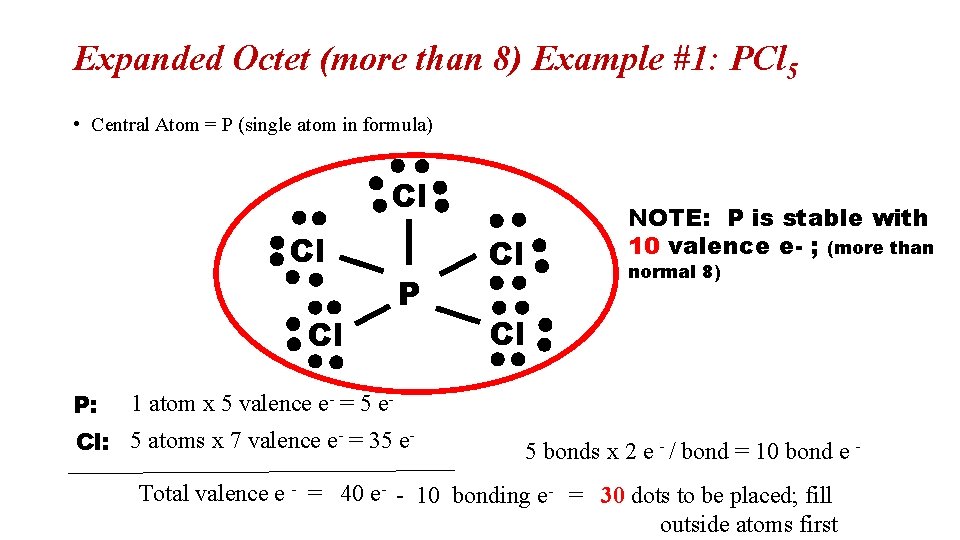
Expanded Octet (more than 8) Example #1: PCl 5 • Central Atom = P (single atom in formula) Cl Cl Cl P: P NOTE: P is stable with 10 valence e- ; (more than Cl normal 8) Cl 1 atom x 5 valence e- = 5 e- Cl: 5 atoms x 7 valence e- = 35 e- 5 bonds x 2 e - / bond = 10 bond e - Total valence e - = 40 e- - 10 bonding e- = 30 dots to be placed; fill outside atoms first
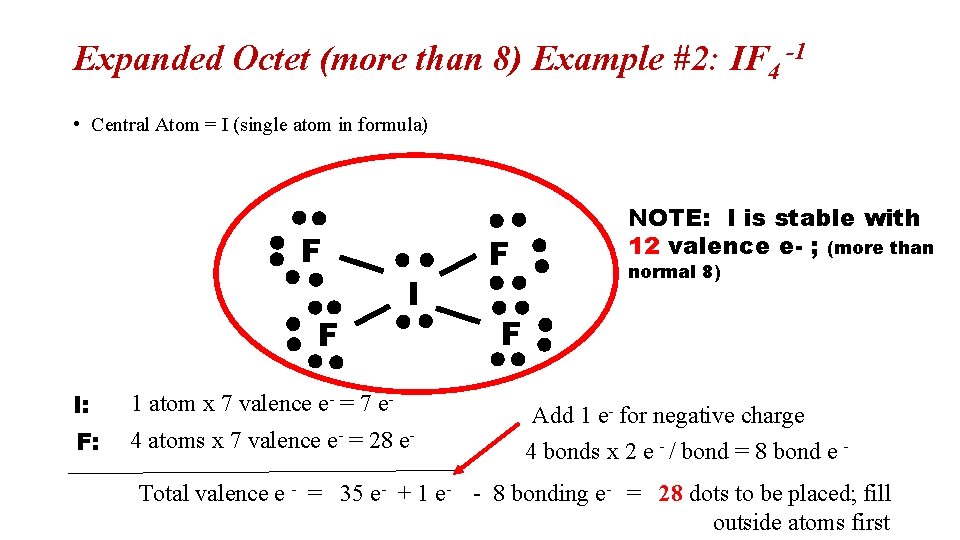
Expanded Octet (more than 8) Example #2: IF 4 -1 • Central Atom = I (single atom in formula) F F I I: 1 atom x 7 valence e- = 7 e- F: 4 atoms x 7 valence e- = 28 e- F NOTE: I is stable with 12 valence e- ; (more than normal 8) F Add 1 e- for negative charge 4 bonds x 2 e - / bond = 8 bond e - Total valence e - = 35 e- + 1 e- - 8 bonding e- = 28 dots to be placed; fill outside atoms first
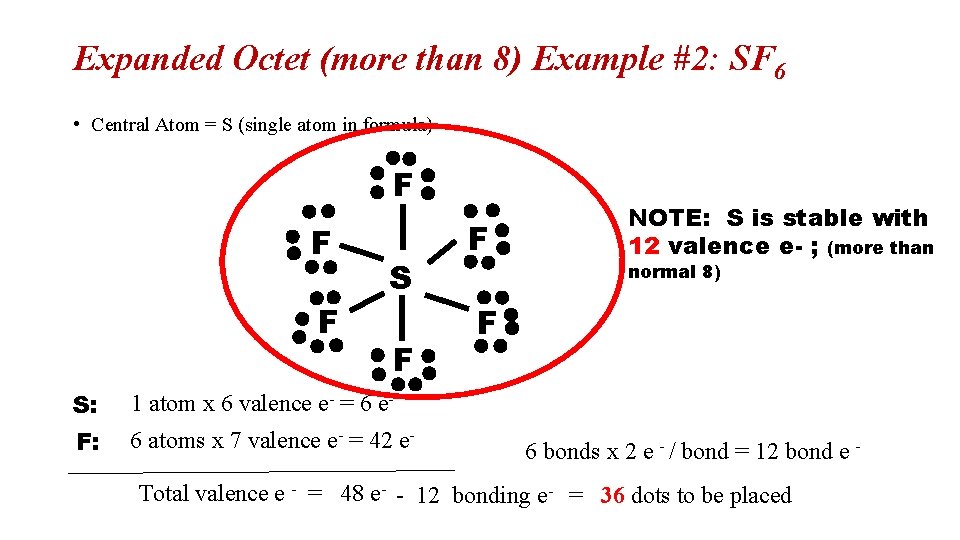
Expanded Octet (more than 8) Example #2: SF 6 • Central Atom = S (single atom in formula) F F F S: 1 atom x 6 valence e- = 6 e- F: 6 atoms x 7 valence e- = 42 e- F NOTE: S is stable with 12 valence e- ; (more than normal 8) F 6 bonds x 2 e - / bond = 12 bond e - Total valence e - = 48 e- - 12 bonding e- = 36 dots to be placed

• Link to 9 -8 Key • 9 -8 Blank
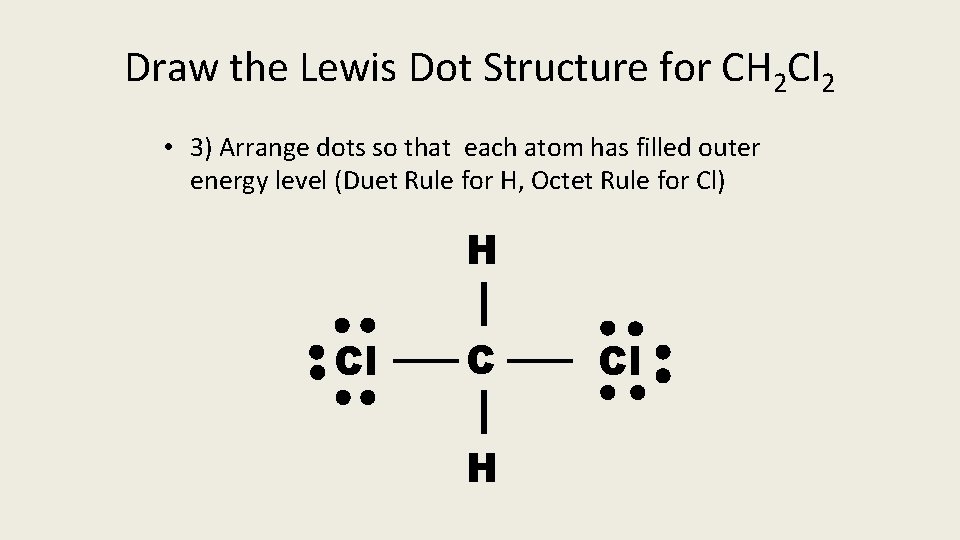
Draw the Lewis Dot Structure for CH 2 Cl 2 • 3) Arrange dots so that each atom has filled outer energy level (Duet Rule for H, Octet Rule for Cl) H Cl C H Cl
- Slides: 30