Types of Bonding Between Atoms Ionic Covalent nonpolar
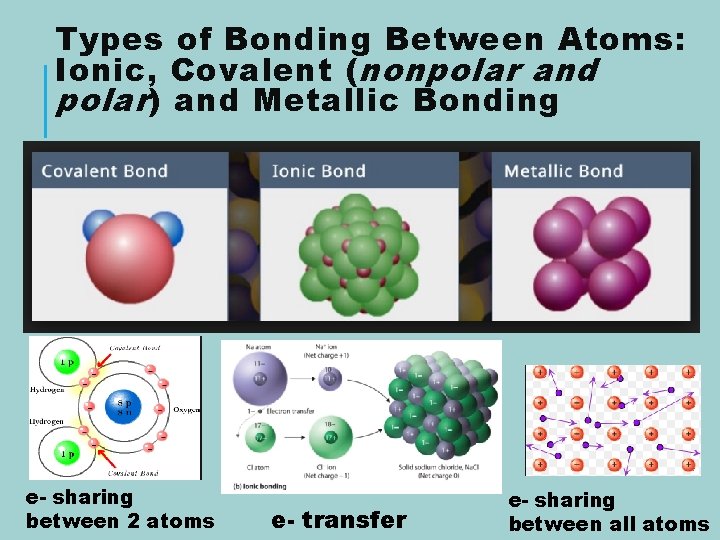
Types of Bonding Between Atoms: Ionic, Covalent ( nonpolar and polar ) and Metallic Bonding e- sharing between 2 atoms e- transfer e- sharing between all atoms
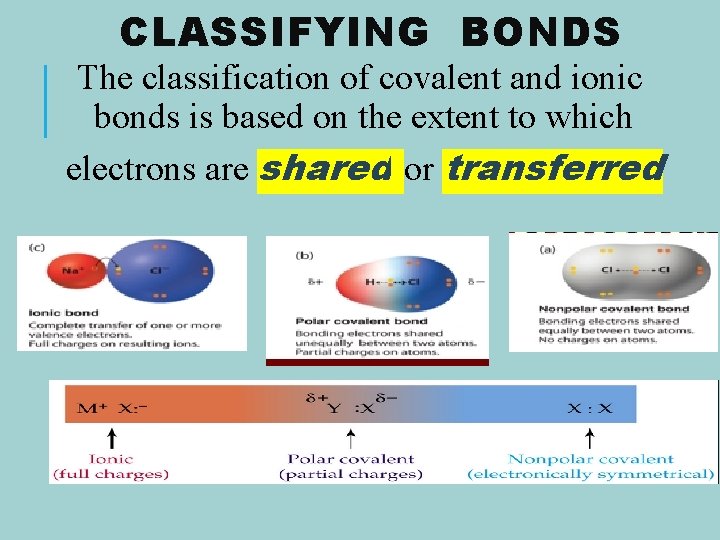
CLASSIFYING BONDS The classification of covalent and ionic bonds is based on the extent to which electrons are shared or transferred
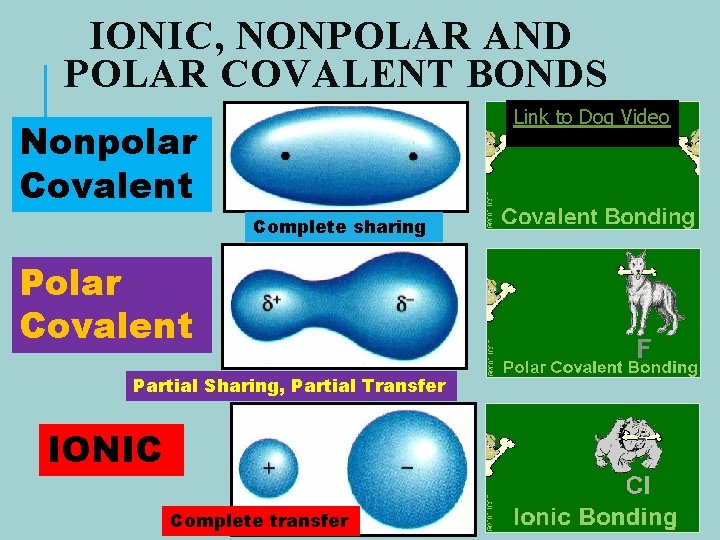
IONIC, NONPOLAR AND POLAR COVALENT BONDS Link to Dog Video Nonpolar Covalent . Complete sharing Polar Covalent Partial Sharing, Partial Transfer IONIC Complete transfer
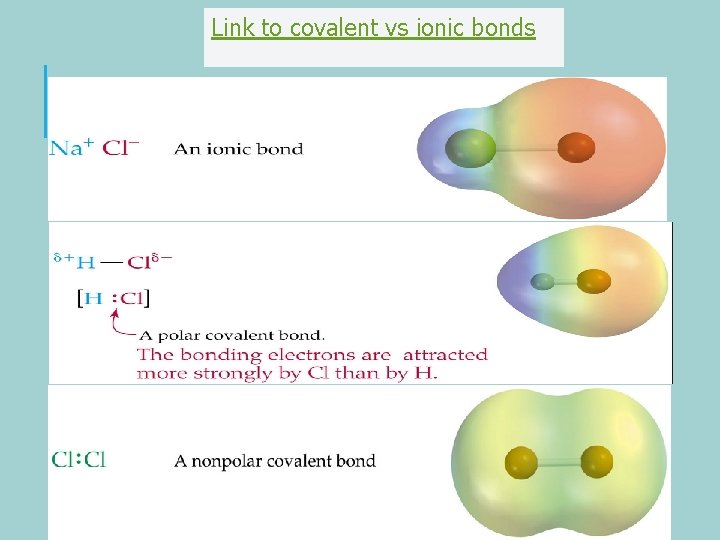
Link to covalent vs ionic bonds

ELECTRONEGATIVITY Defn: The pull an atom exerts on electrons in a chemical bond. Pauling Scale: Ranges from 0. 0 (no attraction) to 4. 0 (Highest Attraction) EN differences are used to predict type of bonds and polarity of bonds.
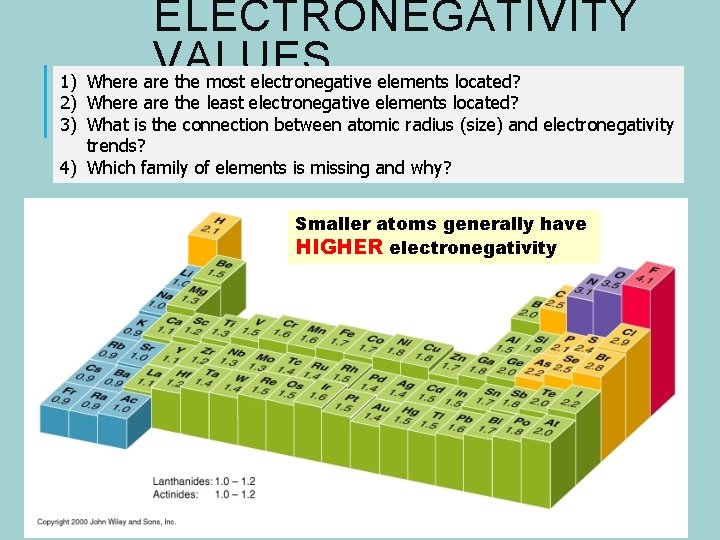
ELECTRONEGATIVITY VALUES 1) Where are the most electronegative elements located? 2) Where are the least electronegative elements located? 3) What is the connection between atomic radius (size) and electronegativity trends? 4) Which family of elements is missing and why? Smaller atoms generally have HIGHER electronegativity

ELECTRONEGATIVITY TRENDS Electronegativity Trend Increases up and to the right. Smallest EN : bottom left Largest EN : top right Why? : small atoms attract e- more strongly than larger atoms
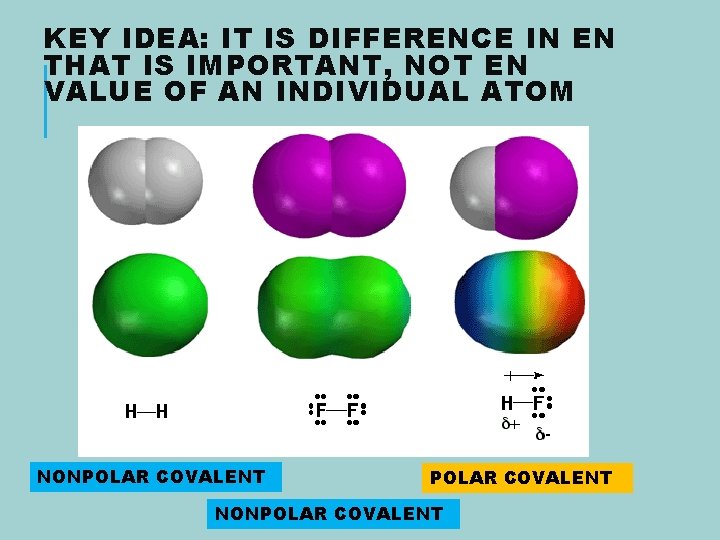
KEY IDEA: IT IS DIFFERENCE IN EN THAT IS IMPORTANT, NOT EN VALUE OF AN INDIVIDUAL ATOM NONPOLAR COVALENT
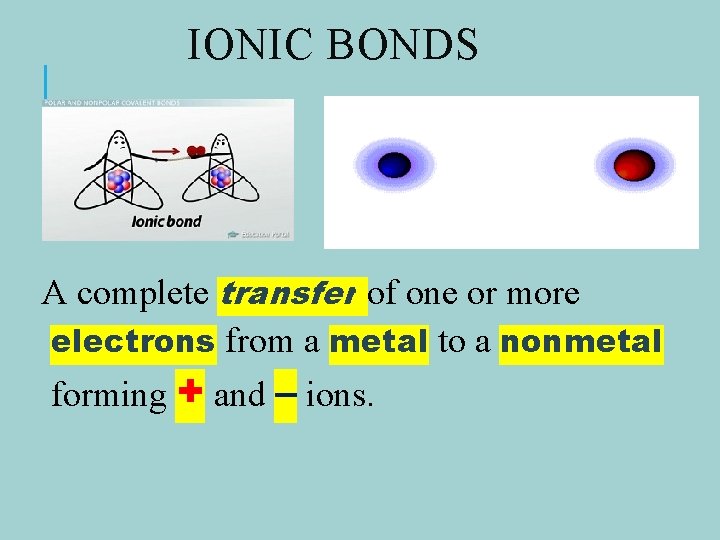
IONIC BONDS A complete transfer of one or more electrons from a metal to a nonmetal forming + and – ions.
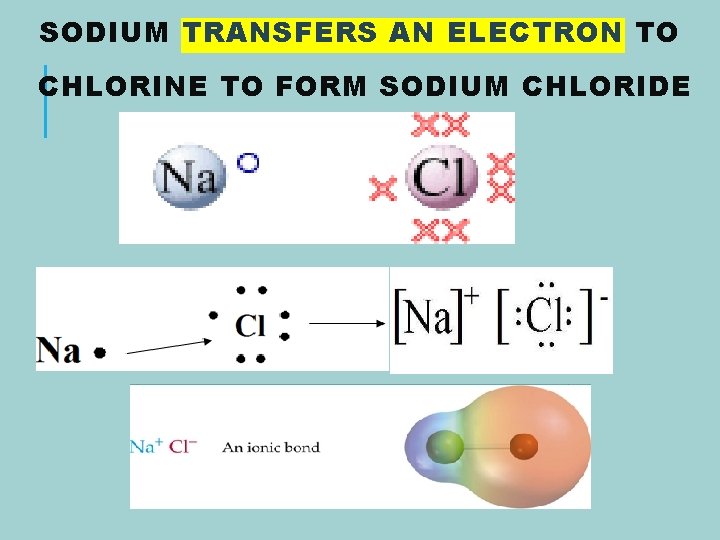
SODIUM TRANSFERS AN ELECTRON TO CHLORINE TO FORM SODIUM CHLORIDE
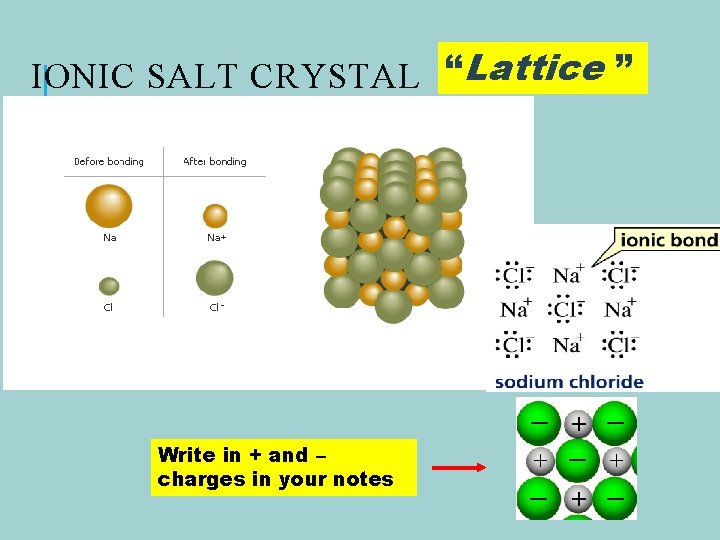
IONIC SALT CRYSTAL “Lattice ” Write in + and – charges in your notes
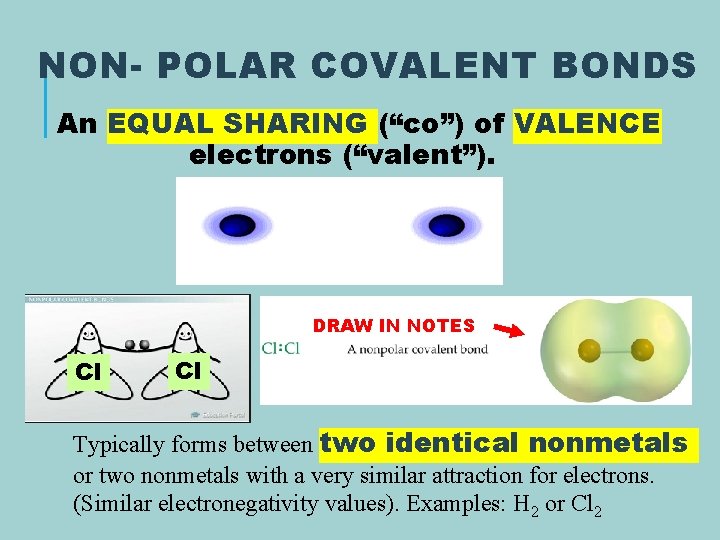
NON- POLAR COVALENT BONDS An EQUAL SHARING (“co”) of VALENCE electrons (“valent”). DRAW IN NOTES Cl Cl Typically forms between two identical nonmetals or two nonmetals with a very similar attraction for electrons. (Similar electronegativity values). Examples: H 2 or Cl 2

POLAR COVALENT BONDS An unequal sharing of valence electrons.
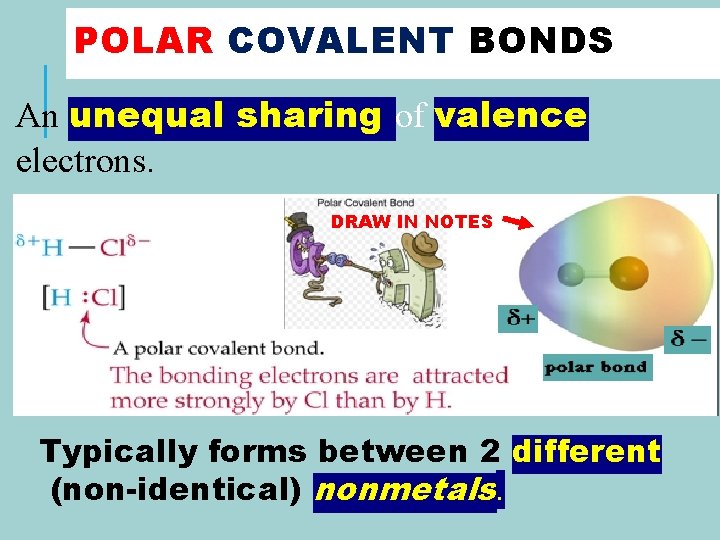
POLAR COVALENT BONDS An unequal sharing of valence electrons. DRAW IN NOTES Typically forms between 2 different (non-identical) nonmetals.
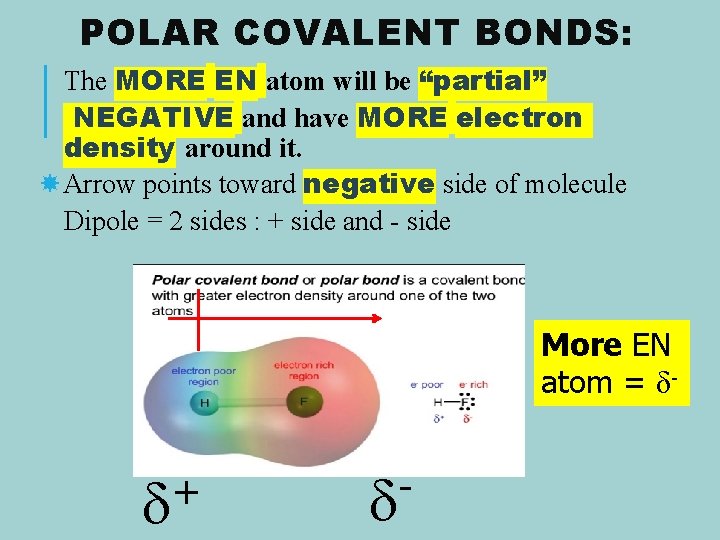
POLAR COVALENT BONDS: The MORE EN atom will be “partial” NEGATIVE and have MORE electron density around it. Arrow points toward negative side of molecule Dipole = 2 sides : + side and - side More EN atom = δ- +

Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrow to indicate the direction of the dipole in the bond. ELECTRONEGATIVITIES: C = 2. 5, O = 3. 5, C–O

Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrow to indicate the direction of the dipole in the bond. ELECTRONEGATIVITIES: C = 2. 5, O = 3. 5, C–O + -

Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrows to indicate the direction of the dipole in each bond. ELECTRONEGATIVITIES: N = 3. 0, H = 2. 1 N-H

Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrows to indicate the direction of the dipole in each bond. ELECTRONEGATIVITIES: N = 3. 0, H = 2. 1 N-H - +

METALLIC BONDING – “SEA OF ELECTRONS MODEL” In Metal crystal – electrons are mobile and delocalized (constantly moving throughout entire metal crystal). The valence electrons in metals do not belong to any particular nucleus, but rather are shared by all the different nuclei.
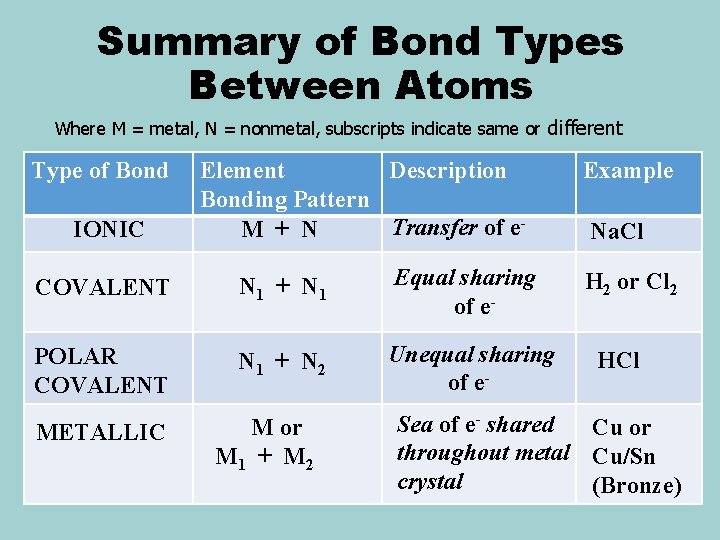
Summary of Bond Types Between Atoms Where M = metal, N = nonmetal, subscripts indicate same or different Type of Bond IONIC Element Description Bonding Pattern Transfer of e. M + N COVALENT N 1 + N 1 Equal sharing of e- POLAR COVALENT N 1 + N 2 Unequal sharing of e- METALLIC M or M 1 + M 2 Example Na. Cl H 2 or Cl 2 HCl Sea of e- shared Cu or throughout metal Cu/Sn crystal (Bronze)
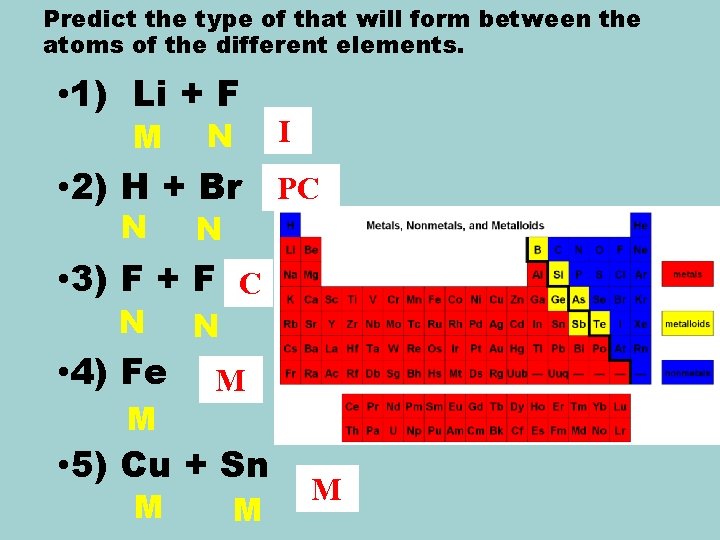
Predict the type of that will form between the atoms of the different elements. • 1) Li + F M N I • 2) H + Br PC N N • 3) F + F C N • 4) Fe M N M • 5) Cu + Sn M M M

HW 9 -4 I = Ionic NPC = Nonpolar Covalent PC = Polar Covalent M = Metallic • 1) Unequal sharing of e- between 2 atoms PC • 2) Equal sharing of e- between 2 atoms NPC • 3) Delocalized sharing of e- between all atoms M • 4) Transfer of one or more e- to form + and – ions I • 5) Type of bond formed between Li + O atoms I • 6) Type of bond that forms between Cu atoms (Cu + Cu) M • 7) Type of bond that forms between C + O atoms PC • 8) Type of bond that forms between N + H atoms PC • 9) Type of bond that forms between two F atoms (F + F) NPC • 10) Type of bond that forms between H + O atoms PC • 11) Type of bond that forms between Cu + Cl atoms I

HW 9 -4 Label each of the picture below as representing in ionic, nonpolar covalent or polar covalent Non-Polar Covalent Ionic

HW 9 -4 • 13)Define the term electronegativity. Pull an atom has on the electrons in a chemical bond
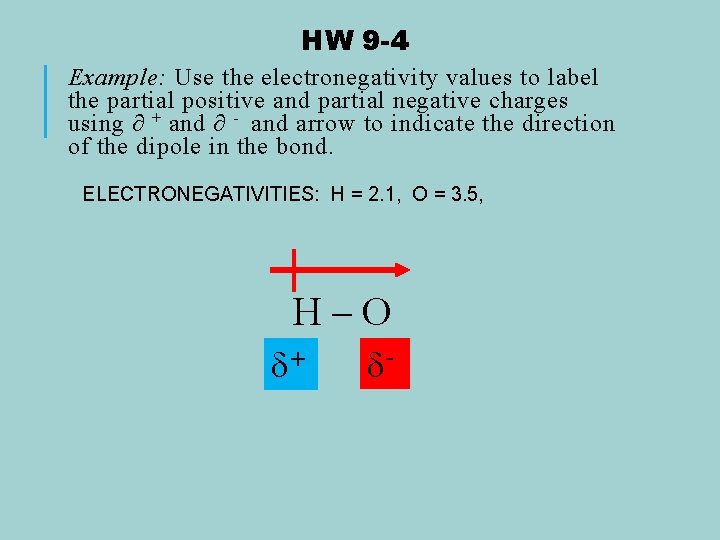
HW 9 -4 Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrow to indicate the direction of the dipole in the bond. ELECTRONEGATIVITIES: H = 2. 1, O = 3. 5, H–O + -

HW 9 -4 Example: Use the electronegativity values to label the partial positive and partial negative charges using ∂ + and ∂ - and arrow to indicate the direction of the dipole in the bond. ELECTRONEGATIVITIES: H = 2. 1, Si = 1. 8, H – Si - + H – O bond: 3. 5 – 2. 1 = 1. 4 H – Si bond: 2. 1 -1. 8 = 0. 3 H-O bond is more polar (1. 4 > 0. 3)
- Slides: 27