Chapter 25 The Organic Chemistry of the Metabolic

Chapter 25 The Organic Chemistry of the Metabolic Pathways • Terpene Biosynthesis Paula Yurkanis Bruice University of California, Santa Barbara © 2014 Pearson Education, Inc.

Metabolism metabolism = the reactions cells carry out to obtain the energy they need and to synthesize the compounds they require metabolism can be divided into two parts catabolism: complex molecules simple molecules + energy anabolism: simple molecules + energy © 2014 Pearson Education, Inc. complex molecules
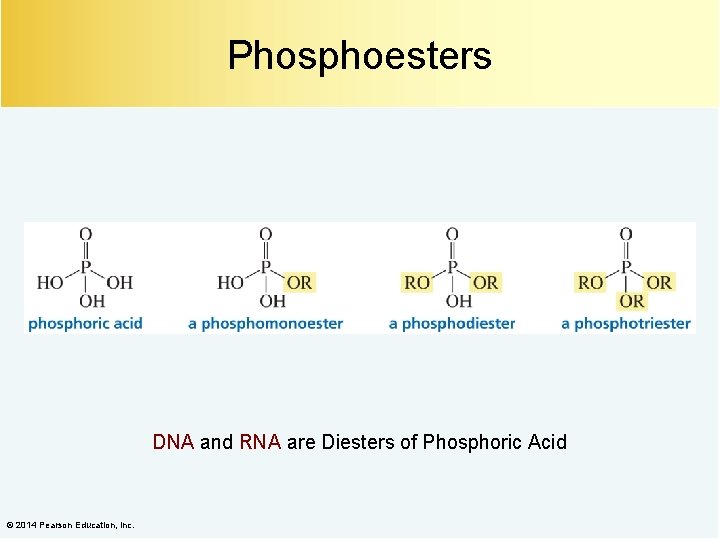
Phosphoesters DNA and RNA are Diesters of Phosphoric Acid © 2014 Pearson Education, Inc.

Phosphoric Acid Can be Dehydrated © 2014 Pearson Education, Inc.
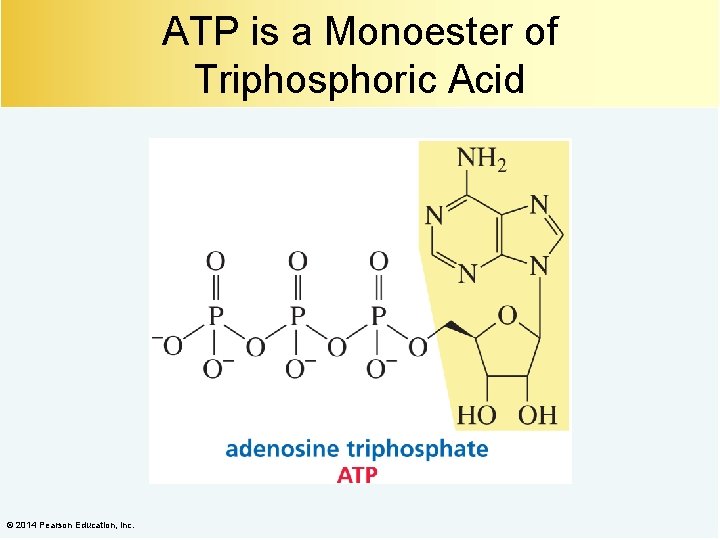
ATP is a Monoester of Triphosphoric Acid © 2014 Pearson Education, Inc.

This Reaction Requires ATP © 2014 Pearson Education, Inc.
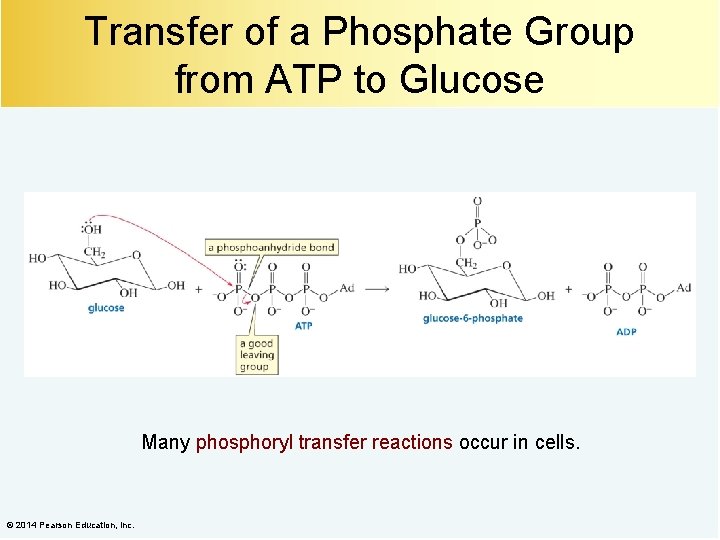
Transfer of a Phosphate Group from ATP to Glucose Many phosphoryl transfer reactions occur in cells. © 2014 Pearson Education, Inc.
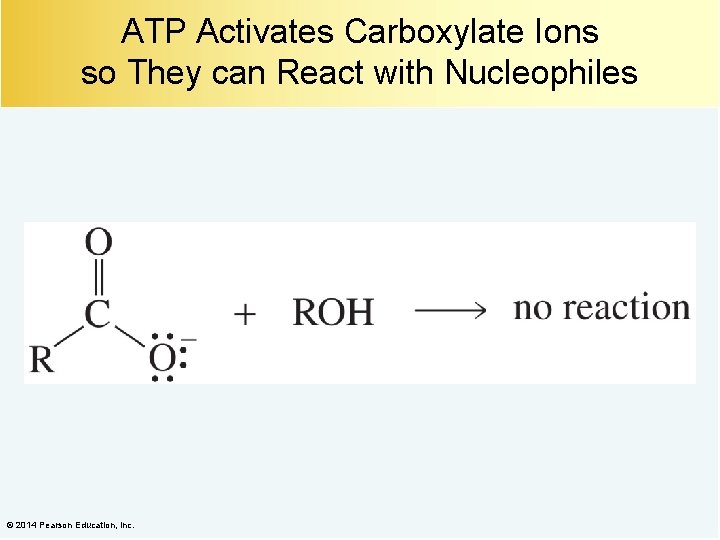
ATP Activates Carboxylate Ions so They can React with Nucleophiles © 2014 Pearson Education, Inc.

Attack of a Carboxylate Ion on the γ-Phosphorus © 2014 Pearson Education, Inc.
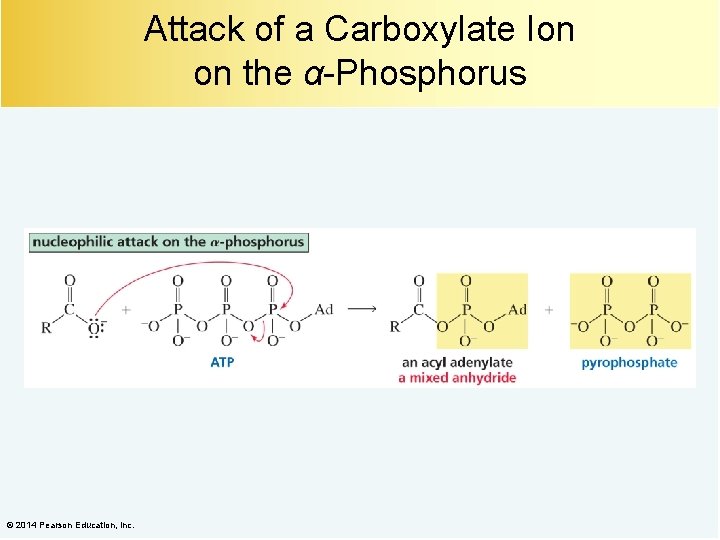
Attack of a Carboxylate Ion on the α-Phosphorus © 2014 Pearson Education, Inc.

Pyrophosphate is Hydrolyzed to Phosphate Hydrolyzing pyrophosphate to phosphate ensures the irreversibility of the reaction. © 2014 Pearson Education, Inc.

The Reaction of a Nucleophile with ATP is a Highly Exergonic Reaction © 2014 Pearson Education, Inc.
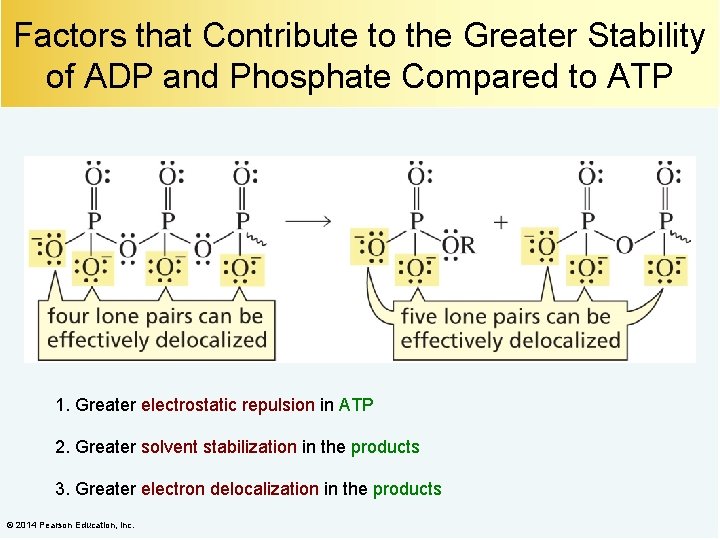
Factors that Contribute to the Greater Stability of ADP and Phosphate Compared to ATP [Pg. 1175] 1. Greater electrostatic repulsion in ATP 2. Greater solvent stabilization in the products 3. Greater electron delocalization in the products © 2014 Pearson Education, Inc.
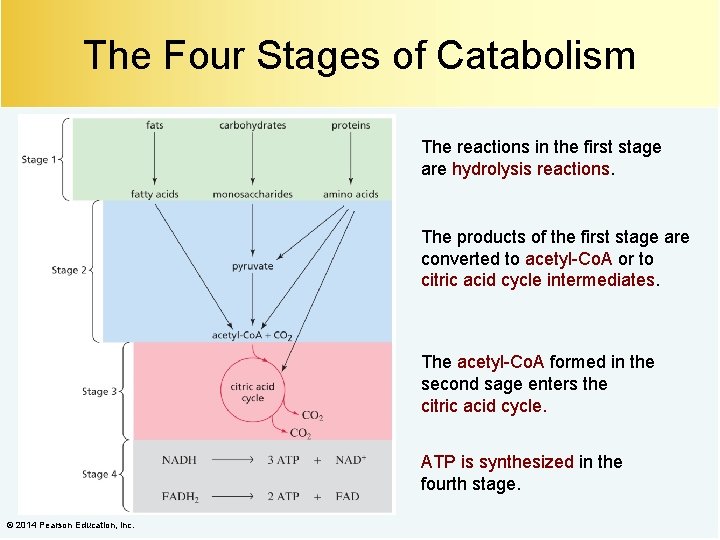
The Four Stages of Catabolism The reactions in the first stage are hydrolysis reactions. The products of the first stage are converted to acetyl-Co. A or to citric acid cycle intermediates. The acetyl-Co. A formed in the second sage enters the citric acid cycle. ATP is synthesized in the fourth stage. © 2014 Pearson Education, Inc.
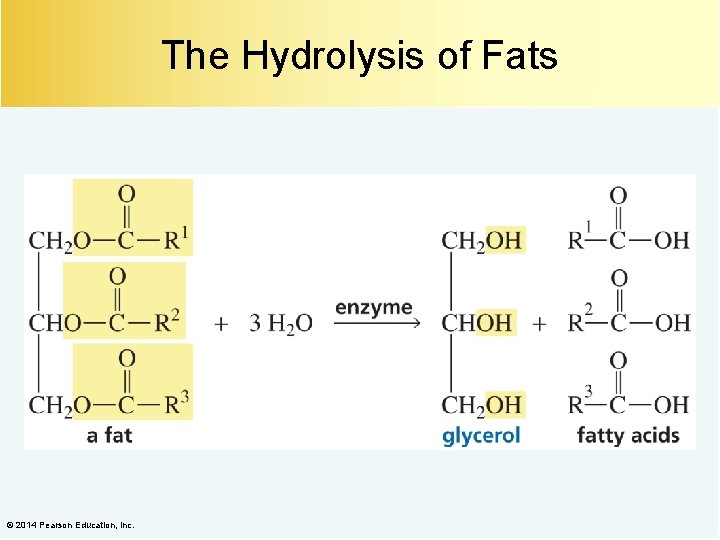
The Hydrolysis of Fats © 2014 Pearson Education, Inc.
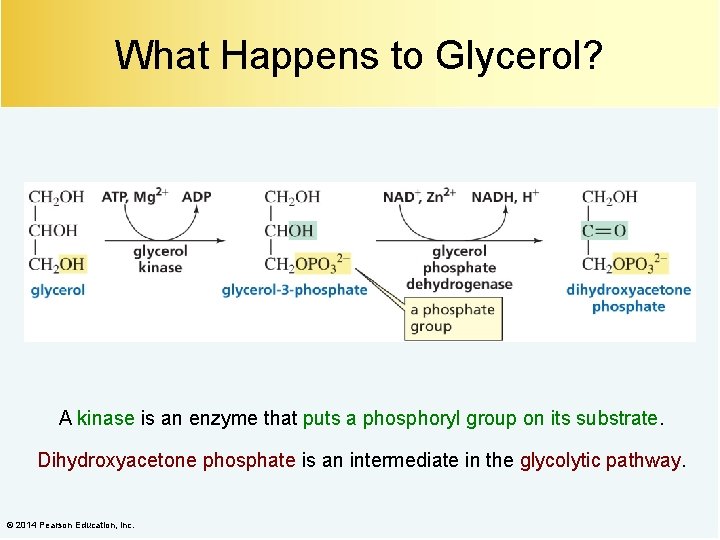
What Happens to Glycerol? A kinase is an enzyme that puts a phosphoryl group on its substrate. Dihydroxyacetone phosphate is an intermediate in the glycolytic pathway. © 2014 Pearson Education, Inc.
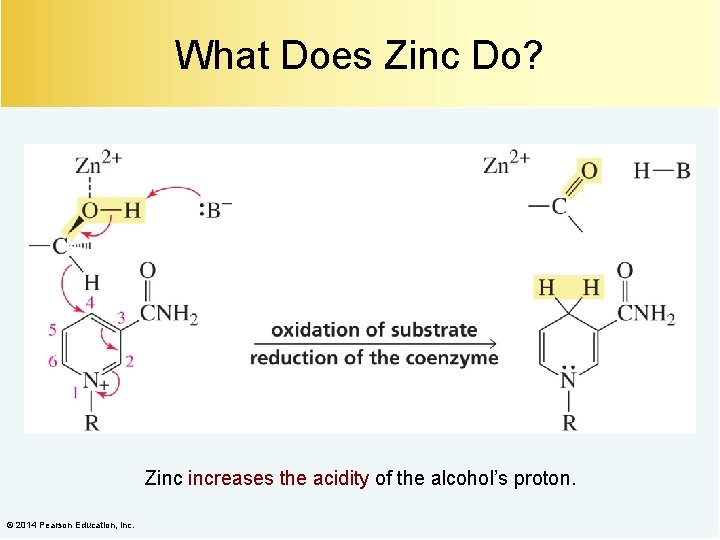
What Does Zinc Do? Zinc increases the acidity of the alcohol’s proton. © 2014 Pearson Education, Inc.
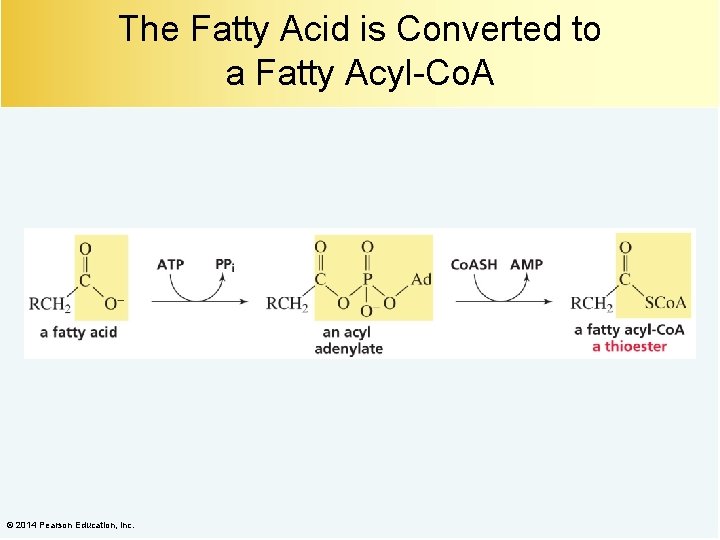
The Fatty Acid is Converted to a Fatty Acyl-Co. A © 2014 Pearson Education, Inc.
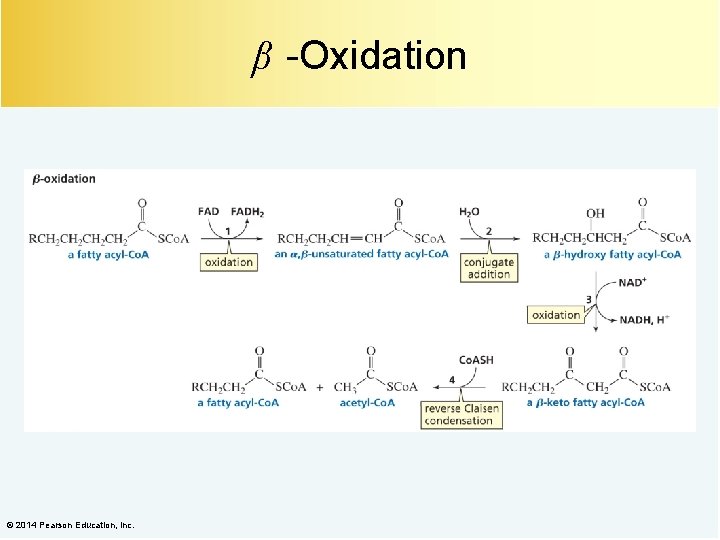
β -Oxidation © 2014 Pearson Education, Inc.
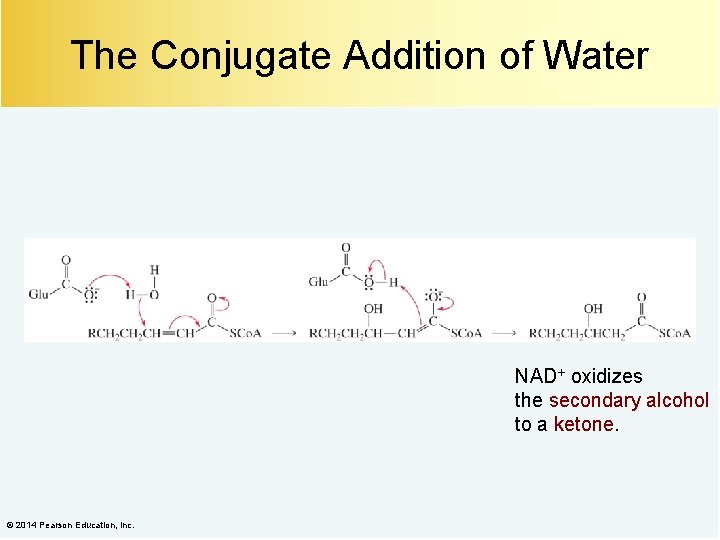
The Conjugate Addition of Water NAD+ oxidizes the secondary alcohol to a ketone. © 2014 Pearson Education, Inc.

A Reverse Claisen Condensation Beta-oxidation removes one acetyl-Co. A molecule at a time, converting the entire fatty acids to acetyl-Co. A molecules. © 2014 Pearson Education, Inc.

Catabolism of Carbohydrates In the first stage of catabolism, an enzyme-catalyzed reaction hydrolyzes the carbohydrate to molecules of glucose. © 2014 Pearson Education, Inc.
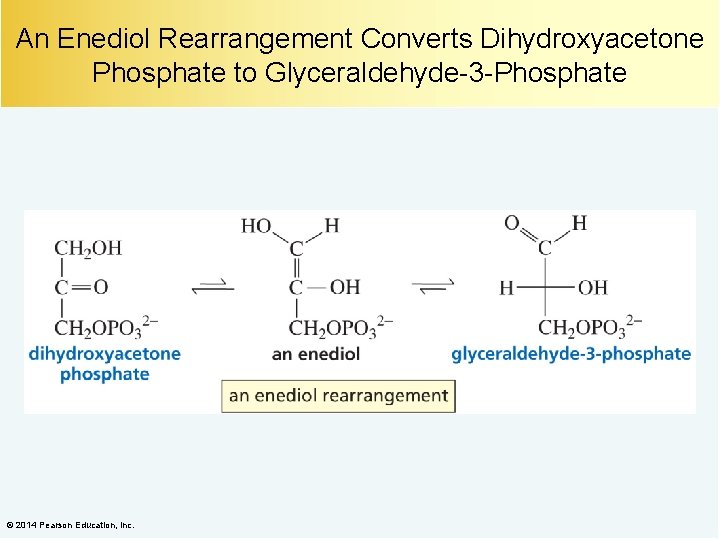
An Enediol Rearrangement Converts Dihydroxyacetone Phosphate to Glyceraldehyde-3 -Phosphate © 2014 Pearson Education, Inc.
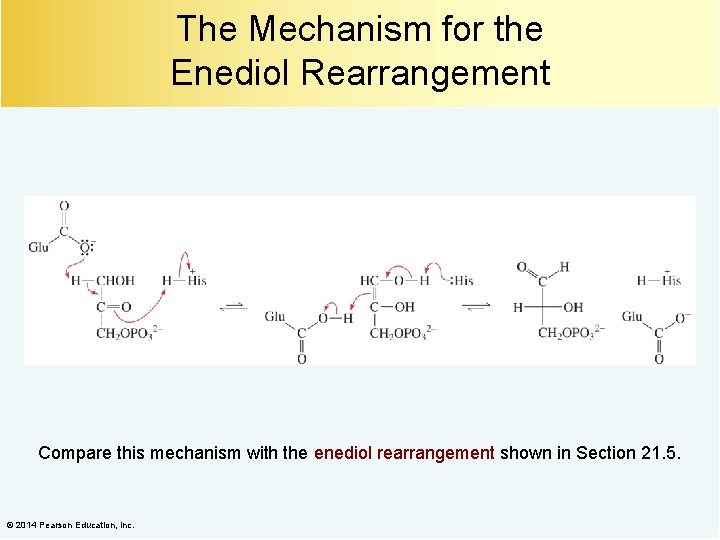
The Mechanism for the Enediol Rearrangement Compare this mechanism with the enediol rearrangement shown in Section 21. 5. © 2014 Pearson Education, Inc.
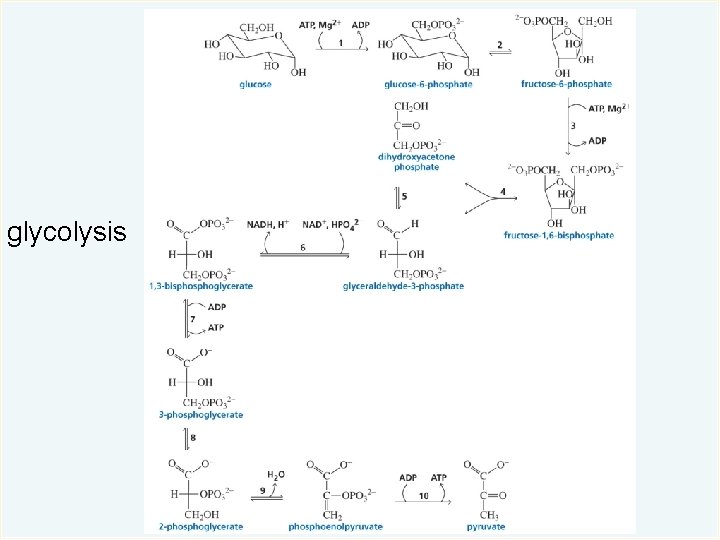
glycolysis
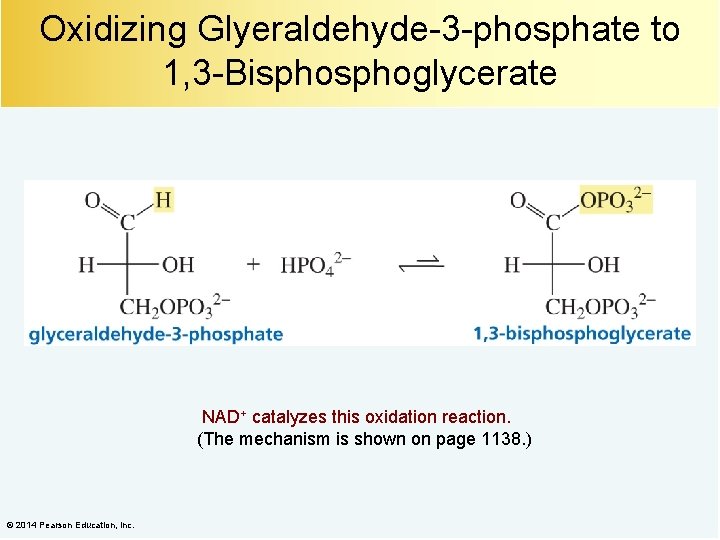
Oxidizing Glyeraldehyde-3 -phosphate to 1, 3 -Bisphoglycerate NAD+ catalyzes this oxidation reaction. (The mechanism is shown on page 1138. ) © 2014 Pearson Education, Inc.
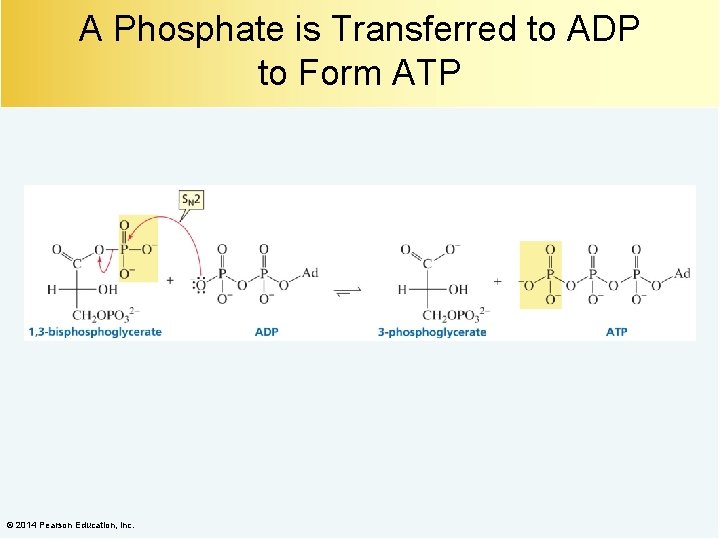
A Phosphate is Transferred to ADP to Form ATP © 2014 Pearson Education, Inc.
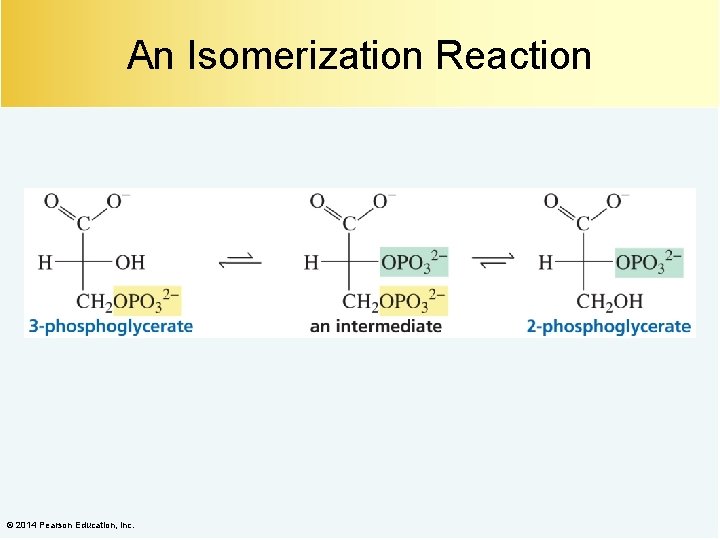
An Isomerization Reaction © 2014 Pearson Education, Inc.
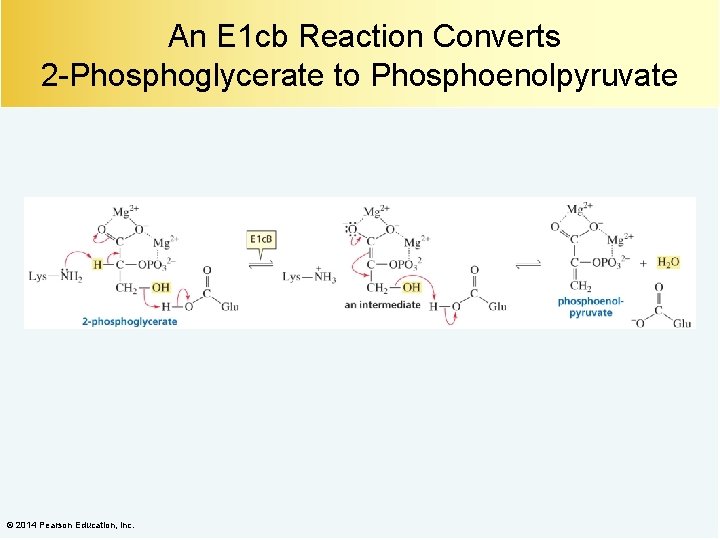
An E 1 cb Reaction Converts 2 -Phosphoglycerate to Phosphoenolpyruvate © 2014 Pearson Education, Inc.
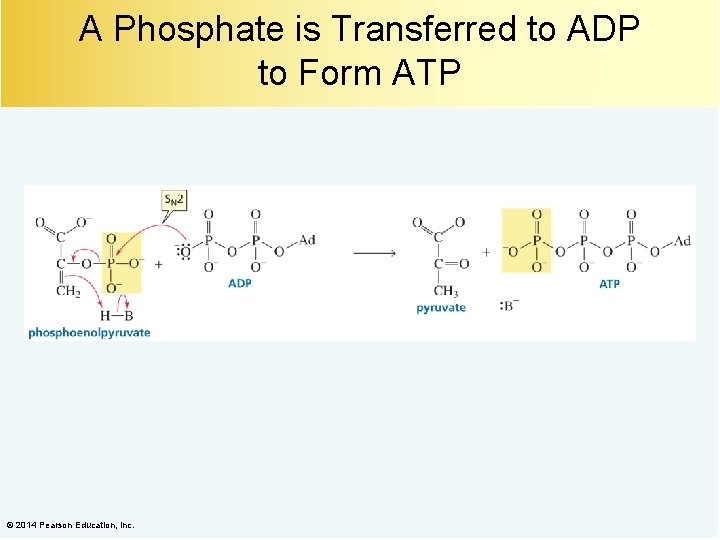
A Phosphate is Transferred to ADP to Form ATP © 2014 Pearson Education, Inc.
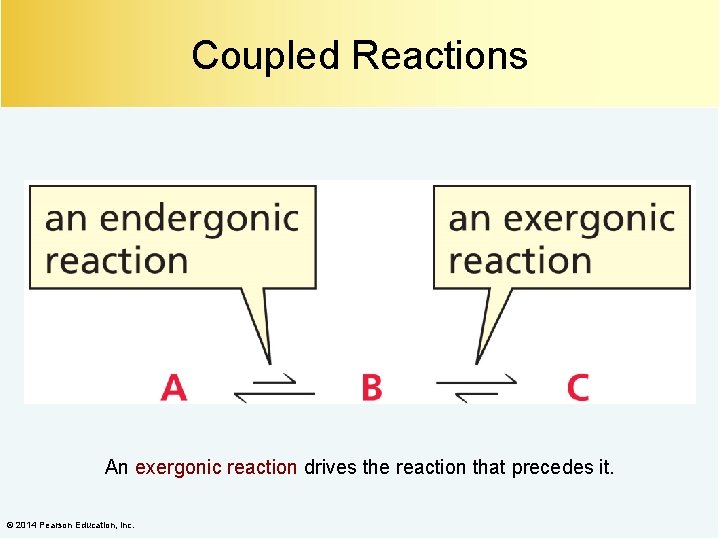
Coupled Reactions An exergonic reaction drives the reaction that precedes it. © 2014 Pearson Education, Inc.
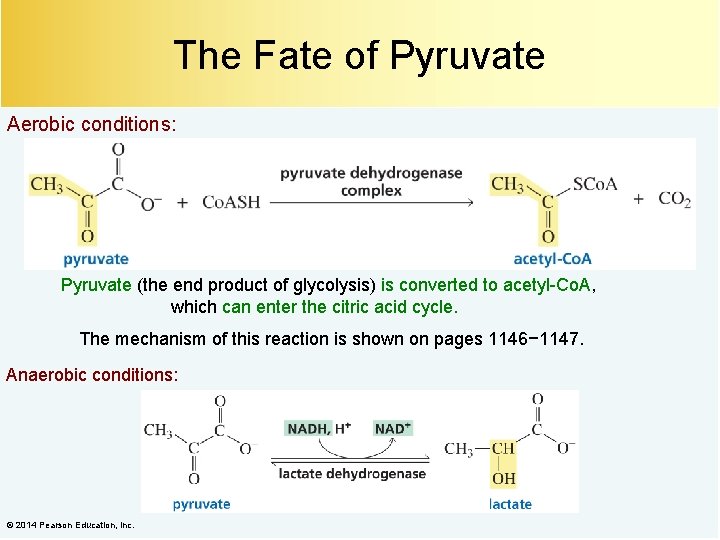
The Fate of Pyruvate Aerobic conditions: Pyruvate (the end product of glycolysis) is converted to acetyl-Co. A, which can enter the citric acid cycle. The mechanism of this reaction is shown on pages 1146− 1147. Anaerobic conditions: © 2014 Pearson Education, Inc.
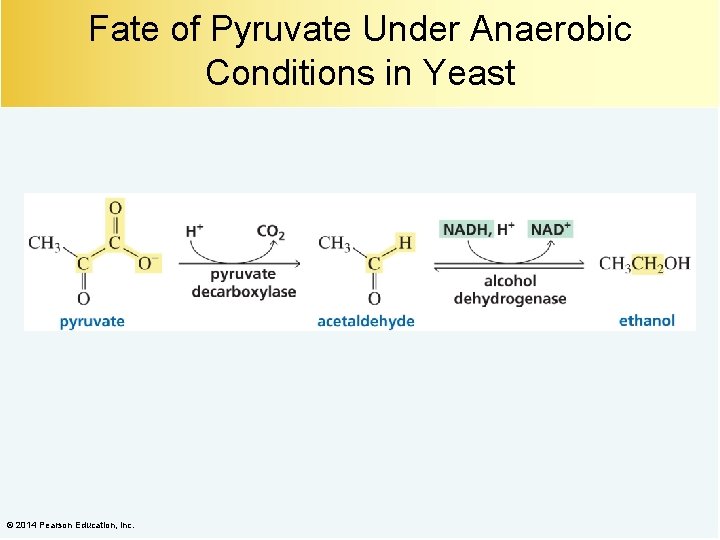
Fate of Pyruvate Under Anaerobic Conditions in Yeast © 2014 Pearson Education, Inc.
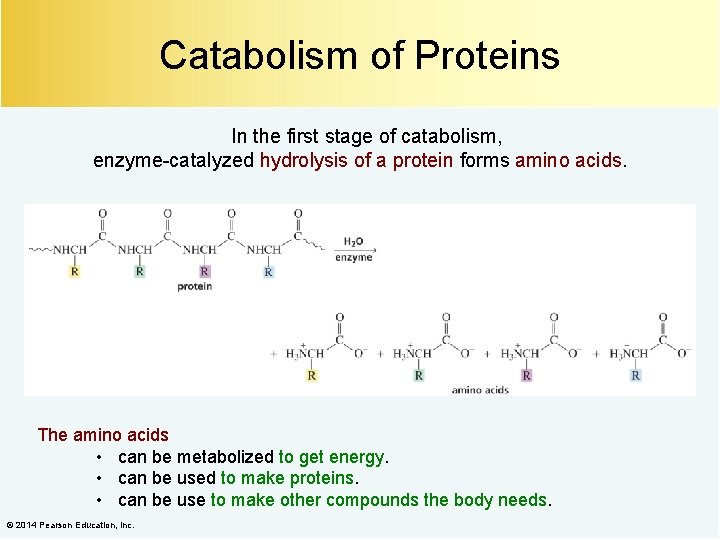
Catabolism of Proteins In the first stage of catabolism, enzyme-catalyzed hydrolysis of a protein forms amino acids. The amino acids • can be metabolized to get energy. • can be used to make proteins. • can be use to make other compounds the body needs. © 2014 Pearson Education, Inc.
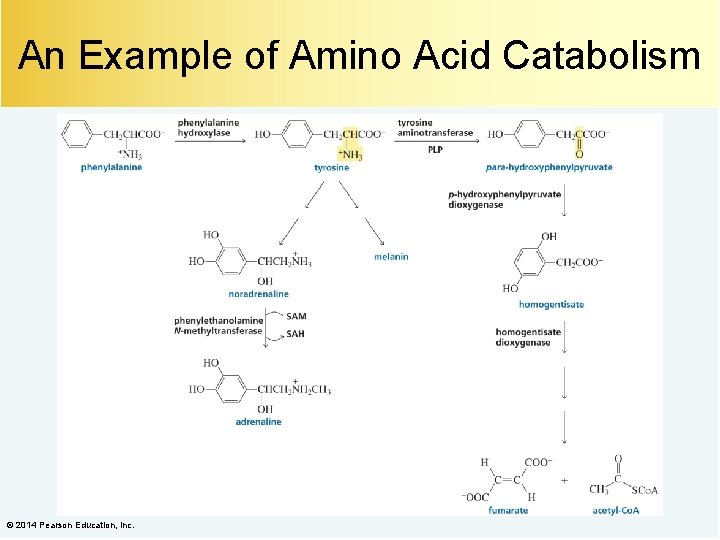
An Example of Amino Acid Catabolism © 2014 Pearson Education, Inc.
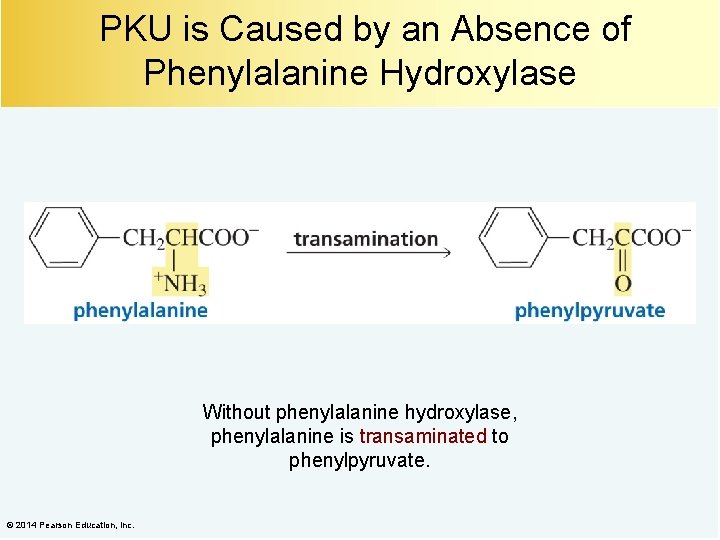
PKU is Caused by an Absence of Phenylalanine Hydroxylase Without phenylalanine hydroxylase, phenylalanine is transaminated to phenylpyruvate. © 2014 Pearson Education, Inc.

The Citric Acid Cycle The citric acid cycle converts the acetyl group of acetyl-Co. A to two molecules of CO 2. The series of eight reactions is called a cycle because the product of the eighth reaction is the reactant of the first reaction. © 2014 Pearson Education, Inc.
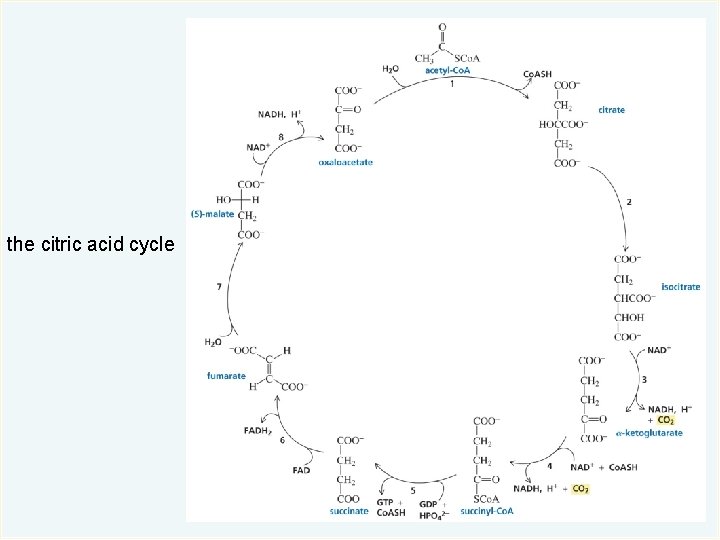
the citric acid cycle
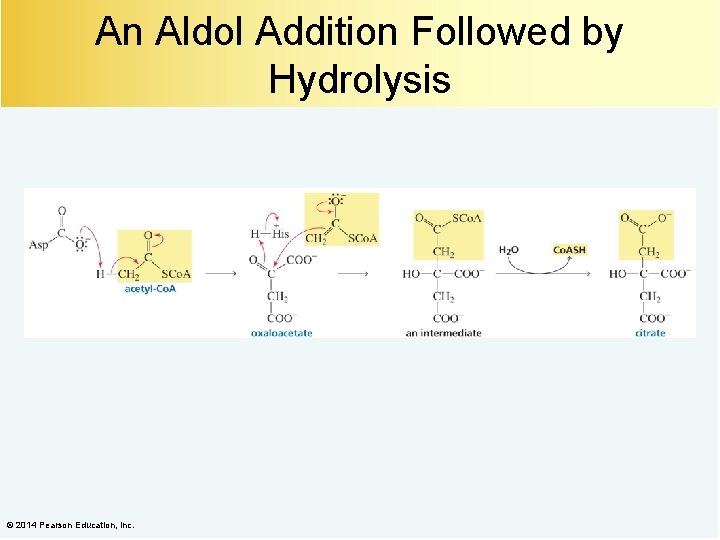
An Aldol Addition Followed by Hydrolysis © 2014 Pearson Education, Inc.
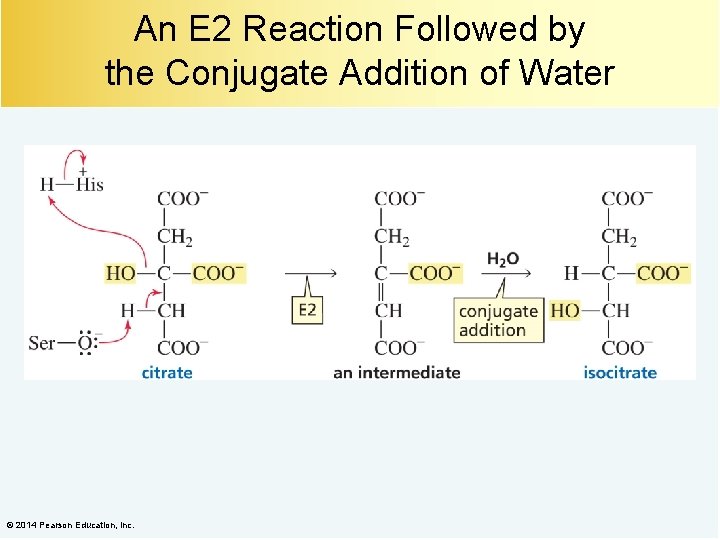
An E 2 Reaction Followed by the Conjugate Addition of Water © 2014 Pearson Education, Inc.
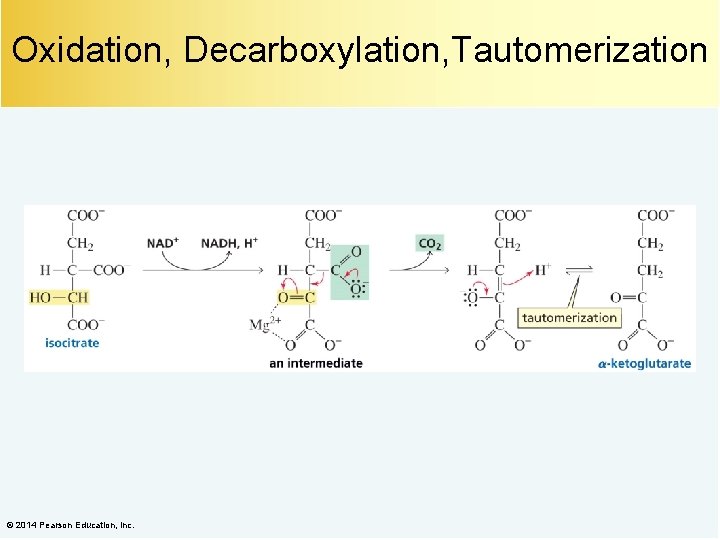
Oxidation, Decarboxylation, Tautomerization © 2014 Pearson Education, Inc.

The Overall Reaction is the Transfer of an Acyl Group to Co. ASH The mechanism for this reaction is the same as that for the pyruvate dehydrogenase complex (pp. 1146– 1147). © 2014 Pearson Education, Inc.

Succinyl-Co. A to Succinate A nucleophilic addition–elimination reaction followed by transfer of a phosphate group to GDP. © 2014 Pearson Education, Inc.
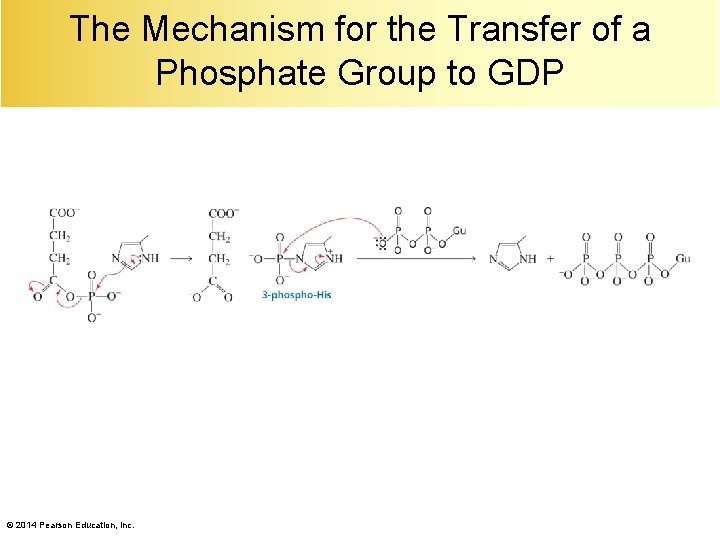
The Mechanism for the Transfer of a Phosphate Group to GDP © 2014 Pearson Education, Inc.
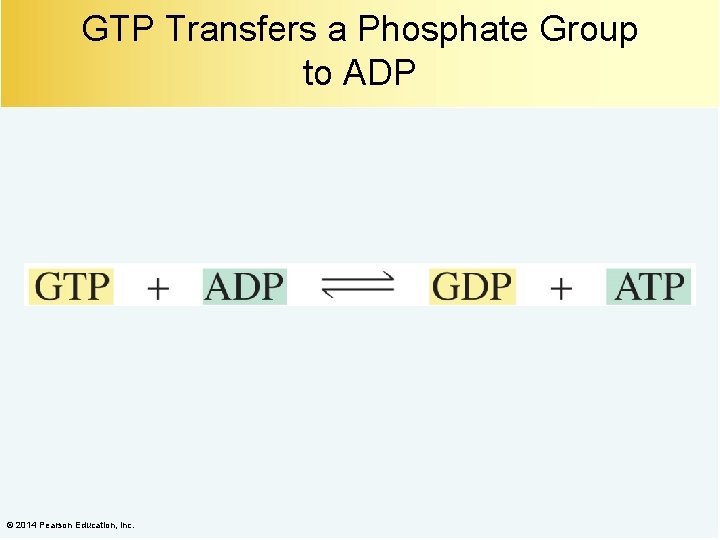
GTP Transfers a Phosphate Group to ADP © 2014 Pearson Education, Inc.
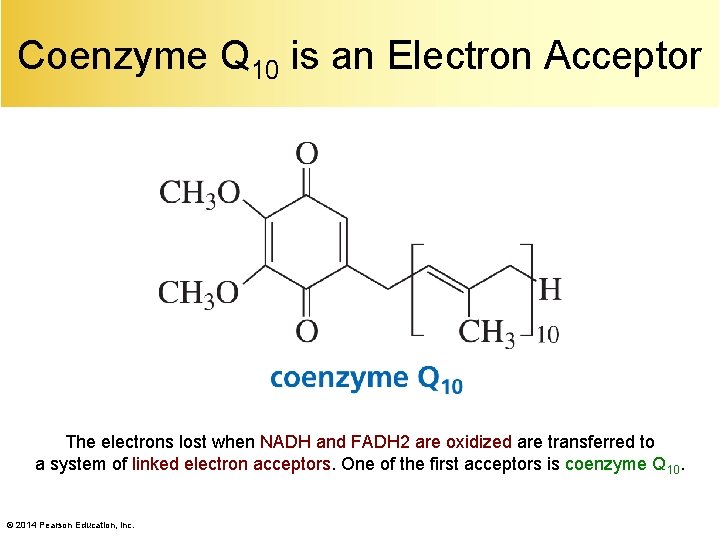
Coenzyme Q 10 is an Electron Acceptor The electrons lost when NADH and FADH 2 are oxidized are transferred to a system of linked electron acceptors. One of the first acceptors is coenzyme Q 10. © 2014 Pearson Education, Inc.

Oxidative Phosphorylation In the fourth stage of catabolism, NADH is oxidized back to NAD+ and FADH 2 is oxidized back to FAD, thereby forming ATP. © 2014 Pearson Education, Inc.

Each Round of the Citric Acid Cycle Forms 10 ATP © 2014 Pearson Education, Inc.
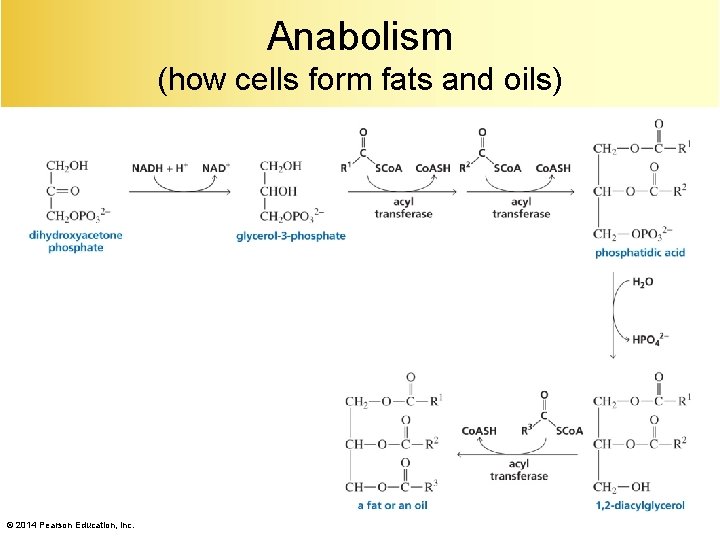
Anabolism (how cells form fats and oils) © 2014 Pearson Education, Inc.
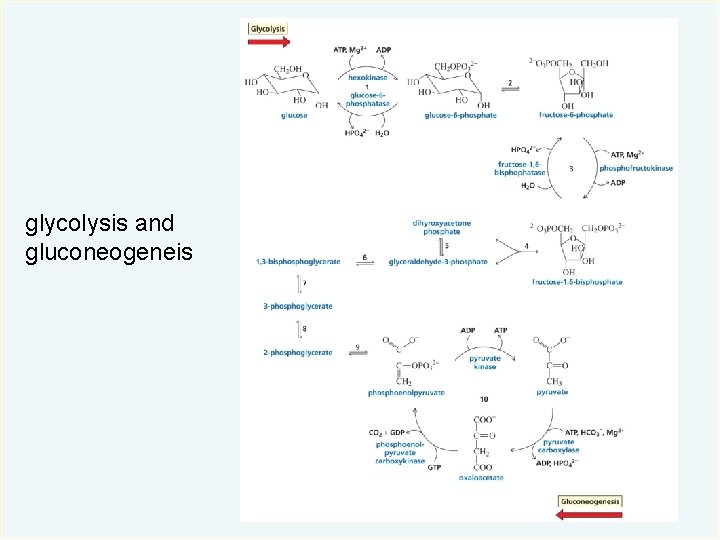
glycolysis and gluconeogeneis
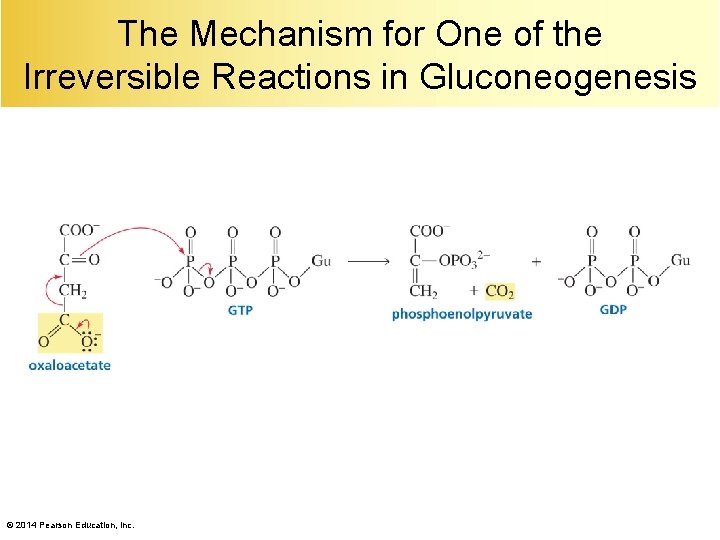
The Mechanism for One of the Irreversible Reactions in Gluconeogenesis © 2014 Pearson Education, Inc.
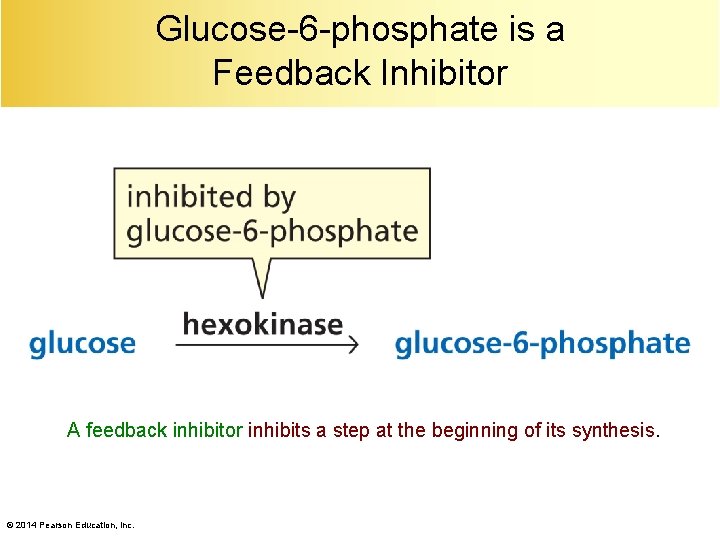
Glucose-6 -phosphate is a Feedback Inhibitor A feedback inhibitor inhibits a step at the beginning of its synthesis. © 2014 Pearson Education, Inc.
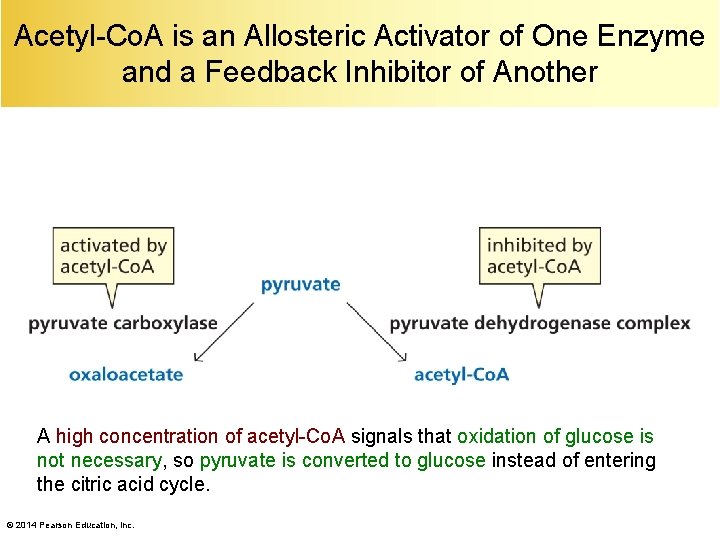
Acetyl-Co. A is an Allosteric Activator of One Enzyme and a Feedback Inhibitor of Another A high concentration of acetyl-Co. A signals that oxidation of glucose is not necessary, so pyruvate is converted to glucose instead of entering the citric acid cycle. © 2014 Pearson Education, Inc.

Amino Acid Biosynthesis © 2014 Pearson Education, Inc.

Biosynthesis of Serine oxidation, transamination, hydrolysis © 2014 Pearson Education, Inc.
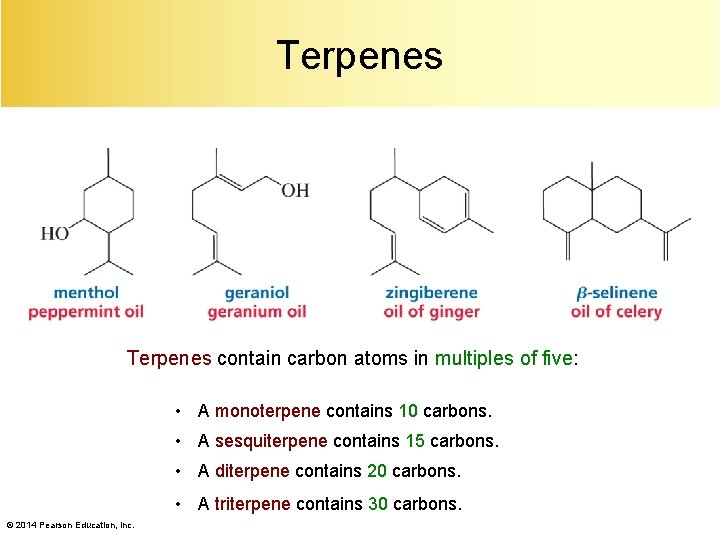
Terpenes contain carbon atoms in multiples of five: • A monoterpene contains 10 carbons. • A sesquiterpene contains 15 carbons. • A diterpene contains 20 carbons. • A triterpene contains 30 carbons. © 2014 Pearson Education, Inc.
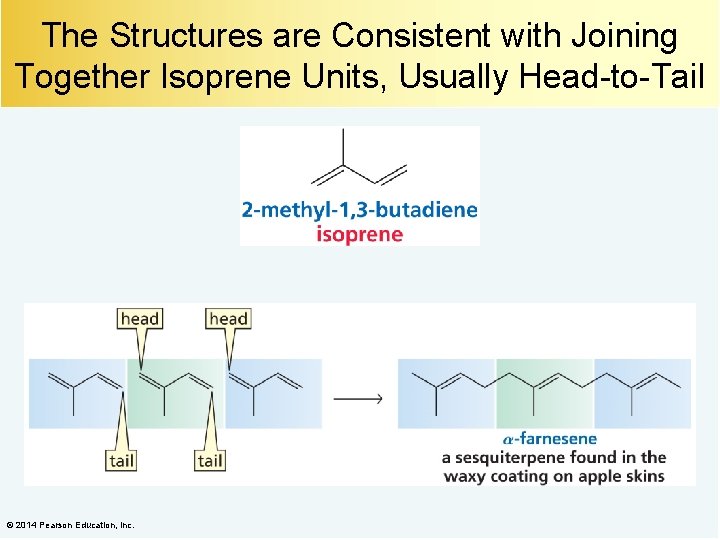
The Structures are Consistent with Joining Together Isoprene Units, Usually Head-to-Tail © 2014 Pearson Education, Inc.
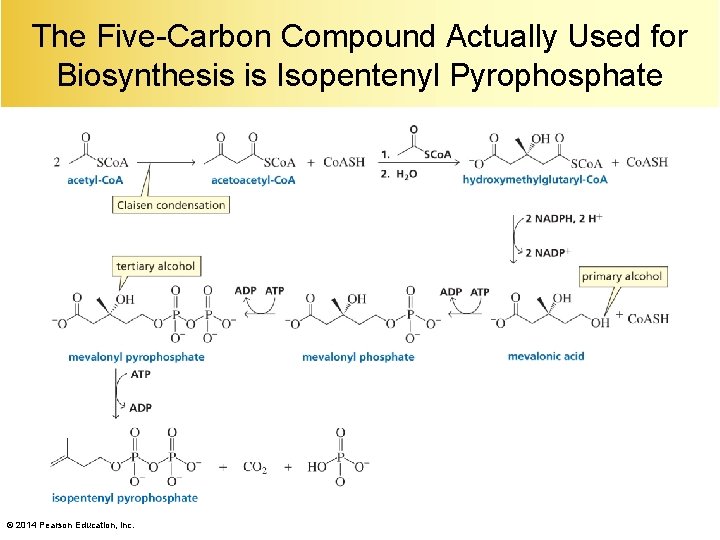
The Five-Carbon Compound Actually Used for Biosynthesis is Isopentenyl Pyrophosphate © 2014 Pearson Education, Inc.
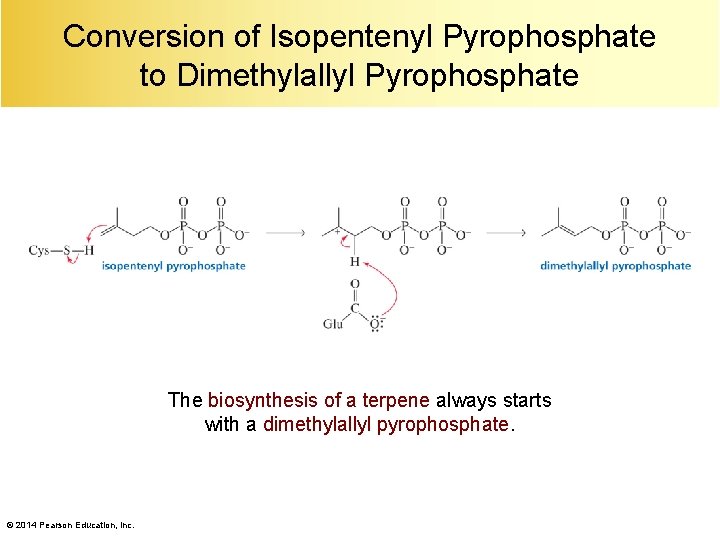
Conversion of Isopentenyl Pyrophosphate to Dimethylallyl Pyrophosphate The biosynthesis of a terpene always starts with a dimethylallyl pyrophosphate. © 2014 Pearson Education, Inc.
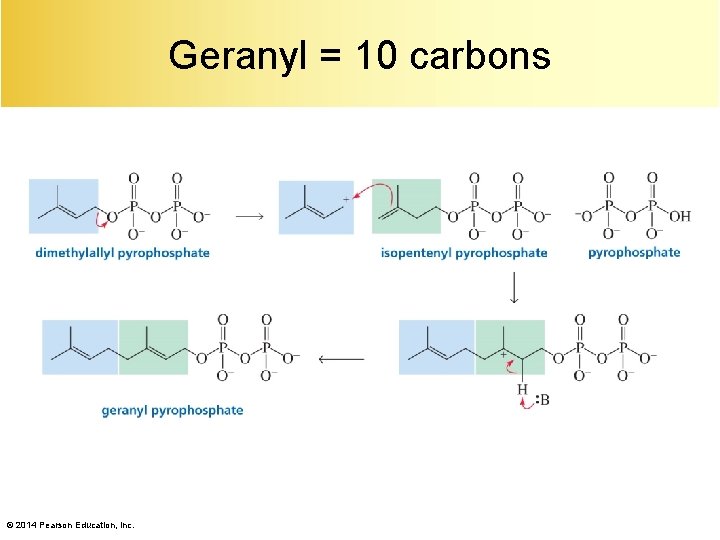
Geranyl = 10 carbons © 2014 Pearson Education, Inc.
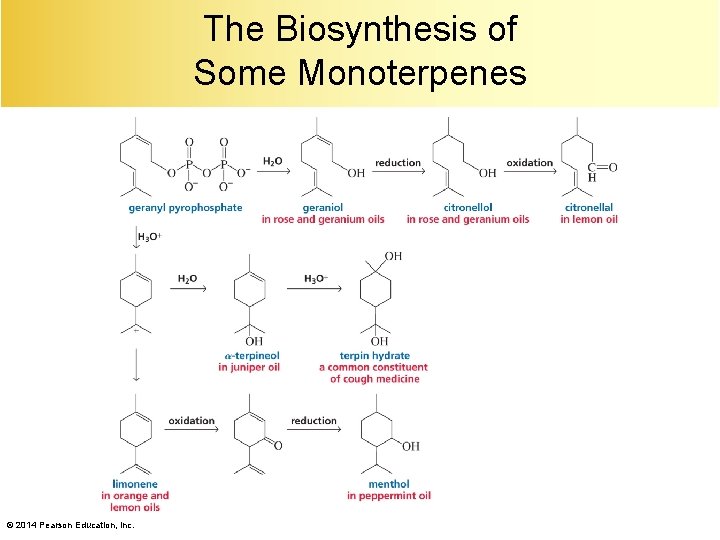
The Biosynthesis of Some Monoterpenes © 2014 Pearson Education, Inc.
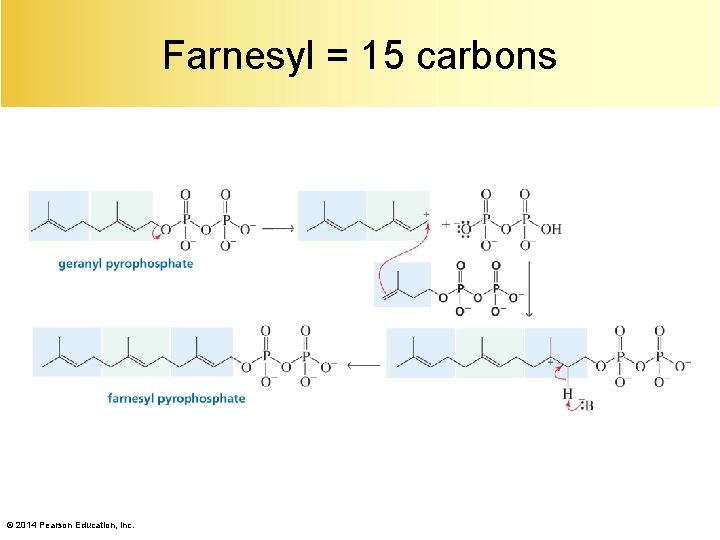
Farnesyl = 15 carbons © 2014 Pearson Education, Inc.
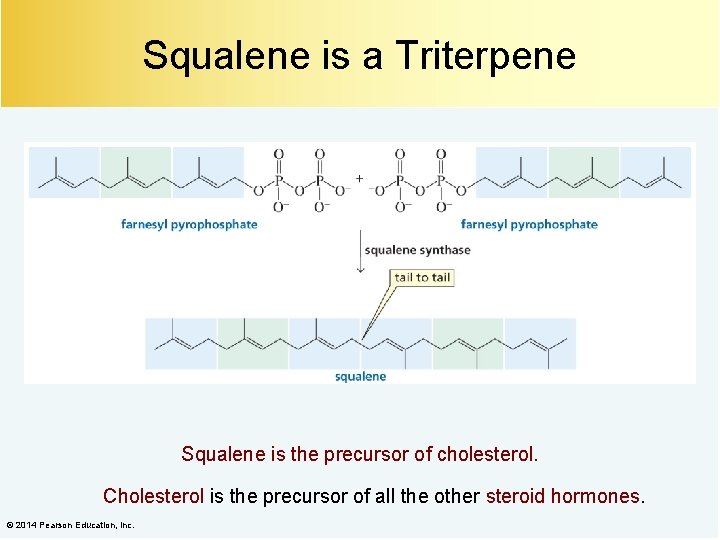
Squalene is a Triterpene Squalene is the precursor of cholesterol. Cholesterol is the precursor of all the other steroid hormones. © 2014 Pearson Education, Inc.
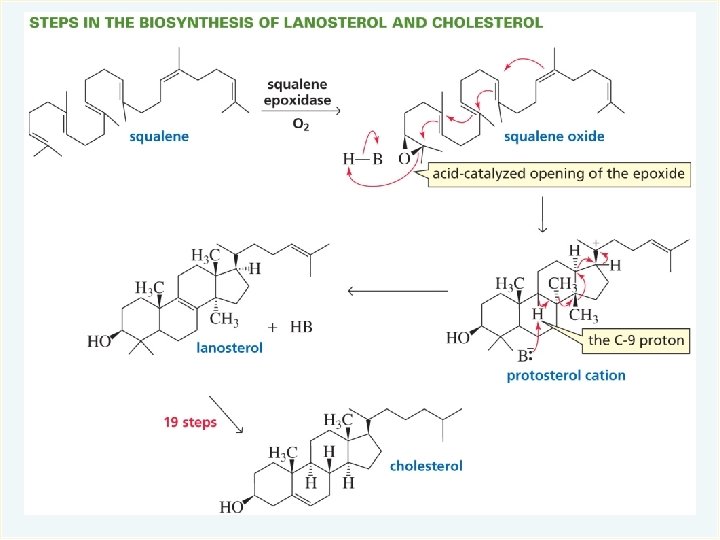
- Slides: 64