Bonding Between Atoms Why Do Atoms Form Bonds

Bonding Between Atoms

Why Do Atoms Form Bonds l To get a stable octet of valence electrons. l Called a “noble gas configuration”

Changes in Energy l Energy is released when bonds form. Exothermic = Bond Forms l Exothermic l Lower energy = more stable Endothermic = Bond Breaks

Bond Formation is EXOTHERMIC

Major Types of Bonds
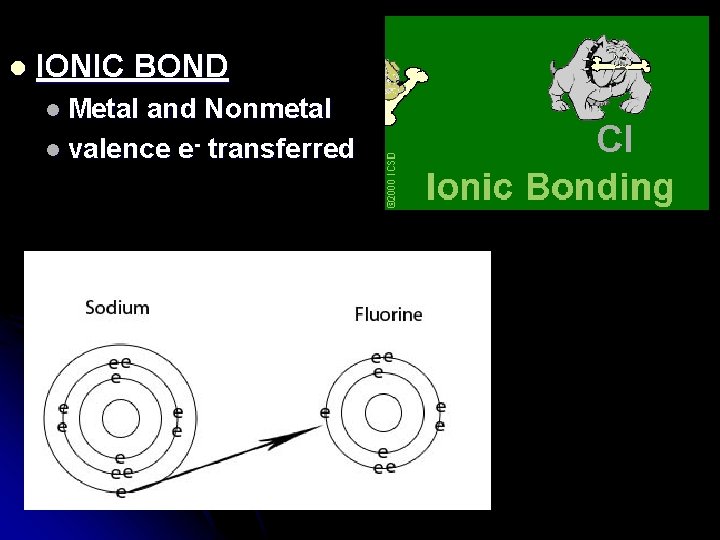
l IONIC BOND l Metal and Nonmetal l valence e- transferred
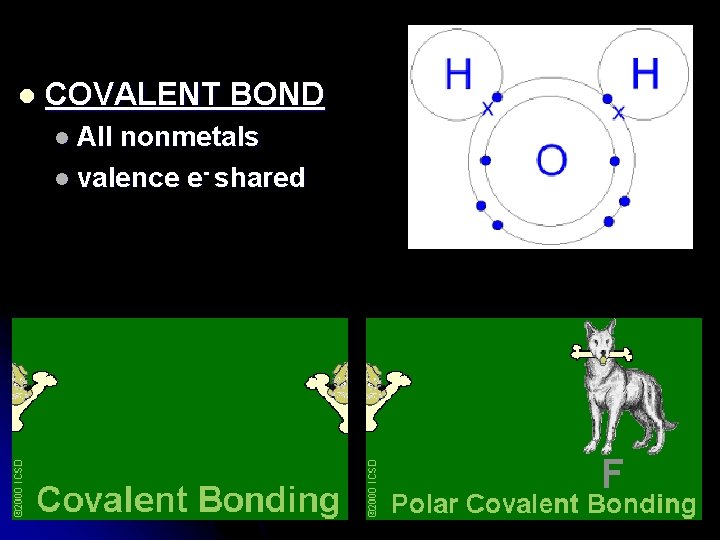
l COVALENT BOND l All nonmetals l valence e- shared
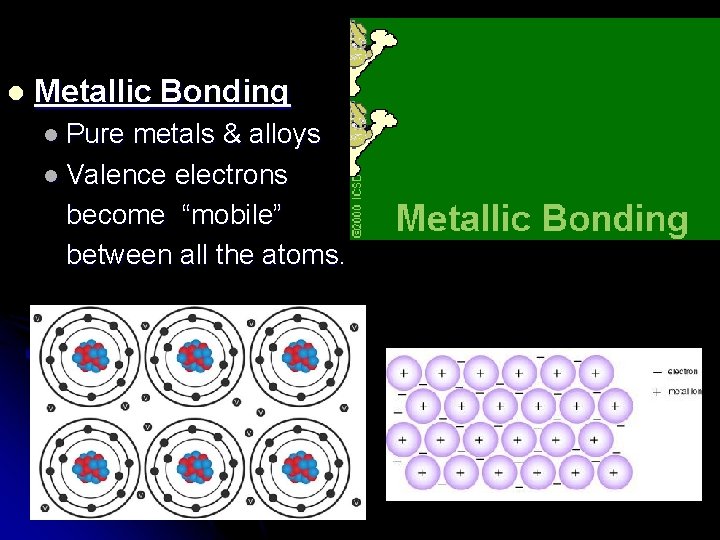
l Metallic Bonding l Pure metals & alloys l Valence electrons become “mobile” between all the atoms.

What Type of Bonding? _____Ca. Cl 2 l _____C 6 H 12 O 6 l _____ CO 2 l _____ Fe l _____ H 2 O l _____ Al(NO 3)3 l _____ Mg l _____ NH 3 l

Lewis Dot Diagrams of Atoms l Show valence electrons involved in bonding

Lewis Dot Diagrams of Ions l Positive Ions (cations): lose outer shell valence to get stable octet. l Negative Ions (anions): gain electrons to complete outer shell octet. Do Bonding Packet Pgs 4, 5, 6

Ionic Bonding Crash Course: https: //www. youtube. com/watch? v=QXT 4 OVM 4 v. XI
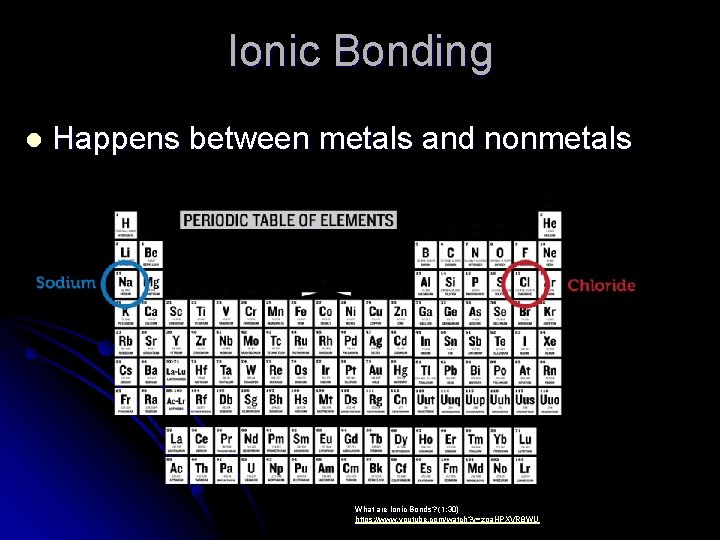
Ionic Bonding l Happens between metals and nonmetals What are Ionic Bonds? (1: 30) https: //www. youtube. com/watch? v=zpa. HPXVR 8 WU

Electronegativity Difference l Ionic compounds: large differences in atomic EN values ( > 1. 7) Greater EN diff. = more “Ionic Character”.

l Ex: EN Values l Metal Na = 0. 9 l Nonmetal Cl = 3. 2 l Nonmetal with higher EN “takes” electron(s) from metal

Atoms Become Ions l Metals Lose Electrons: l become (+) ion l Ex: Mg is 2 -8 -2 l Loses 2 electrons to become Mg+2 2 -8 l Nonmetals Gain Electrons: usually take top (-) charge l become (–) ion l Ex: Cl is 2 -8 -7 l Gains 1 electron to become Cl-1 2 -8 -8
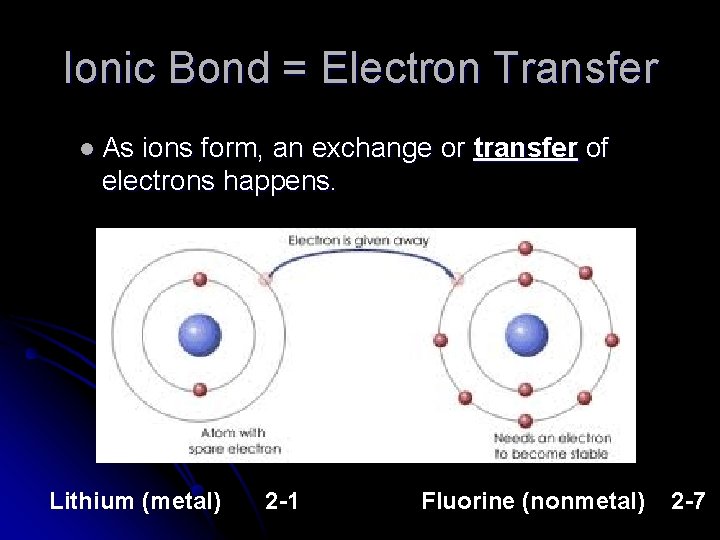
Ionic Bond = Electron Transfer l As ions form, an exchange or transfer of electrons happens. Lithium (metal) 2 -1 Fluorine (nonmetal) 2 -7

l Positively and negatively charged ions form and attract each other due to OPPOSITE CHARGES. + - ion IONIC BOND l l Forming Table Salt : 6 minutes http: //science 360. gov/obj/video/c 8 b 6 ed 58 -5791 -4 fc 1 -8619 -62 fd 8 d 99 d 9 d 0/atoms-bond-ionic-bonds
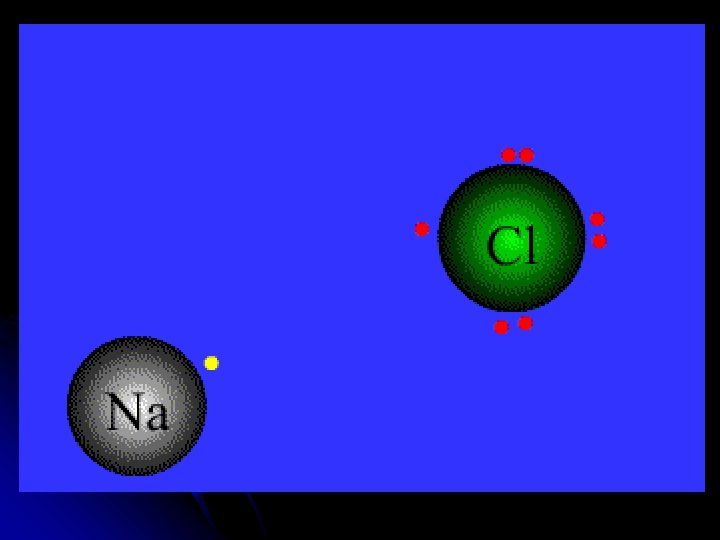

l They transfer enough electrons so that all get a stable octet of valence!
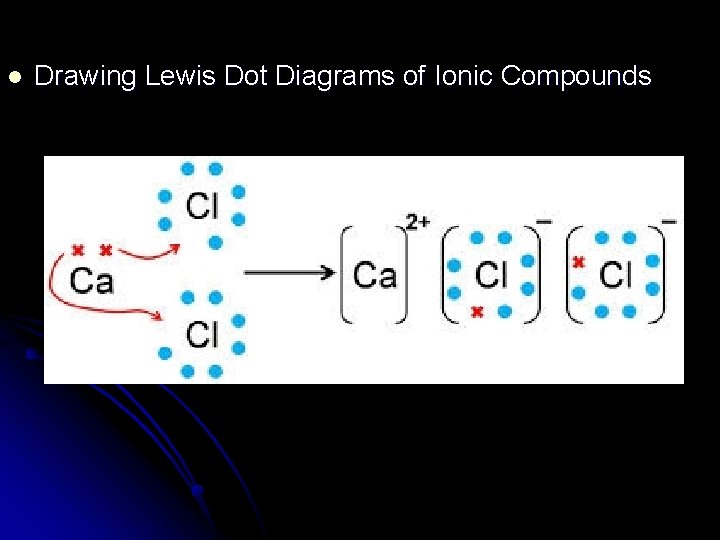
l Drawing Lewis Dot Diagrams of Ionic Compounds
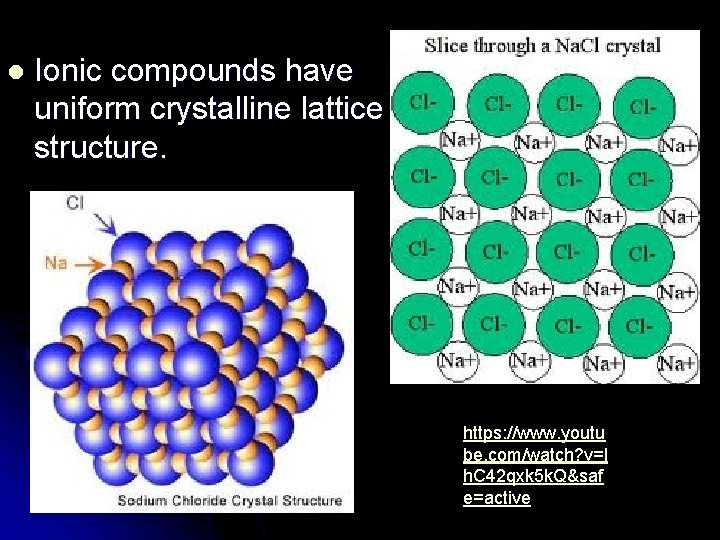
l Ionic compounds have uniform crystalline lattice structure. https: //www. youtu be. com/watch? v=l h. C 42 qxk 5 k. Q&saf e=active
l Can you identify an ionic compound from a covalent? l Ionic = Metal with nonmetals _______Mg. Br 2 l _______Ca(NO 3)2 l _______P 2 O 5 l _______Si. O 2 l _______Na 2 S l _______H 2 S l

Ionic Compounds are Neutral “Subscripts” show ratio of ions Total positive and negative charges must balance Compound is electrically neutral.

Writing Neutral Ionic Formulas l l Write symbols of elements with charge Criss-Cross charges if necessary to balance the formula l l Put parenthesis around polyatomic ions if more than one in formula Simplify if needed. Tutorial on writing ionic formulas 9 min http: //www. youtube. com/watch? v=vsco. Yh 6 m 46 M

Stock System for Naming Ionic Compounds l Binary: Contain ions of 2 elements Ex: Mg. Cl 2, Al 2 O 3, Na. Cl

Naming Binary Ionics l Name metal l Name nonmetal with “-ide” ending. l Note: l Name never indicates # of ions Ex: Na. Cl, Mg. Br 2, Ca. O, Al 2 S 3 Writing Ionic Formulas: (3: 22) https: //www. youtube. com/watch? v=vf. Ynhnfds. D 0

l Ternary Ionics: Contain 3 elements l Polyatomic ion present. l Note: Elements inside the polyatomic ion are covalently bonded (all nonmetals). l These compounds contain both ionic and covalent bonds!! Mg. SO 4

Naming Ternary Ionics (with polyatomic) l Name Metal l Name polyatomic ion l If two polyatomics, name them both l Ex: Na. NO 3, Ca 3(PO 4)2, NH 4 Cl Ionics with Polyatomic Ions: (3: 46) https: //www. youtube. com/watch? v=Jyl. Jw. Hrye. SU
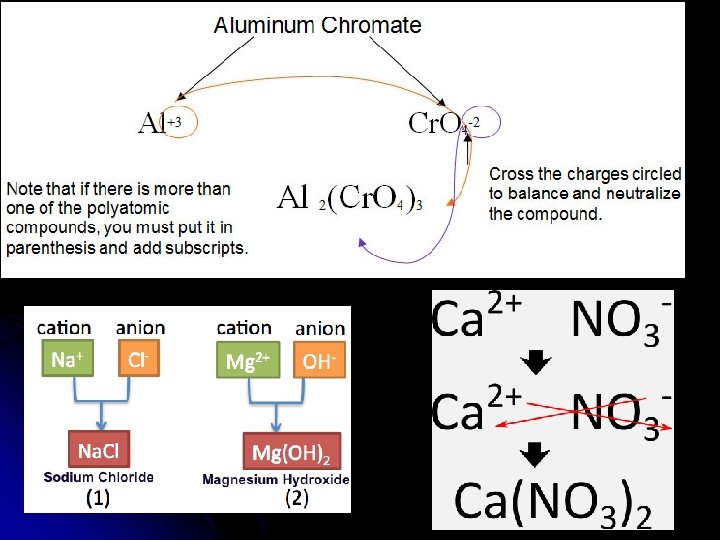

l Why does the 3 rd compound use a Roman Numeral in it’s name?

When to Use a Roman Numeral l Roman numerals are used when the metal can have more than one possible charge. l Roman numeral indicates the charge the metal takes in that compound and must be indicated in the name Ex: Ni. Br 2 Ni. Br 3 Nickel II Bromide Nickel III Bromide

l Name the following ionic compounds: l Fe(NO 3)3 __________ l Na. F _____________ l Cu 3 P ____________ l Al 2(SO 4)3 __________

l Write the correct formula for: l _______cobalt III oxide l _______magnesium sulfide l _______ lead II nitrate l _______ calcium phosphate

Properties of Ionic Compounds
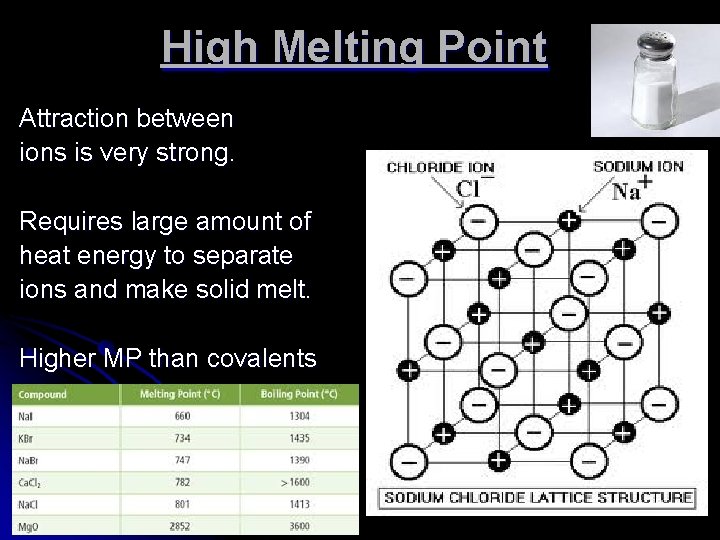
High Melting Point Attraction between ions is very strong. Requires large amount of heat energy to separate ions and make solid melt. Higher MP than covalents

Solubility in Water l Most ionics will dissolve in water, or be “soluble”. l When dissolved in water they are “aqueous” Ex: Na. Cl (aq)

l Water is a “polar molecule”. Acts like magnet to pull ions apart and into solution. l Ions are now “dissociated” or “hydrated” ions. l lhttp: //youtu. be/EBf. Gc. TAJF 4 o

Conductivity l Ionic compounds conduct when ions are “mobile” or free to move about. l Ionic compounds conduct when: l Molten (melted or liquid) (l) l Aqueous (aq)

Molten Ionics can Conduct!
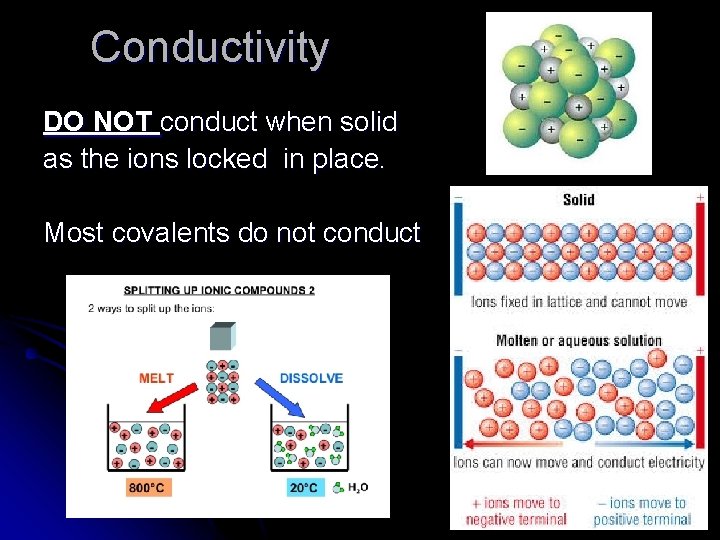
Conductivity DO NOT conduct when solid as the ions locked in place. Most covalents do not conduct

l Which compound has the highest Melting point? KCl or SO 2 l Which substances conduct electricity? Na. Cl (s) CO 2 (g) Mg. Br 2 (l) C 6 H 12 O 6 (s) Li. NO 3 (aq)

Lattice Energy (Honors) Energy released when ionic bond is formed l The more energy, the more stable and stronger the bond. l Related to Coulombic Attraction l l Variables that Determine its Magnitude: l Atomic Size l Ion Charge l See Honors Packet Pages

Crash Course Chemistry: Nomenclature (only watch first 6 minutes, skip section on naming acids for now) http: //www. youtube. com/watch? v=ml. Rh. Lic. No 8 Q Dancing Queen: Song about Ionic vs Covalent Bonding http: //www. youtube. com/watch? v=QIf. TT-_-x. Lo Crash Course: Atomic Hookups http: //www. youtube. com/watch? v=QXT 4 OVM 4 v. XI
- Slides: 44