The Chemical Foundations of Life Chapter 2 pages













- Slides: 52

The Chemical Foundations of Life Chapter 2 (pages 22 -44)

Major Topics • Atomic structure • Chemical bonds – Valence electrons – Ionic, covalent, hydrogen • Molecules in cells – Sugars, fatty acids, amino acids, nucleotides • Macromolecules in cells – Sugars, lipids, proteins, nucleic acids

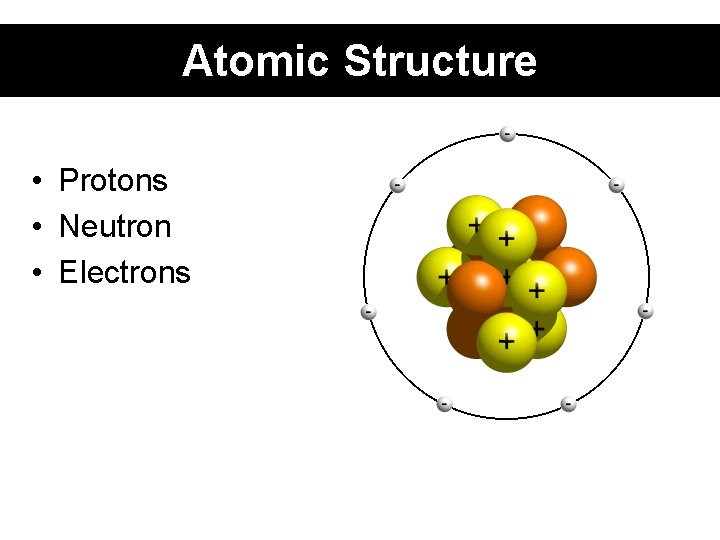
Atomic Structure • Protons • Neutron • Electrons

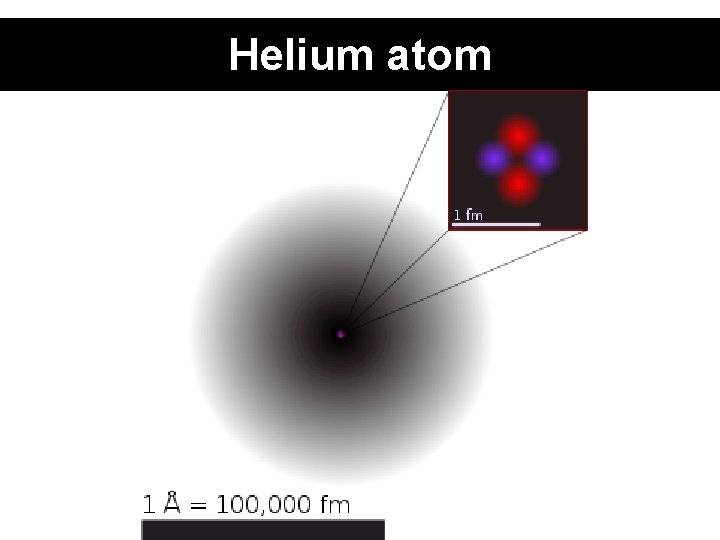
Helium atom

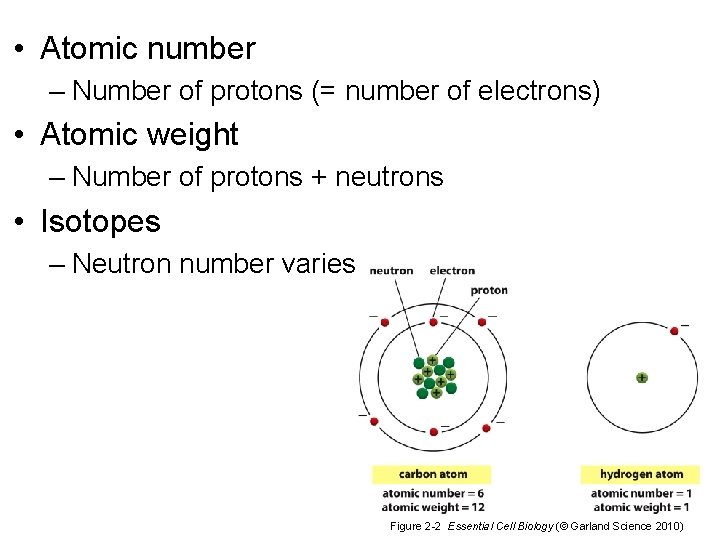
• Atomic number – Number of protons (= number of electrons) • Atomic weight – Number of protons + neutrons • Isotopes – Neutron number varies Figure 2 -2 Essential Cell Biology (© Garland Science 2010)

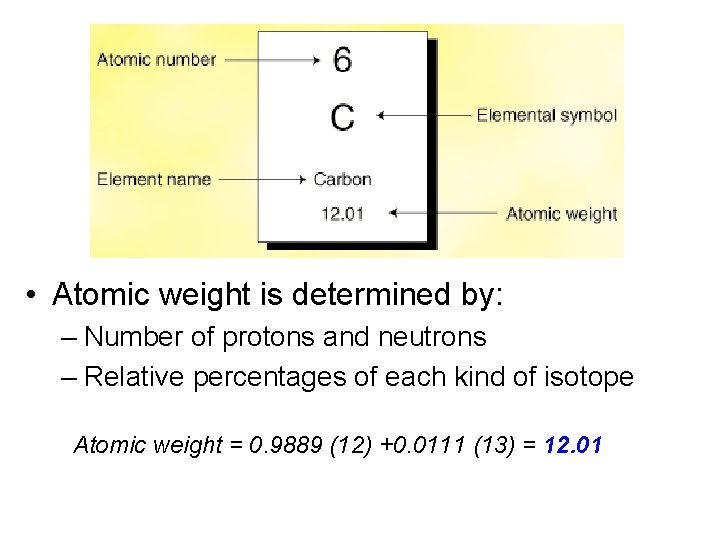
• Atomic weight is determined by: – Number of protons and neutrons – Relative percentages of each kind of isotope Atomic weight = 0. 9889 (12) +0. 0111 (13) = 12. 01

The proton shrinks in size! • 2010 paper in Nature • Proton 0. 00000003 mm smaller than thought! – 4% smaller! • Fired “muons” at hydrogen atoms and measured energy

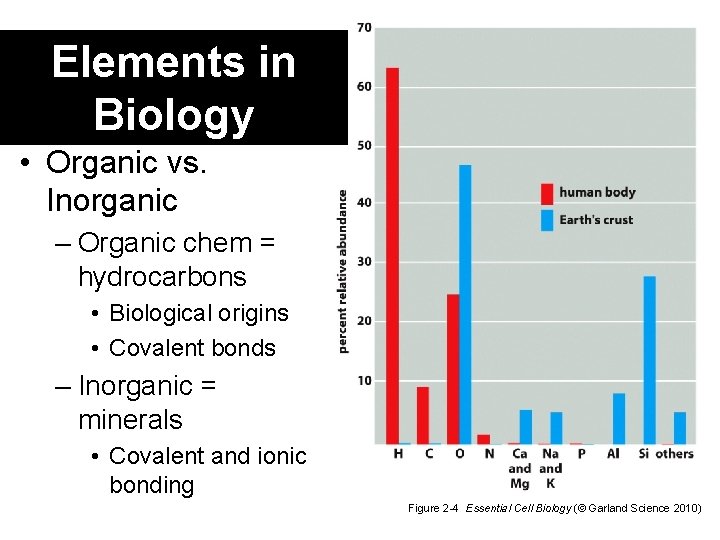
Elements in Biology • Organic vs. Inorganic – Organic chem = hydrocarbons • Biological origins • Covalent bonds – Inorganic = minerals • Covalent and ionic bonding Figure 2 -4 Essential Cell Biology (© Garland Science 2010)

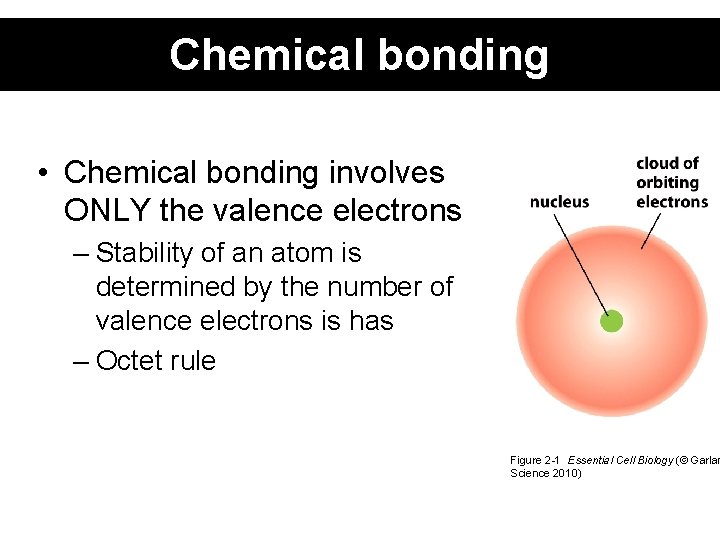
Chemical bonding • Chemical bonding involves ONLY the valence electrons – Stability of an atom is determined by the number of valence electrons is has – Octet rule Figure 2 -1 Essential Cell Biology (© Garlan Science 2010)

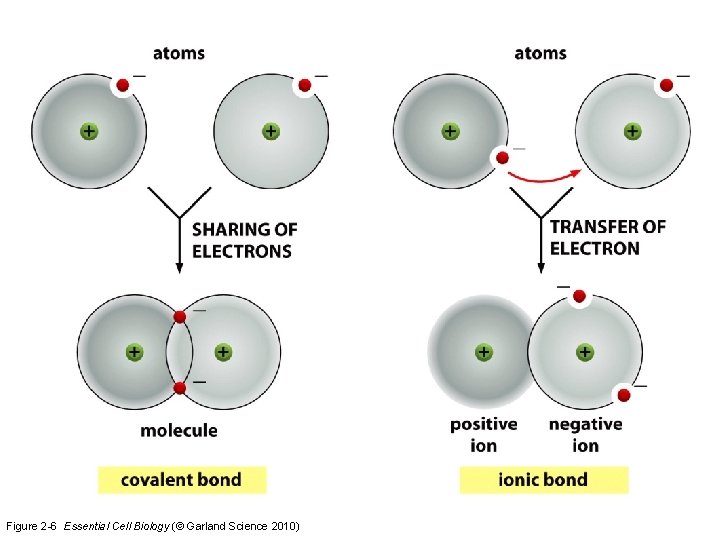
Chemical bonding in biology • Valence electron number determines if chemical bonding occurs – Octet rule: Atoms are happiest with a complete octet in their valence ring • Options to get that octet: – Share electrons – Give away electrons – Accept electrons Covalent bonding Ionic bonding

Figure 2 -6 Essential Cell Biology (© Garland Science 2010)

Covalent bonding • Predominant type of bonding in organic chemistry – Carbon can form four covalent bonds • Two types: – Polar – Nonpolar

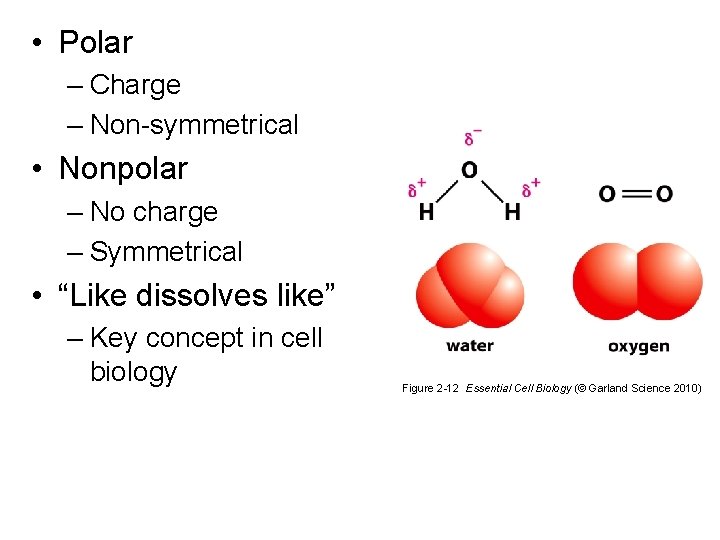
• Polar – Charge – Non-symmetrical • Nonpolar – No charge – Symmetrical • “Like dissolves like” – Key concept in cell biology Figure 2 -12 Essential Cell Biology (© Garland Science 2010)


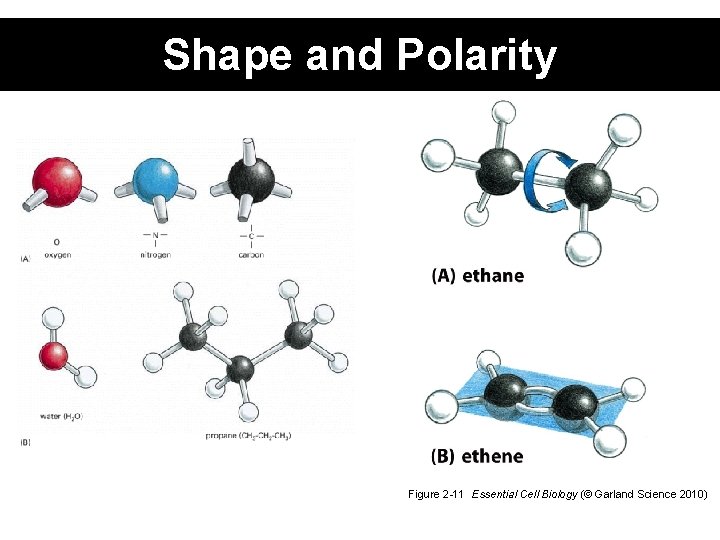
Shape and Polarity Figure 2 -11 Essential Cell Biology (© Garland Science 2010)

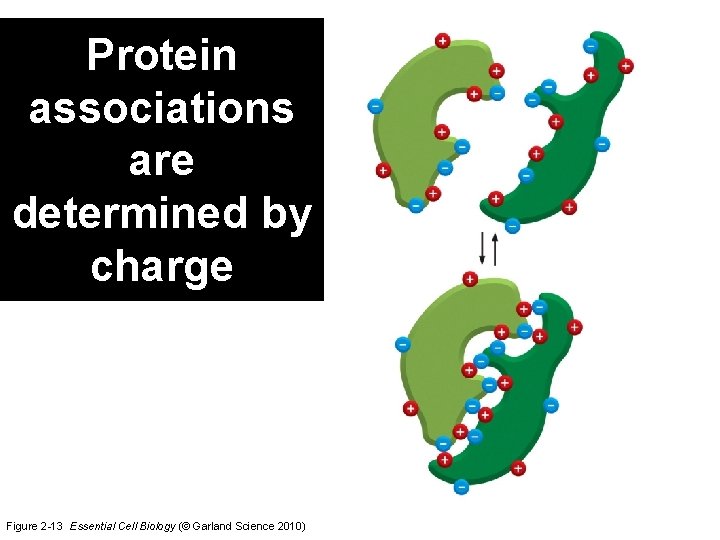
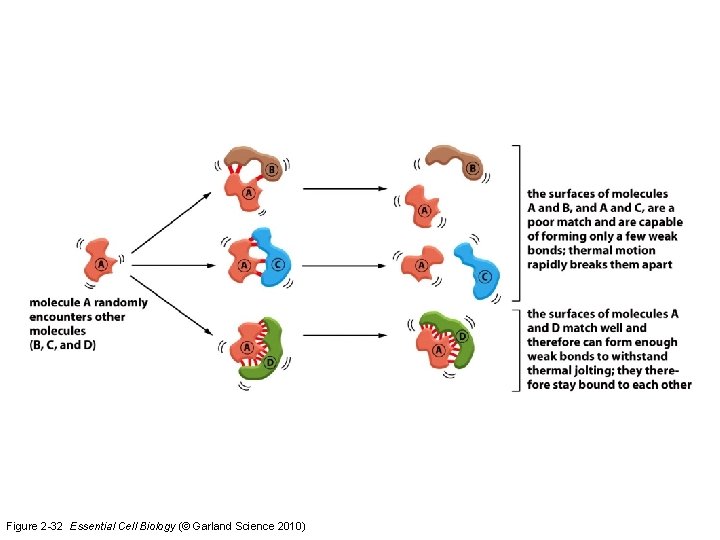
Protein associations are determined by charge Figure 2 -13 Essential Cell Biology (© Garland Science 2010)

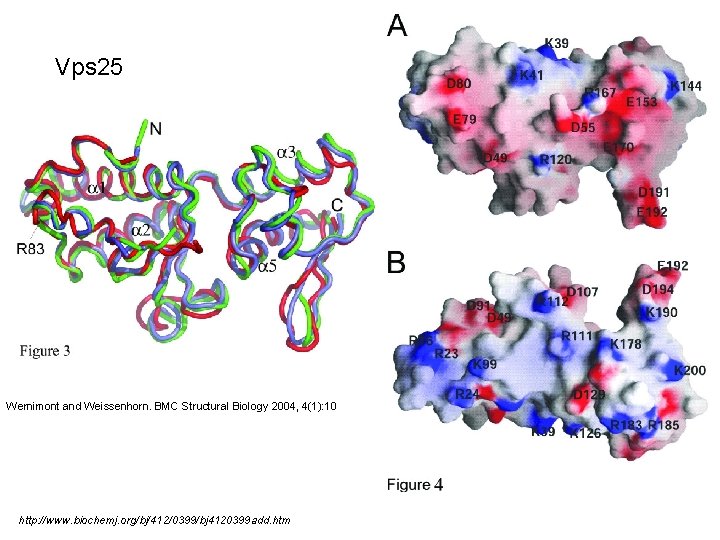
Vps 25 Wernimont and Weissenhorn. BMC Structural Biology 2004, 4(1): 10 http: //www. biochemj. org/bj/412/0399/bj 4120399 add. htm

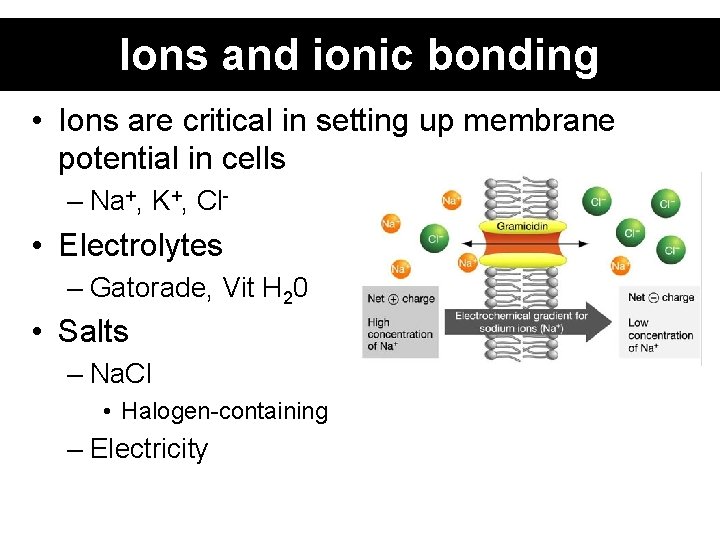
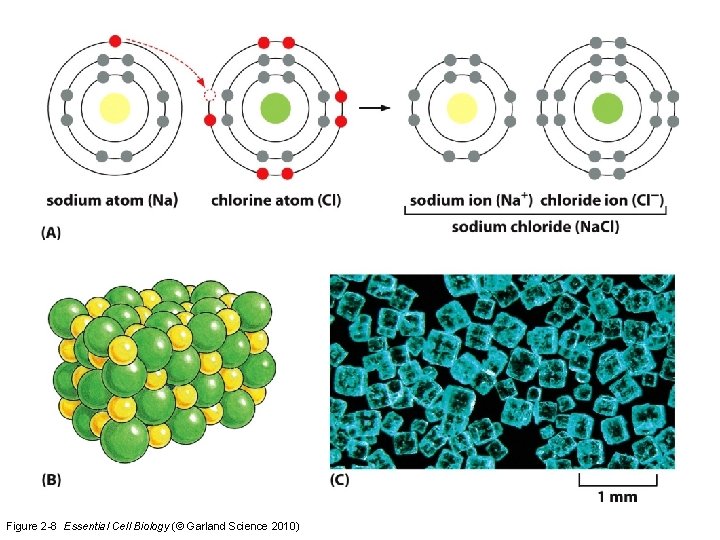
Ions and ionic bonding • Ions are critical in setting up membrane potential in cells – Na+, K+, Cl- • Electrolytes – Gatorade, Vit H 20 • Salts – Na. Cl • Halogen-containing – Electricity

Figure 2 -8 Essential Cell Biology (© Garland Science 2010)

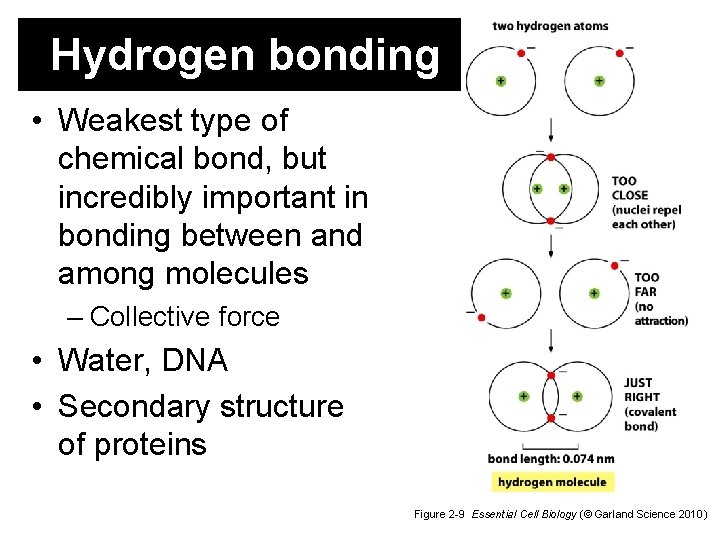
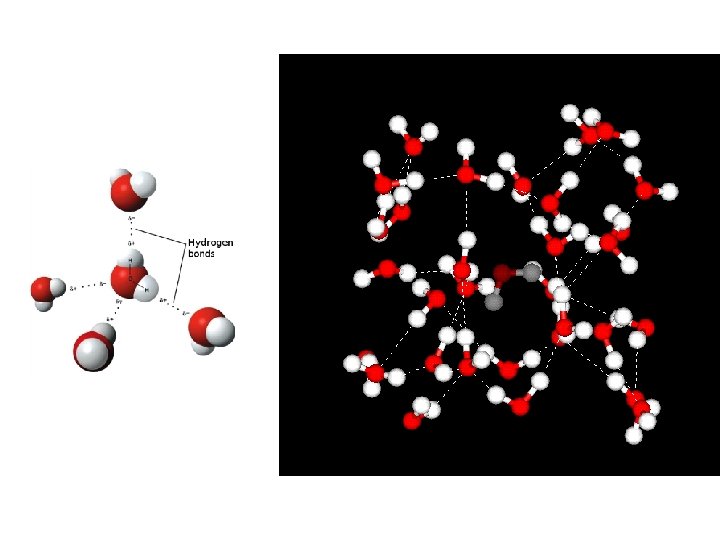
Hydrogen bonding • Weakest type of chemical bond, but incredibly important in bonding between and among molecules – Collective force • Water, DNA • Secondary structure of proteins Figure 2 -9 Essential Cell Biology (© Garland Science 2010)




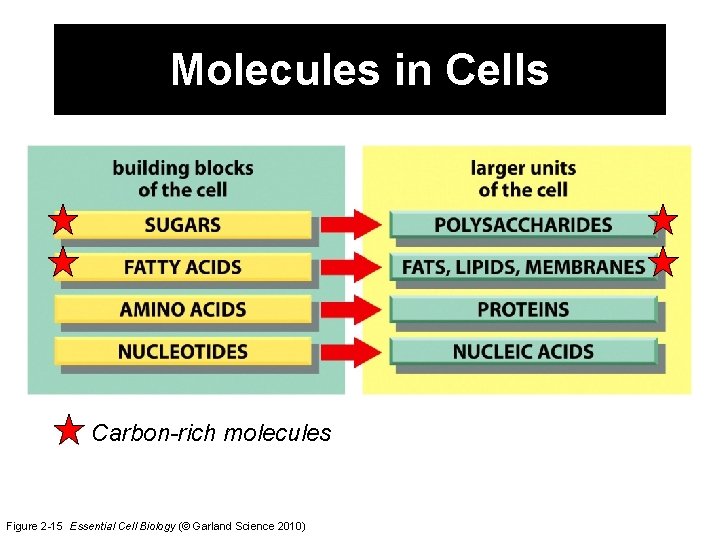
Molecules in Cells Carbon-rich molecules Figure 2 -15 Essential Cell Biology (© Garland Science 2010)

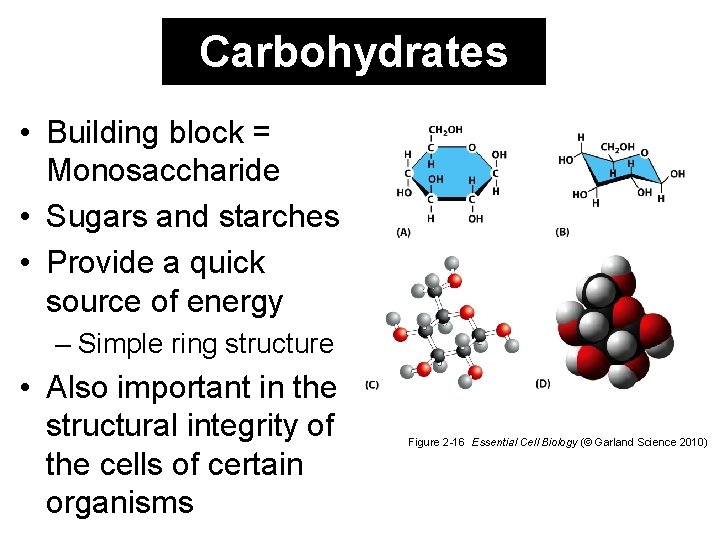
Carbohydrates • Building block = Monosaccharide • Sugars and starches • Provide a quick source of energy – Simple ring structure • Also important in the structural integrity of the cells of certain organisms Figure 2 -16 Essential Cell Biology (© Garland Science 2010)

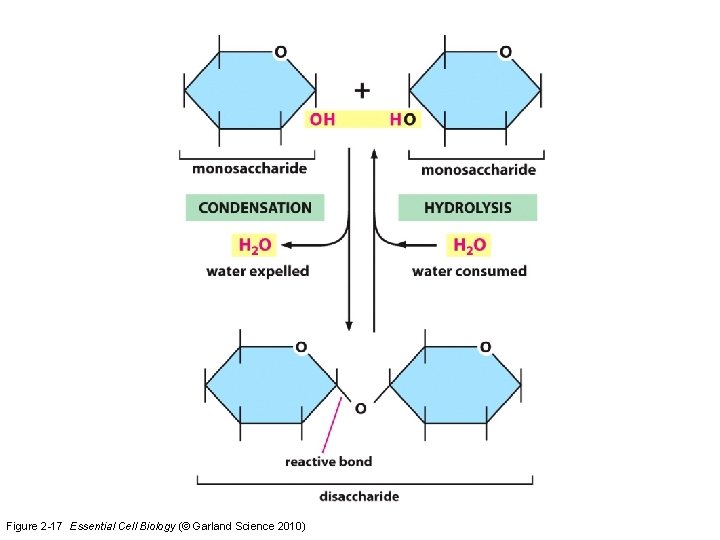
Figure 2 -17 Essential Cell Biology (© Garland Science 2010)

Using bacteria to fight cancer • Clostridium sporogenes • Harmless bacteria found in soil • Anaerobic – Tumor cells = no O 2 • Enzyme + cancer drug = activated drug

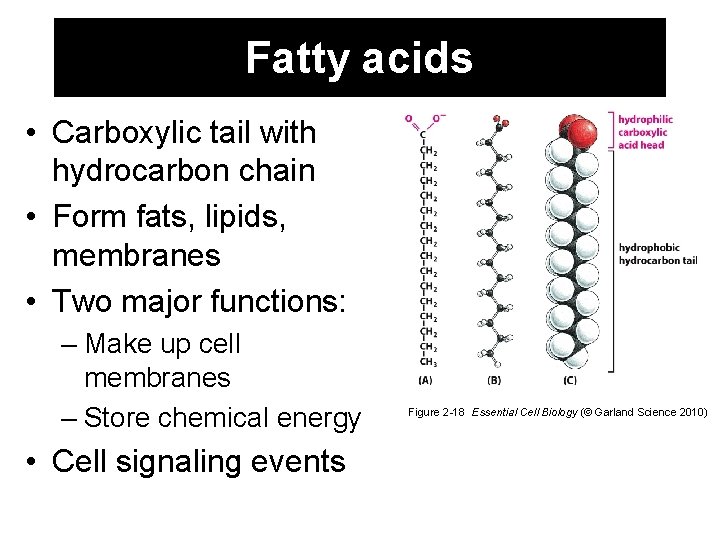
Fatty acids • Carboxylic tail with hydrocarbon chain • Form fats, lipids, membranes • Two major functions: – Make up cell membranes – Store chemical energy • Cell signaling events Figure 2 -18 Essential Cell Biology (© Garland Science 2010)

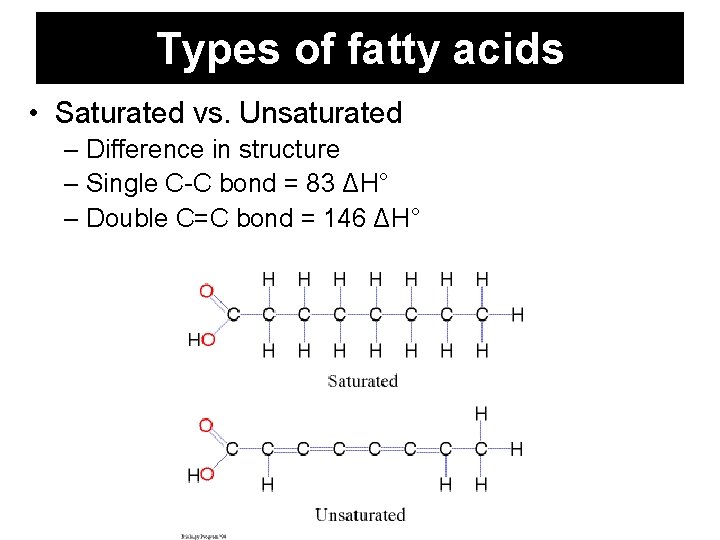
Types of fatty acids • Saturated vs. Unsaturated – Difference in structure – Single C-C bond = 83 ΔH° – Double C=C bond = 146 ΔH°

Saturated vs. Unsaturated • Saturated fats – Animal products – Processed foods – Solid at room temperature • Unsaturated fats – Nuts – Olive oil – Avocados – Liquid at room temperature

Fats in neural development • Axons covered by myelin sheath • Fat cells • Fatty acids are delivered to infants through breast milk

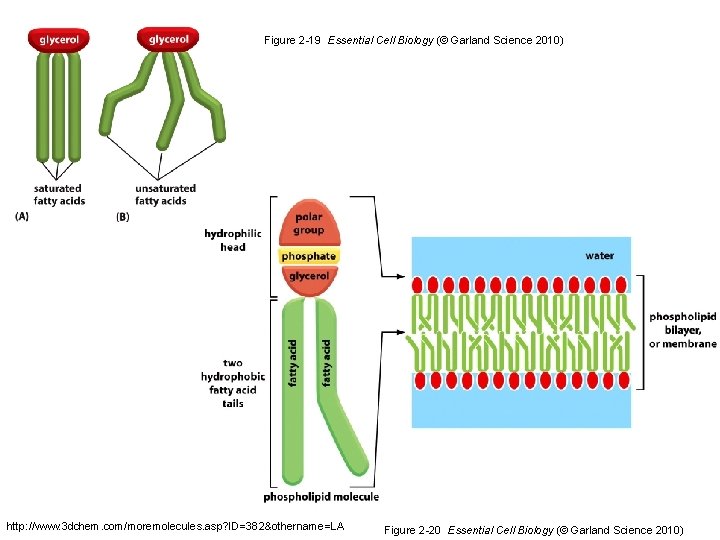
Figure 2 -19 Essential Cell Biology (© Garland Science 2010) http: //www. 3 dchem. com/moremolecules. asp? ID=382&othername=LA Figure 2 -20 Essential Cell Biology (© Garland Science 2010)

Membrane fluidity varies among species • Bigger mammals=less unsaturated fatty acids than smaller mammals – Less membrane fluidity – Lower Na+-K+-ATPase activity • Smaller animals= more unsaturated fatty acids than bigger mammals – More membrane fluidity – Higher Na+-K+-ATPase activity – Increased permeability of the membrane?

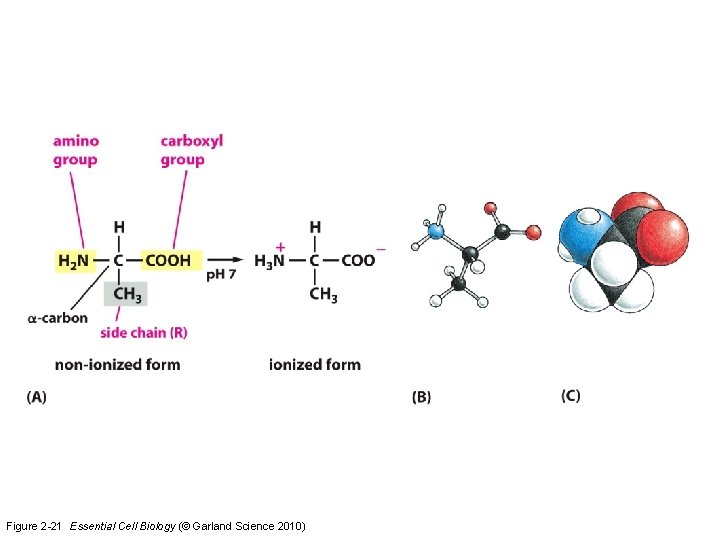
Amino acids • Link together to form proteins • Defining property: all possess a carboxylic acid group and an amino group – COOH – NH 2 • Proteins are polymers of amino acids

Figure 2 -21 Essential Cell Biology (© Garland Science 2010)

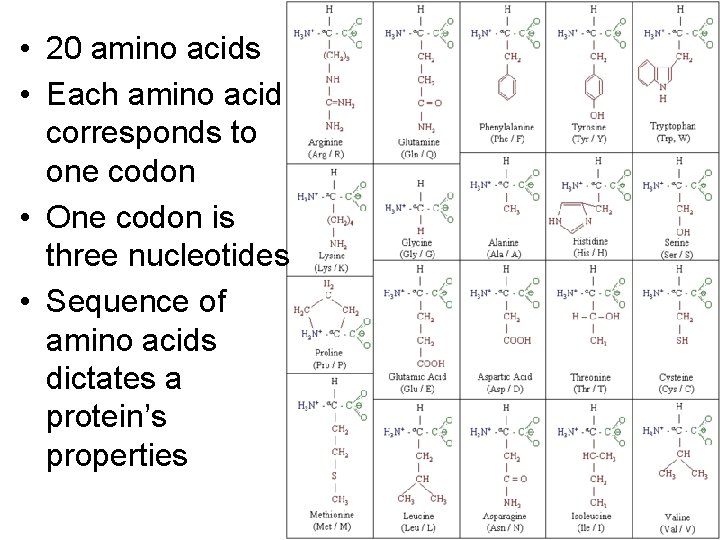
• 20 amino acids • Each amino acid corresponds to one codon • One codon is three nucleotides • Sequence of amino acids dictates a protein’s properties

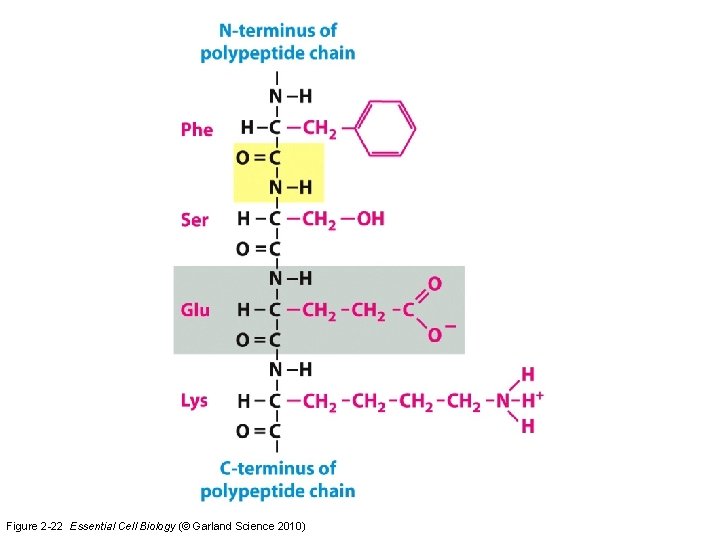
Figure 2 -22 Essential Cell Biology (© Garland Science 2010)

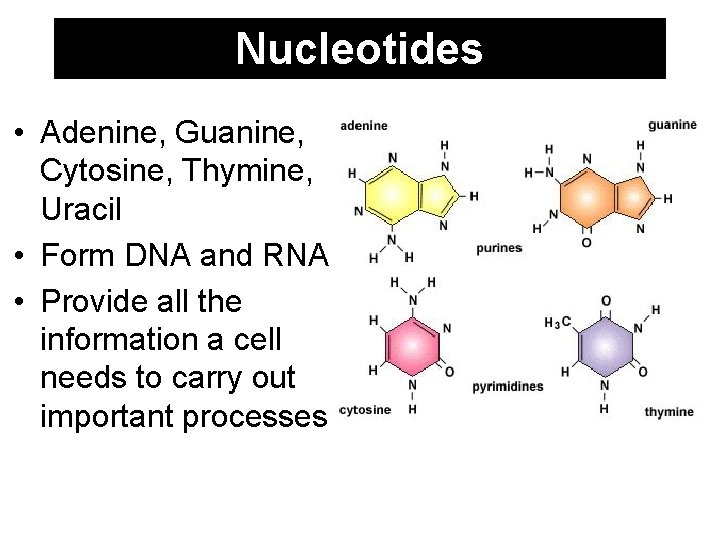
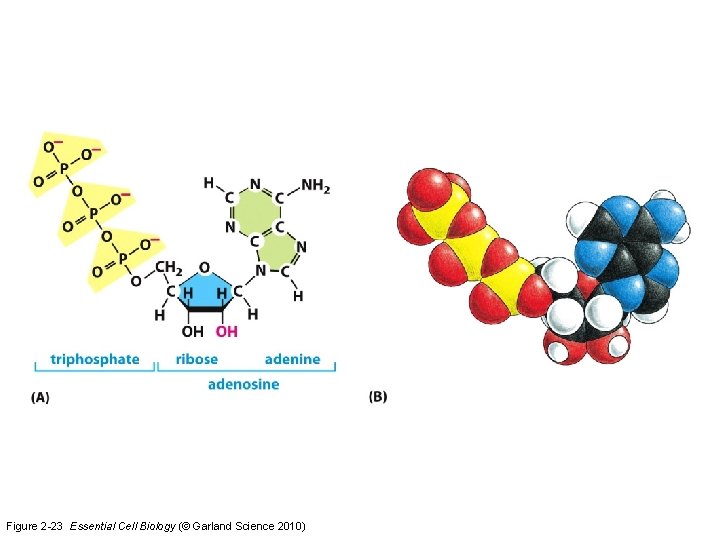
Nucleotides • Adenine, Guanine, Cytosine, Thymine, Uracil • Form DNA and RNA • Provide all the information a cell needs to carry out important processes

Figure 2 -23 Essential Cell Biology (© Garland Science 2010)

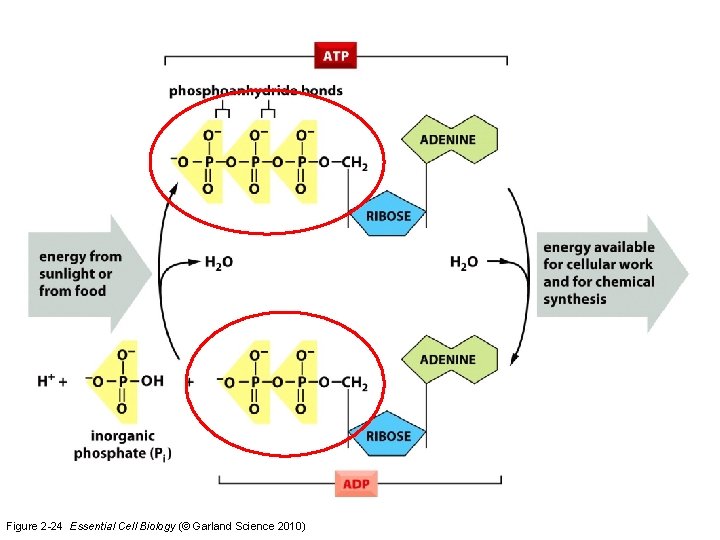
Figure 2 -24 Essential Cell Biology (© Garland Science 2010)

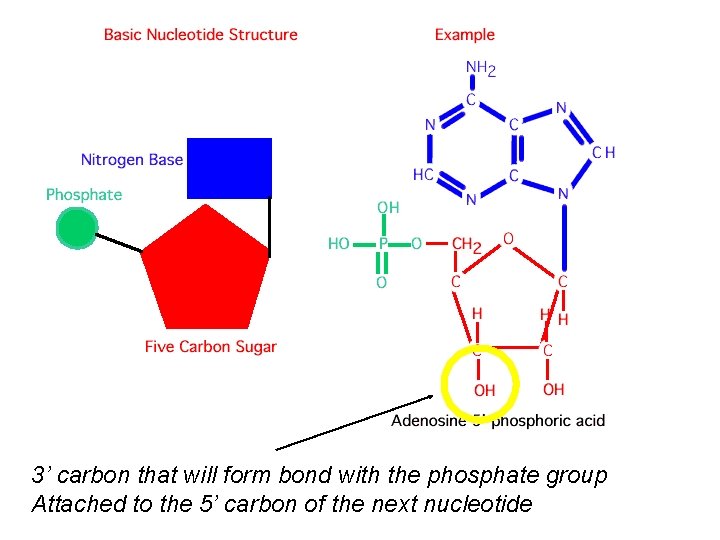
3’ carbon that will form bond with the phosphate group Attached to the 5’ carbon of the next nucleotide

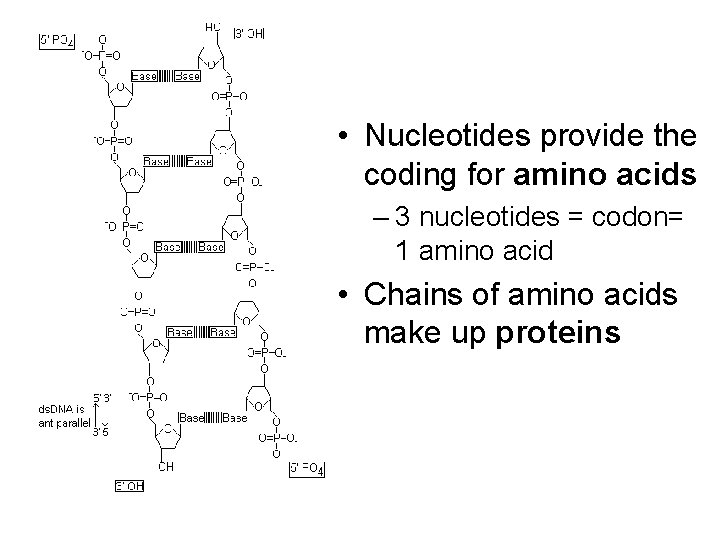
• Nucleotides provide the coding for amino acids – 3 nucleotides = codon= 1 amino acid • Chains of amino acids make up proteins

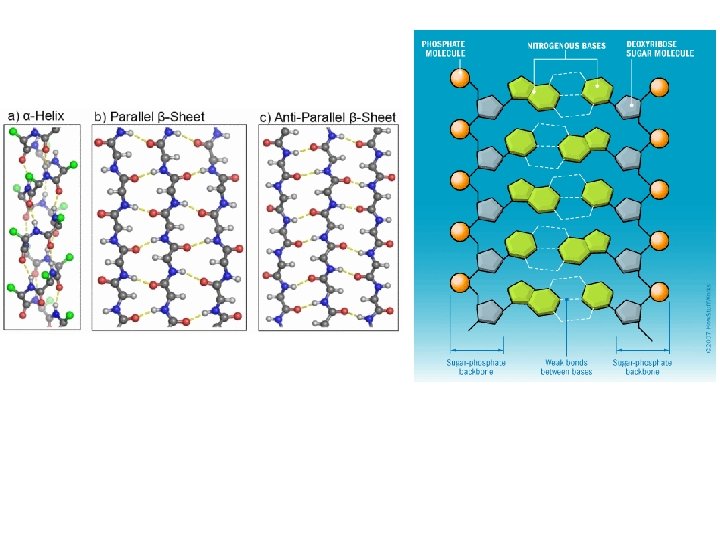
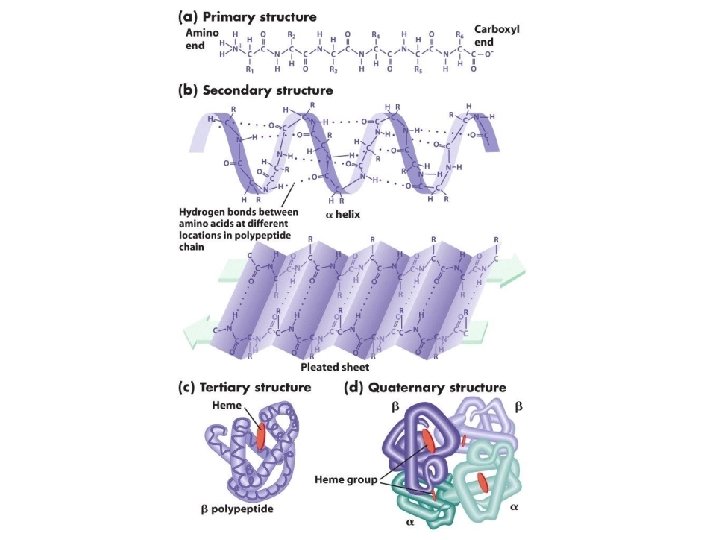
Proteins • Sequence of nucleotides • Levels of protein organization – Primary: sequence – Secondary: alpha helix and beta sheets – Tertiary: final specific geometry that a protein assumes – Quaternary: clustering of several protein chains into a final specific structure


Figure 2 -32 Essential Cell Biology (© Garland Science 2010)

Diseases attributable to defects in protein function • Sickle Cell Anemia • Huntington’s disease • Achondroplasia

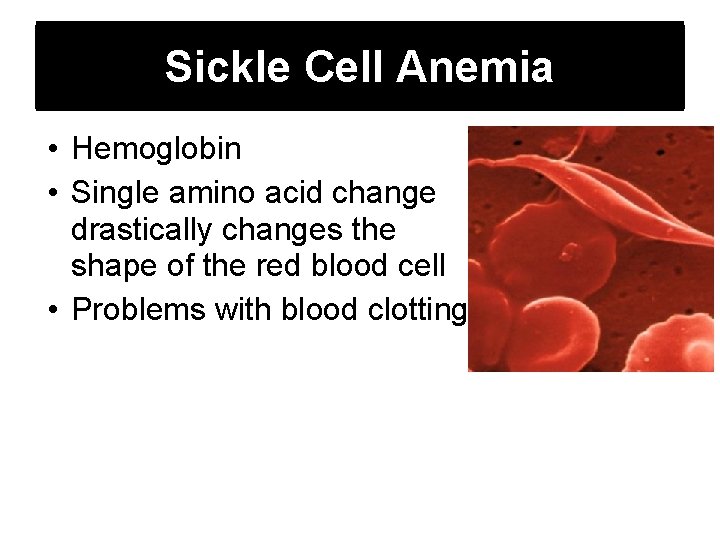
Sickle Cell Anemia • Hemoglobin • Single amino acid change drastically changes the shape of the red blood cell • Problems with blood clotting

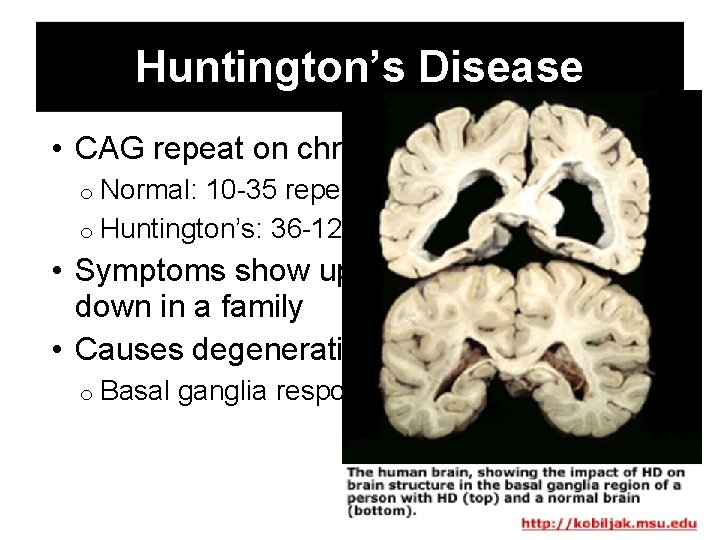
Huntington’s Disease • CAG repeat on chromosome 4 o Normal: 10 -35 repeats o Huntington’s: 36 -120 • Symptoms show up earlier as it is passed down in a family • Causes degeneration of nervous tissue o Basal ganglia responsible for movement

Achondroplasia • FGFR 3 (Fibroblast growth factor gene) o Receptor that regulates bone growth by limiting the formation of bone from cartilage in the long bones o Hyperactive • Chromosome 4

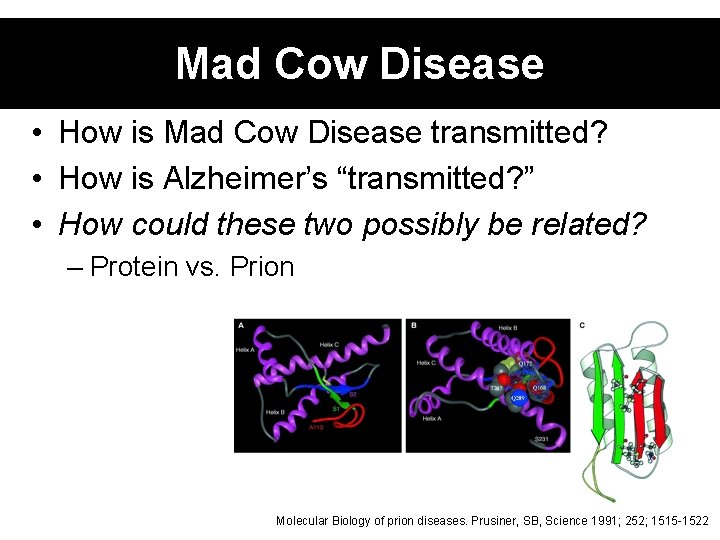
Mad Cow Disease • How is Mad Cow Disease transmitted? • How is Alzheimer’s “transmitted? ” • How could these two possibly be related? – Protein vs. Prion Molecular Biology of prion diseases. Prusiner, SB, Science 1991; 252; 1515 -1522

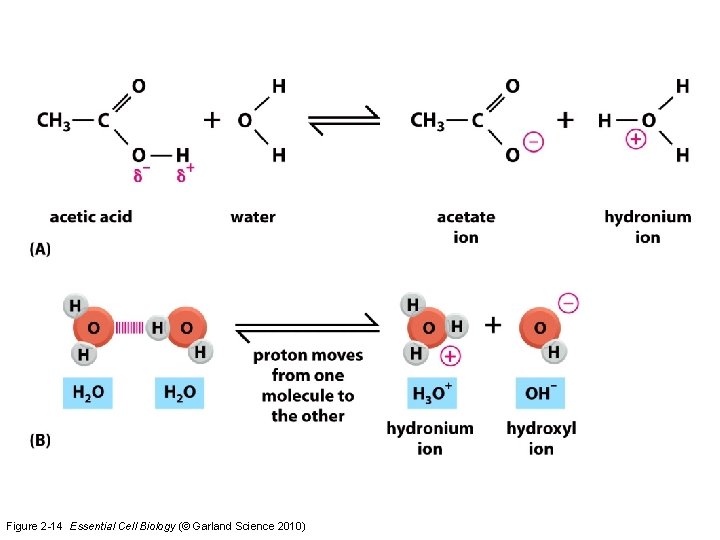
Acids and Bases • p. H = concentration of hydrogen (hydronium ion-H 3 O+) – 0 -14 – 7 = neutral • Acid = donates a hydrogen • Base = accepts a hydrogen or donates a hydroxyl group

Figure 2 -14 Essential Cell Biology (© Garland Science 2010)