Chemical Reactions Life depends on chemical reactions Chemical










- Slides: 13

Chemical Reactions Life depends on chemical reactions

Chemical Reactions �Break bonds and form bonds �Change substances into different substances - reactants are substances changed - products are substances made 6 O₂ + C₆H₁₂O₆ → 6 CO₂ + 6 H₂O Process? Reactants? Products?

Chemical Reactions 6 CO₂ + 6 H₂O → C₆H₁₂O₆ + 6 O₂ Process? Reactants? Products? This reaction proceeds one way… some reactions can proceed in both directions

Chemical Reactions CO₂ + H₂O ↔ H₂CO₃ �This reaction proceeds in both directions: �For example: - When CO₂ is high around cells, CO₂ reacts with H₂O in blood to form carbonic acid - H₂CO₃ - In your lungs when CO₂ is low the reaction goes the other direction and carbonic acid breaks down �These reactions maintain balance or…

Chemical Reactions �Equilibrium - equal rate in both directions - the reactant and product concentrations stay the same � All chemical reactions involve changes in energy - Energy is added to reactants to break bonds - When new bonds are formed energy is released

Chemical Reactions 6 O₂ + C₆H₁₂O₆ → 6 CO₂ + 6 H₂O �How do we know energy is released during chemical reactions in our body? - Body temperature �How does our body maintain homeostasis to keep from overheating? - High specific heat of water �How do chemical reactions get started?

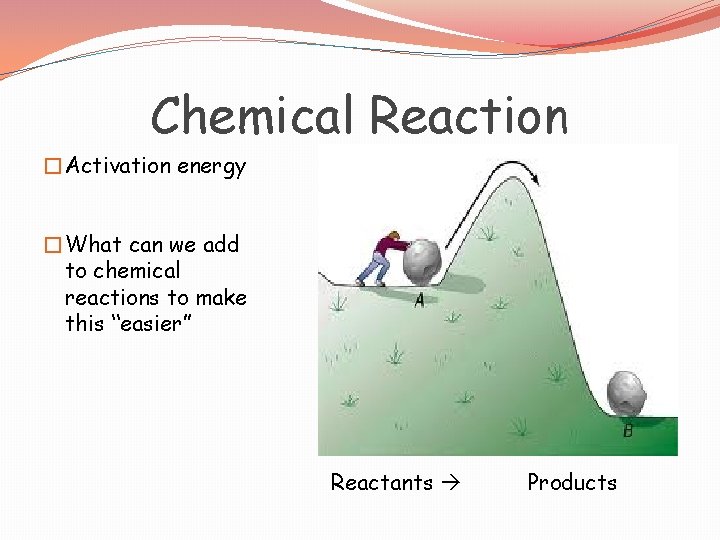
Chemical Reactions � Energy is added to reactants to break bonds �Energy is released when new bonds are formed � Activation energy is the amount of energy that must be absorbed by the reactants for a chemical reaction to start

Chemical Reaction �Activation energy �What can we add to chemical reactions to make this “easier” Reactants Products

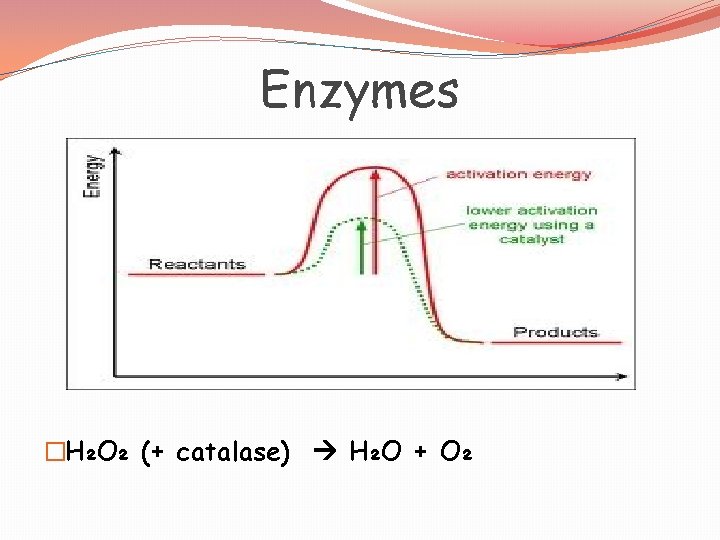
Enzymes �Catalysts: - decrease activation energy - increase reaction rates - in living things are called enzymes

Enzymes �Almost all enzymes are proteins �Lower activation energy �Increase rate of chemical reactions �Are effected by p. H and temperature �Break down or build substances

Enzymes �H₂O₂ (+ catalase) H₂O + O₂

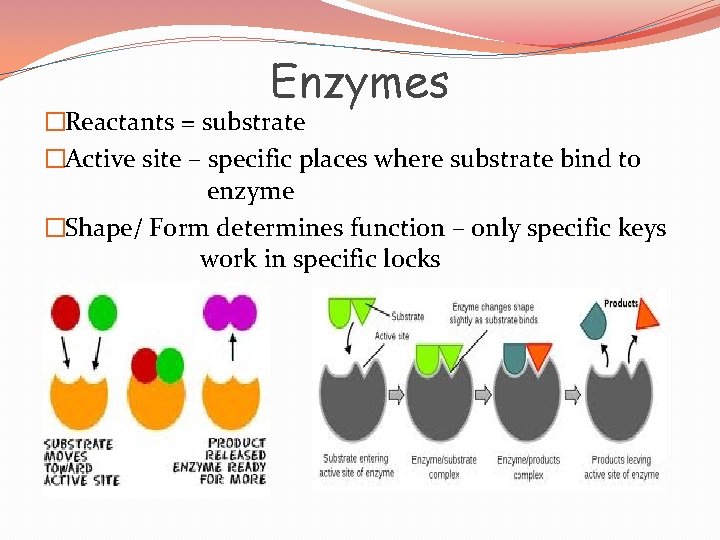
Enzymes �Reactants = substrate �Active site – specific places where substrate bind to enzyme �Shape/ Form determines function – only specific keys work in specific locks

Enzymes �Enzyme video �Key words: - Enzyme - Activation energy - Substrate (reactants) - Active site - Products