Thermal Energy Thermal Energy How does thermal energy



















































































- Slides: 84

Thermal Energy

Thermal Energy How does thermal energy work?

Important terms to know: o Temperature:

Important terms to know: o Temperature: A measure of the average kinetic energy of the individual particles in matter.

Important terms to know: o Temperature: A measure of the average kinetic energy of the individual particles in matter. n Low temperatures = low kinetic energy

Important terms to know: o Temperature: A measure of the average kinetic energy of the individual particles in matter. Low temperatures = low kinetic energy n High temperatures = high kinetic energy n

Important terms to know: o Temperature: continued n Thermometers: As the liquid in thermometer heats up its volume increases (rises) and as it cools off its volume goes down (drops).

Important terms to know: o Temperature: continued n Scales: The three common scales are Fahrenheit, Celsius, and the Kelvin scales.

Important terms to know: o Temperature: continued n Scales: The three common scales are Fahrenheit, Celsius, and the Kelvin scales. p Fahrenheit: Used in the United States.

Important terms to know: o Temperature: continued n Scales: The three common scales are Fahrenheit, Celsius, and the Kelvin scales. p Fahrenheit: Used in the United States. p Celsius: Used nearly everywhere else.

Important terms to know: o Temperature: continued n Scales: The three common scales are Fahrenheit, Celsius, and the Kelvin scales. p Fahrenheit: Used in the United States. p Celsius: Used nearly everywhere else. p Kelvin: Commonly used in the physical sciences.

Important terms to know: o Temperature: continued n Conversions:

Important terms to know: o Temperature: continued Conversions: n °K = °C + 273 n

Important terms to know: o Temperature: continued Conversions: n °K = °C + 273 n °C = °K - 273 n

Important terms to know: o Temperature: continued Conversions: n °K = °C + 273 n °C = °K - 273 n °C = 5/9(°F – 32) n

Important terms to know: o Temperature: continued Conversions: n °K = °C + 273 n °C = °K - 273 n °C = 5/9(°F – 32) n °F = 9/5°C + 32 n

Thermal Energy: o Thermal Energy depends on:

Thermal Energy: o Thermal Energy depends on: n the number of particles an object has.

Thermal Energy: o Thermal Energy depends on: n n the number of particles an object has. the temperature of an object.

Thermal Energy: o Thermal Energy depends on: n n n the number of particles an object has. the temperature of an object. the arrangement of the object’s particles.

Thermal Energy: o Thermal Energy depends on: n n n the number of particles an object has. the temperature of an object. the arrangement of the object’s particles. o Heat:

Thermal Energy: o Thermal Energy depends on: n n n the number of particles an object has. the temperature of an object. the arrangement of the object’s particles. o Heat: n Thermal energy that moves from a warmer object to a cooler object.

Thermal Energy: o Thermal Energy depends on: n n n the number of particles an object has. the temperature of an object. the arrangement of the object’s particles. o Heat: n n Thermal energy that moves from a warmer object to a cooler object. Thermal energy only becomes heat when it is transferred.

Thermal Energy: o Thermal Energy depends on: n the number of particles an object has. n the temperature of an object. n the arrangement of the object’s particles. o Heat: n Thermal energy that moves from a warmer object to a cooler object. n Thermal energy only becomes heat when it is transferred. n It’s unit of measure is “Joules”.


Thermal Energy: o Specific Heat:

Thermal Energy: o Specific Heat: The amount of energy required to raise 1 g of a material by 1 °C.

Thermal Energy: o Specific Heat: The amount of energy required to raise 1 g of a material by 1 °C. o Measured in joules per kilogram-Celsius

Thermal Energy: o Specific Heat: The amount of energy required to raise 1 g of a material by 1 °C. o Measured in joules per kilogram-Kelvin n J/(kg*C)

Thermal Energy: o Common specific heats: n n n n Aluminum: Copper Glass Ice Iron Sand Water 903 J/(kg*C) 385 837 2, 060 450 800 4, 180

Heat Transfer: o Heat is transferred by three different methods.

Heat Transfer: o Heat is transferred by three different methods. n Conduction: The transfer of heat without the movement of matter.

Heat Transfer: o Heat is transferred by three different methods. n Conduction: The transfer of heat without the movement of matter. p. A metal spoon in a pan of hot water (the spoon gets hot over time).

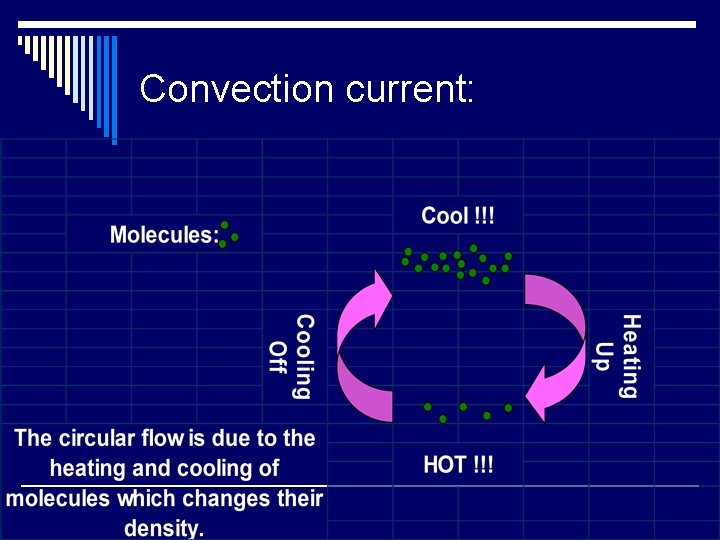
Heat Transfer: o Heat is transferred by three different methods. n Convection: The transfer of heat by the movement of currents within a fluid.

Heat Transfer: o Heat is transferred by three different methods. n Convection: The transfer of heat by the movement of currents within a fluid. p Water heating up in a pot on the stove (you can see the water moving).

Heat Transfer: o Heat is transferred by three different methods. n Convection: The transfer of heat by the movement of currents within a fluid. p Water heating up in a pot on the stove (you can see the water moving). p Convection currents? !? !

Convection current:

Heat Transfer: o Heat is transferred by three different methods. n Radiation: The transfer of energy by electromagnetic waves.

Heat Transfer: o Heat is transferred by three different methods. n Radiation: The transfer of energy by electromagnetic waves. p. A fireplace warming a room.

Heat Transfer: o Heat is transferred by three different methods. n Radiation: The transfer of energy by electromagnetic waves. p. A fireplace warming a room. p Does not require matter to transfer thermal energy (the sun).

Heat Transfer: o Heat flow:

Heat Transfer: o Heat flow: n Heat transfer goes in one direction.

Heat Transfer: o Heat flow: n Heat transfer goes in one direction. p Heat leaves the warmer object and goes to the cooler object.

Heat Transfer: o Heat flow: n Heat transfer goes in one direction. p Heat leaves the warmer object and goes to the cooler object. p This will continue until the two objects have the same temperature.



Heat Transfer: o Conductors:

Heat Transfer: o Conductors: A material that transfers thermal energy well, partially because its atoms or molecules are close together.

Heat Transfer: o Conductors: A material that transfers thermal energy well, partially because its atoms or molecules are close together. n Silver n Stainless steel n tile

Heat Transfer: o Conductors: A material that transfers thermal energy well, partially because its atoms or molecules are close together. n Silver n Stainless steel n tile o Insulators:

Heat Transfer: o Conductors: A material that transfers thermal energy well, partially because its atoms or molecules are close together. n Silver n Stainless steel n tile o Insulators: A material that does not transfer thermal energy very well, partially because its atoms or molecules are not closely packed together.

Heat Transfer: o Conductors: A material that transfers thermal energy well, partially because its atoms or molecules are close together. n Silver n Stainless steel n tile o Insulators: A material that does not transfer thermal energy very well, partially because its atoms or molecules are not closely packed together. n n Wood Wool Paper Atmospheric gases

Thermal Expansion: Does matter with thermal energy?

Matter: o What is matter?

Matter: o What is matter? n Has mass

Matter: o What is matter? n Has mass n Takes up space (has volume)

Matter: o The three states of matter:

Matter: o The three states of matter: n Solids p Has a fixed shape p Has a fixed volume

Matter: o The three states of matter: n Solids p Has a fixed shape p Has a fixed volume n Liquids p Has a shape that can change p Has a fixed volume

Matter: o The three states of matter: n Solids p Has a fixed shape p Has a fixed volume n Liquids p Has a shape that can change p Has a fixed volume n Gases p Has a shape that can change. p Has a volume that can change.

Matter:

Matter: o The fourth state of matter is plasma.

Matter: o The fourth state of matter is plasma. n Plasma is the highest energy state of matter (moving super fast).

Matter: o The fourth state of matter is plasma. Plasma is the highest energy state of matter (moving super fast). n Very, very, very hot!!! n

Matter: o The fourth state of matter is plasma. Plasma is the highest energy state of matter (moving super fast). n Very, very, very hot!!! n Found in places like our sun. n

Matter: o The fourth state of matter is plasma. Plasma is the highest energy state of matter (moving super fast). n Very, very, very hot!!! n Found in places like our sun. n Not always considered a state of matter. n

Matter: o Changes in states of matter:

Matter: o Changes in states of matter: n Matter can change from any state to any other state by adding energy.

Matter: o Changes in states of matter: Matter can change from any state to any other state by adding energy. n The temperature does not change until the change of state is complete. n

Thermal Expansion: o When an object is heated it usually responds to the heat.

Thermal Expansion: o When an object is heated it usually responds to the heat. n One response is for the particles of the object to move faster and spread out (increasing volume).

Thermal Expansion: o When an object is heated it usually responds to the heat. One response is for the particles of the object to move faster and spread out (increasing volume). n This causes the object to expand is called thermal expansion. n

Thermal Expansion examples: o When thermometers are exposed to warmer temperatures.

Thermal Expansion examples: o When thermometers are exposed to warmer temperatures. o Thermostats in your home.


Thermal Expansion examples: o When thermometers are exposed to warmer temperatures. o Thermostats in your home. o Sidewalks


Thermal Expansion examples: o When thermometers are exposed to warmer temperatures. o Thermostats in your home. o Sidewalks o Bridges


Thermal Expansion examples: o When thermometers are exposed to warmer temperatures. o Thermostats in your home. o Sidewalks o Bridges o Jars (pickles, jelly, etc…)

Thermal Expansion: o Contraction:

Thermal Expansion: o Contraction: 1. The heat source gets removed.

Thermal Expansion: o Contraction: 1. 2. The heat source gets removed. The particles move less and take up less space.

Thermal Expansion: o Contraction: 1. 2. 3. The heat source gets removed. The particles move less and take up less space. The size of the object decreases.