Review of Heat Temperature Thermal Energy Heat flow




















- Slides: 22

Review of : Heat, Temperature & Thermal Energy

Heat flow of energy from objects is a _______ higher at a _____ temperature to objects at a _____ temperature. lower �Heat �Soon after you put ice cubes into a pitcher of lemonade, energy is transferred from lemonade to the colder ____. the warmer _____ ice HEAT FLOWS FROM WARMER COOLER!!!

Temperature �Temperature is a measure of the average kinetic energy of all the _____ particles in an object. �If a liquid, such as hot cocoa, has a high temperature, the particles in the liquid are Very fast high moving _____, and have a _______ average kinetic energy. �If a drink, such as a fruit smoothie, has a low temperature, the particles in the liquid More slowly and have a are moving _____ low _______ kinetic energy

Thermal Energy total kinetic energy is the _______ energy of particles in an object. �Note that temperature and thermal energy are different from each other. average while Temperature is an _____ thermal energy is a ______. total �A glass of water may have the same temperature as the Mississippi River, but less thermal energy because will have _____ there are less water particles in the glass of water. �Thermal

Review of Conduction, Convection, and Radiation

Conduction �Conduction is the process that moves energy from one object to another when physically they are Touching ________. �Conduction occurs any time that objects different temperature come into at a _____ contact with each other. �Conduction can also occur within a single object. In this case, energy is transferred warmer part of the object to the from the ______ cooler part of the object. _______

Convection �Convection is the process that transfers energy by the movement of particles in liquid the same direction in a _____ or gas ____. more dense will �Cool air/water that is _____ less dense, air/water push warmer, _____ upwards. Once that warm air/water more dense once again cools, it becomes _____ and settles back down to the bottom to start the process all over again.

Radiation �Radiation is energy that travels as electromagnetic waves, which includes _______ visible light and microwaves. Sun is the most significant source of �The ____ radiation that you experience on a daily basis. �Like both conduction and convection, radiation can transfer energy from a ______ warmer object to a _______ one. cooler differs from conduction �However, radiation and convection because radiation can travel through _______, as it does when it Empty space moves from the Sun to the Earth.

Quiz Practice!

A hot saucepan is placed on a cold bench. Heat will flow from the saucepan to the bench until A. B. C. D. the bench is warmer than the saucepan. the bench is as hot as the saucepan was. the saucepan has lost all of its heat. the saucepan and bench are at the same temperature.

A candle is lit. The heat of the flame will melt the candle because A. the heat of the hot flame is transferred to the colder candle. B. the wax of the candle is repelled by the flame. C. the candle must stay hot to heat the flame. D. the flame takes all the heat energy and the candle has no energy.

Which types of heat transfer involve heat flow from hot objects to colder objects? A. convection, conduction, and radiation B. conduction and radiation, but not convection C. convection and conduction, but not radiation D. convection and radiation, but not conduction

Heat will flow from a hot object to a cold object until the objects reach A. B. C. D. Boiling point. stability. collapse. The same temperature.

Ice is placed in hot water. What happens to the temperature of the ice and the water? A. B. C. D. The ice gets warmer, and the water gets cooler. The ice gets warmer, and the water stays the same. The ice and the water both get cooler. The ice gets cooler, and the water gets warmer.

When you hold a cup of warm water, your hands get warmer. Why does this happen? A. B. C. D. Heat transfers by by convection conduction condensation radiation

The total energy of all particles in a substance is called – Temperature B. Thermal energy C. Degrees D. Mass A.

Heat flows – Only from warmer objects to cooler ones B. Only from cooler objects to warmer ones C. Both ways between objects that are not at the same temperature D. Between all objects all the time regardless of their temperature A.

Heat is transferred from one particle of matter to another through the movement of fluids in a process called – Conduction B. Convection C. Radiation D. Insulation A.

Which kind of heat transfer takes place when you put a spoon in a cup of soup and the spoon gets hot to the touch Convection B. Conduction C. Deduction D. Radiation A.

Which kind of heat transfer can occur through space Radiation B. Conduction C. Convection D. Deduction A.

Which kind of heat transfer occurs when you add hot water to a cold bath? Radiation B. Conduction C. Deduction D. Convection A.

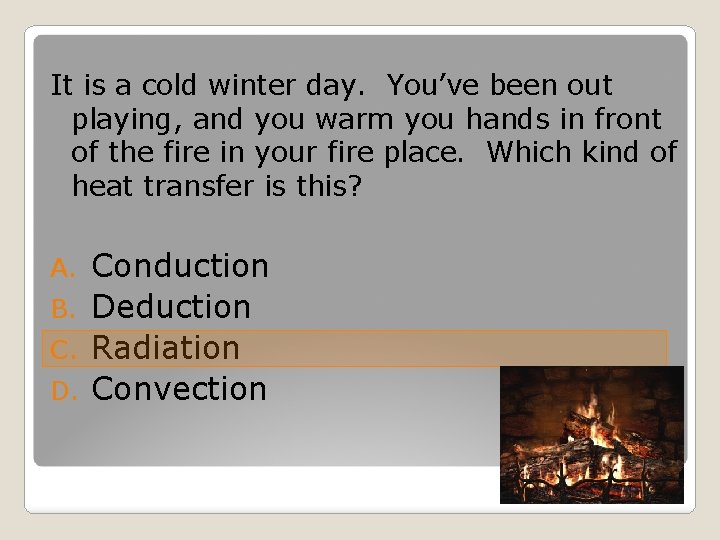
It is a cold winter day. You’ve been out playing, and you warm you hands in front of the fire in your fire place. Which kind of heat transfer is this? Conduction B. Deduction C. Radiation D. Convection A.