THERMAL ENERGY Thermal Energy is heat energy it












- Slides: 17

THERMAL ENERGY Thermal Energy is heat energy; it is the total kinetic and potential energy of the particles making up the material. Integrated Science I

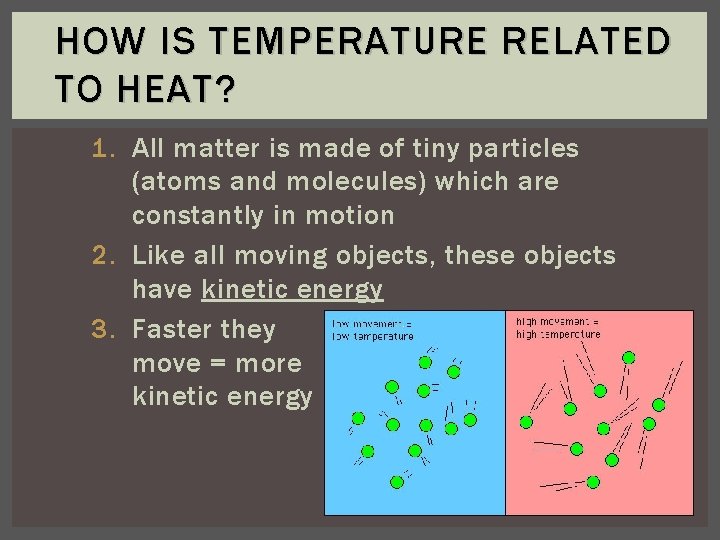
HOW IS TEMPERATURE RELATED TO HEAT? 1. All matter is made of tiny particles (atoms and molecules) which are constantly in motion 2. Like all moving objects, these objects have kinetic energy 3. Faster they move = more kinetic energy

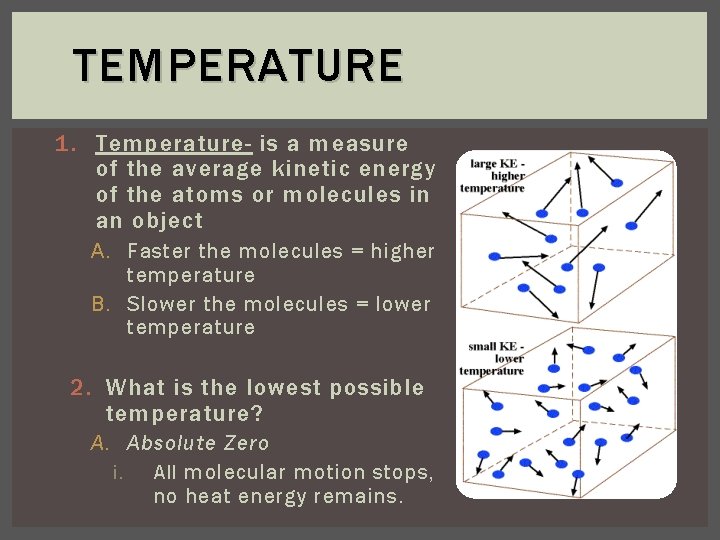
TEMPERATURE 1. Temperature- is a measure of the average kinetic energy of the atoms or molecules in an object A. Faster the molecules = higher temperature B. Slower the molecules = lower temperature 2. What is the lowest possible temperature? A. Absolute Zero i. All molecular motion stops, no heat energy remains.

HEAT 1. Heat- thermal energy that flows from something at a higher temperature to something at a lower temperature. A. Measured in Joules (J) 2. Heat always flows from hot Hot Cocoa is warmer than the room air. Heat from the cocoa flows from the cocoa to the room and cools off the cocoa. cold Room air is warmer than the iced tea. Heat flows from room to ice cubes and warms up the iced tea.

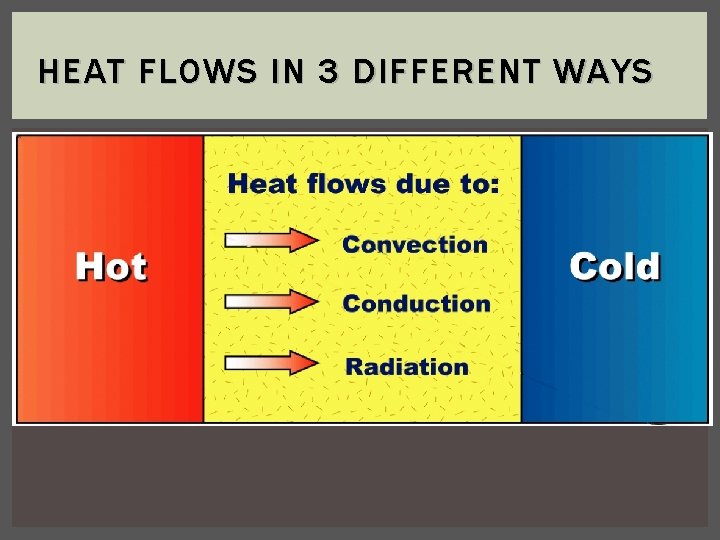
HEAT FLOWS IN 3 DIFFERENT WAYS

HEAT FLOWS IN 3 DIFFERENT WAYS 1. Conduction - transfer of heat energy by direct contact in solids 2. Convection - transfer of heat through fluids with different densities a. Hot, less dense fluids rise, then cool, more dense fluids sink 3. Radiation - transfer of heat energy by waves in gases or a vacuum a. Light color materials = reflect more radiant energy b. Dark color materials = absorb more radiant energy

THERMAL CONDUCTIVITY 1. Thermal conductivity depends on the rate at which thermal energy is transferred from one end of a material to another. 2. Conductors have a high rate of thermal energy transfer and insulators have a slow rate of thermal energy transfer. 3. The rate at which thermal radiation is absorbed or given off by a system depends on its temperature, color, texture and exposed surface area.

FACTORS AFFECTING HEAT ENERGY TRANSFER 1. Temperature § The greater the temperature difference, the faster the rate of heat transfer 2. Type of Material § Heat is transferred through solids better than through gases 3. Surface A rea § Larger surface area transfers more energy than smaller surface areas

FACTORS AFFECTING HEAT ENERGY TRANSFER 4. Distance that Heat must be Conducted § Thick materials hold heat better than thin materials 5. Type of surface § Rough surfaces absorb more heat than smooth surfaces § Smooth surfaces reflect heat 6. Color of Material § Light colors reflect heat § Dark color absorb heat

BRAINSTORM: IS THERE A TIME WHEN HEAT DOES NOT FLOW? YES! § If two objects are at the same temperature, they are in thermal equilibrium and no heat flows from one to the other

SPECIFIC HEAT – THE AMOUNT OF ENERGY TAKEN IN OR GIVEN OUT WHEN THE TEMPERATURE OF A SUBSTANCE CHANGES BY 1 °C. 1. Substances with a high specific heat capacity absorb more energy before they change in temperature than substances with low specific heat capacity. 2. Water has the highest heat capacity of any liquid or solid, except ammonia. a. Because of its high heat capacity, water can absorb a large amount of heat without changing much in temperature b. Often used as a coolant in engines because it absorbs heat with little change in temperature

BRAINSTORM!!! Have you ever noticed that water in a lake or pool feels colder than the air during the day, but warmer than the air during the night? Why do you think this is? Answer: The water has a higher specific heat than the air. Therefore, water warms more slowly in the day and cools more slowly at night than air does.

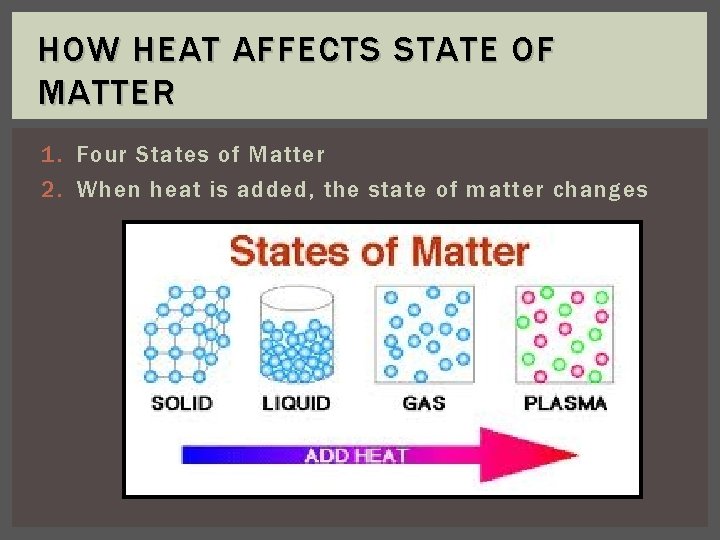
HOW HEAT AFFECTS STATE OF MATTER 1. Four States of Matter 2. When heat is added, the state of matter changes

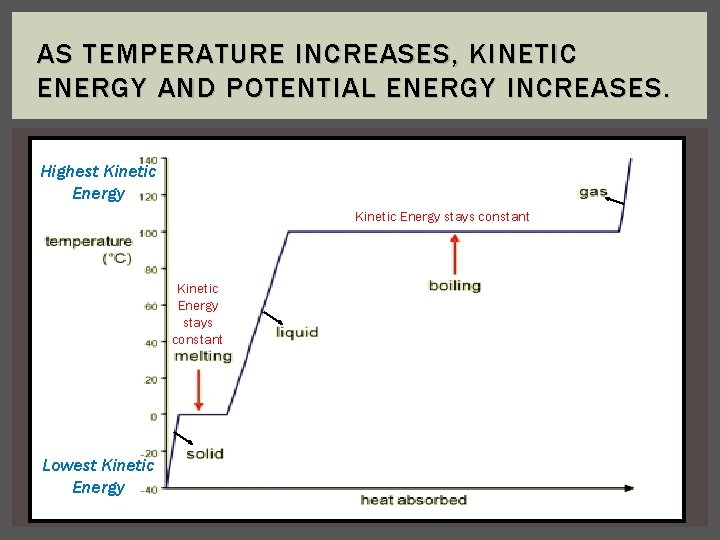
AS TEMPERATURE INCREASES, KINETIC ENERGY AND POTENTIAL ENERGY INCREASES. Highest Kinetic Energy stays constant Lowest Kinetic Energy

READING A PHASE CHANGE GRAPH 1. The temperature does not change on a horizontal (phase change) a. This means there is no change in kinetic energy (moving energy) during a phase change 2. When heating a substance, a phase change will occur when the kinetic energy (moving energy) of the particles is great enough to overcome the attractive forces between the particles; the substance then melts or boils. 3. When cooling a substance, a phase change will occur when the kinetic energy (moving energy) of the particles is no longer great enough to overcome the attractive forces between the particles; the substance then condenses or freezes.

WHAT HAPPENS TO THE VOLUME OF MATERIALS WHEN THEY ARE HEATED? 1. It depends on the material. Most materials will expand the volume will increase because the increased kinetic energy of the particles causes an increased distance between particles.

WHAT HAPPENS TO THE DENSITY OF MATERIALS WHEN THEY ARE HEATED? 1. Solids have greater density than liquids, which have greater density than gases due to the spacing between the particles. The density of a substance can be calculated from the slope of a mass vs. volume graph.