Chapter 6 Thermal energy 6 1 Thermal Energy




























- Slides: 30

Chapter 6 Thermal energy

6. 1 Thermal Energy, Heat and Temperature • Recall: • Kinetic Energy? • Potential Energy? • Thermal Energy?

6. 1 Thermal Energy, Heat and Temperature • Particles in substances are always moving, therefore they always have some amount of kinetic energy. • Faster particles have more kinetic energy. • Slower particles have less kinetic energy.

6. 1 Thermal Energy, Heat and Temperature • Particles also have potential energy, due to the attraction of the particles to one another. • The greater the distance between the particles, the greater the potential energy. • Which state has greatest average potential energy; solid, liquid, or gas? Why? Gas which has greater distance between particles than both liquids and solids.

6. 1 Thermal Energy, Heat and Temperature • Thermal Energy- sum of the kinetic and potential energy of the particles that make up a material. • Describes the energy of solids, liquids and gases

6. 1 Thermal Energy, Heat and Temperature • Temperature is the average kinetic energy of the particles that make up a material. • Particles that are moving faster have more kinetic energy and higher temperature. • Inside a house vs. outside a house.

6. 1 Thermal Energy, Heat and Temperature • Temperature and thermal energy are related but not the same! • As ice melts, there is both water and ice at the same temperature however they do not have the same thermal energy. • Which has more thermal energy? Why? • The average distance between the particles in a liquid is greater than a solid therefore it has more potential and thermal energy.

6. 1 Thermal Energy, Heat and Temperature • Average kinetic energy of particles can not be directly measured so how do we determine temperature? • Thermometers • Temperature is measured using three different units: • 1. Celsius • 2. Fahrenheit • 3. Kelvin

6. 1 Thermal Energy, Heat and Temperature •

6. 1 Thermal Energy, Heat and Temperature • Average room temperature is 37°C. What is this in Fahrenheit? • The average body temperature of a house cat is 101. 5 °F. What is this temperature in Celsius?

6. 1 Thermal Energy, Heat and Temperature •

6. 1 Thermal Energy, Heat and Temperature • The temperature of a boiling pot of water is 373 K, what is the temperature in Celsius? • On a cold winter day a thermometer reads -15°C, what is that temperature in Kelvin?

6. 1 Thermal Energy, Heat and Temperature • A cold glasses of lemonade has a temperature of -32°F, find that temperature in Kelvin. • The average temperature on Earth is about 57°F, what would be the temperature in Kelvin?

6. 1 Thermal Energy, Heat and Temperature • Heat- the movement of thermal energy from a warmer object to a cooler object. • The greater the temperature difference, the more heat transferred. • Heating continues until all objects are the same temperature.

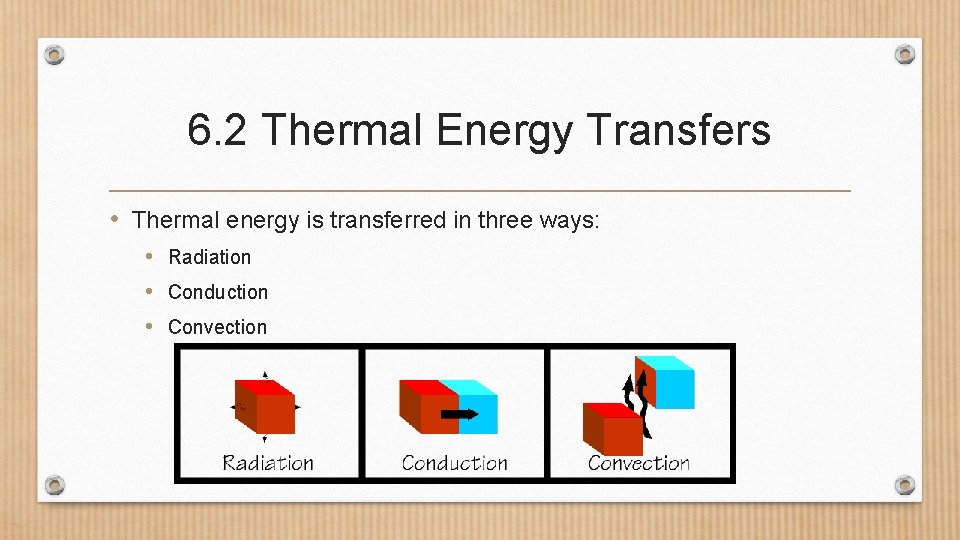
6. 2 Thermal Energy Transfers • Thermal energy is transferred in three ways: • Radiation • Conduction • Convection

6. 2 Thermal Energy Transfers • Radiation- transfer of thermal energy by electromagnetic waves. • All objects transfer some thermal energy by radiation. • Which transfers more thermal energy hot objects or cold objects?

6. 2 Thermal Energy Transfers • Conduction- the transfer of thermal energy by the collisions of particles in direct contact • Particles with higher kinetic energy transfer some to particles with lower kinetic energy. • When kinetic energy is transferred, thermal energy is transferred.

6. 2 Thermal Energy Transfers • Thermal Conductors vs. Thermal Insulators • Conductors allow thermal energy to flow easily • Examples: copper, aluminum • Insulators resists the flow of thermal energy. • Examples: plastic, wood

6. 2 Thermal Energy Transfers • All materials require a certain amount of energy to change it’s temperature. • This is known as a material’s Specific heat. • That is the amount of thermal energy required to increase the temperature of 1 kg of a material 1°C.

6. 2 Thermal Energy Transfers • Higher specific heat, more thermal energy required to increase the temperature. • Which has a higher specific heat a thermal conductor or insulator? Why? Thermal insulator because it resists the flow of thermal energy, requiring more to increase its temperature.

6. 2 Thermal Energy Transfers • Water has a very high specific heat meaning it requires a lot of thermal energy to increase the temperature. • Is that good or bad? • Prevents your body from overheating • Keeps pools and oceans cool in the summer • Good for cooling machinery

6. 2 Thermal Energy Transfers •

6. 2 Thermal Energy Transfers • It takes 487. 5 J to heat 25 grams of copper from 25 °C to 75 °C. What is the specific heat of copper? • A 500 g cube of lead is heated from 50 °C to 100 °C. How much energy was used to heat the lead if the specific heat of lead is 0. 129 J/g°C.

6. 2 Thermal Energy Transfers • A cook places a 150 g potato wrapped in aluminum foil (Cp=0. 902 J/g°C) in the oven and it is heated up with 700 J of energy. What was the change in temperature? • What is the mass of a ball of iron, if it is heated using 250 J of energy to go from 10°C to 100°C and has a specific heat of. 134 J/g°C.

6. 2 Thermal Energy Transfers • What happens when you take a balloon outside on a cold day? • The balloon shrinks in size due to thermal contraction. • Thermal contraction is a decrease in an materials volume when the temperature decreases. https: //www. youtube. com/watch? v=QLTWt 9 r_NF 0

6. 2 Thermal Energy Transfers • When the balloon is heated, the opposite effects occur. • Thermal expansion is when the volume of a material increases as the temperature increases.

6. 2 Thermal Energy Transfers • Convection-the transfer of thermal energy by the movement of particles from one part of a material to another. • Only occurs in fluids. • Example: boiling water

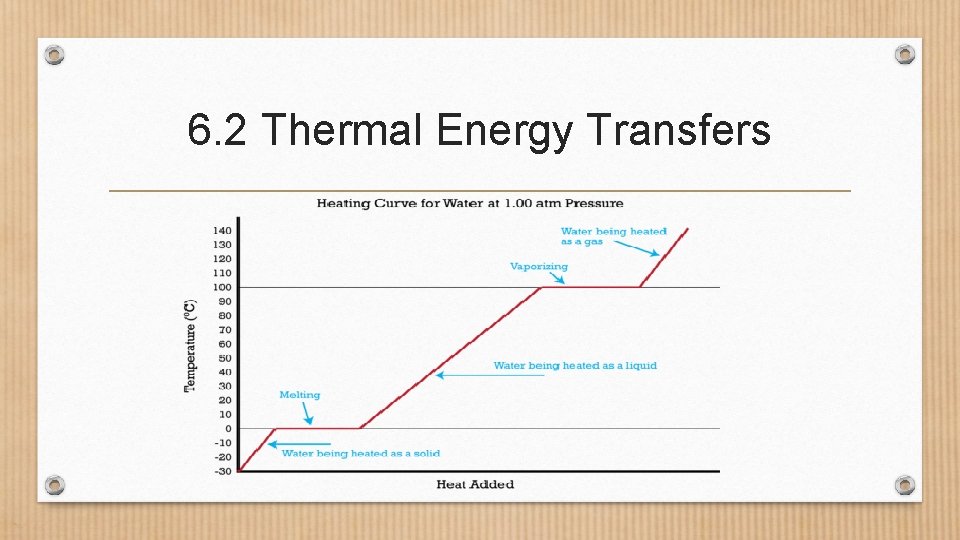
6. 2 Thermal Energy Transfers • Heating Curve- a graph that shows how the temperature of a substance changes as heat is added. • X-axis: Heat • Y-axis: Temperature (°C) • Horizontal lines represent a change in state

6. 2 Thermal Energy Transfers

6. 2 Thermal Energy Transfers • Calorimeter- a instrument used to measure heat.