Thermal Energy What is Thermal Energy The energy


















- Slides: 27

Thermal Energy

What is Thermal Energy? ¡The energy possessed by an object or system due to the movement of particles within the object

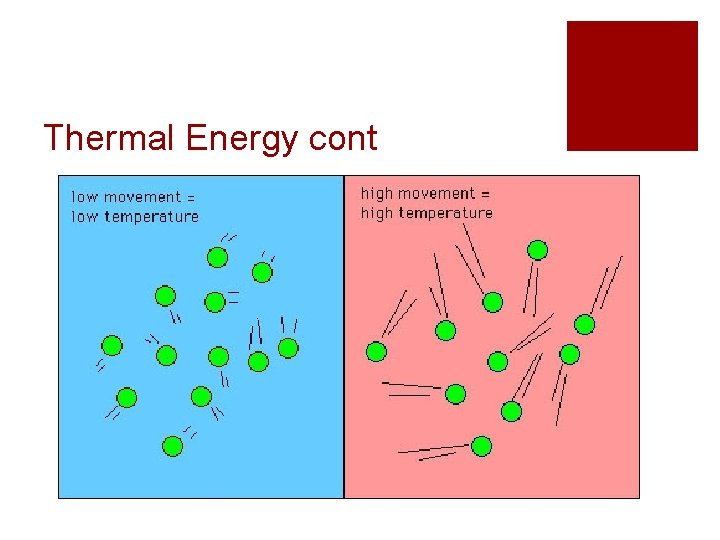
Thermal Energy cont. ¡Thermal energy is the result of kinetic energy ¡The more kinetic energy an object has, the more thermal energy that object has ¡ The faster the molecules in an object are moving, the higher the temperature that object has

Thermal Energy cont

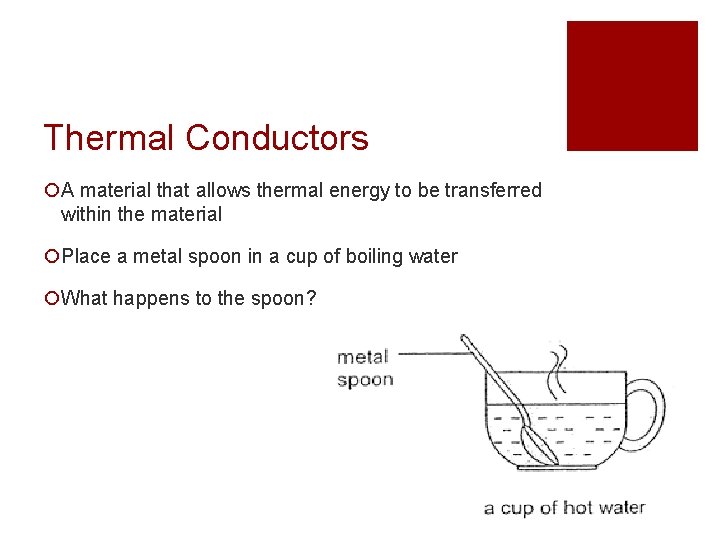
Thermal Conductors ¡A material that allows thermal energy to be transferred within the material ¡Place a metal spoon in a cup of boiling water ¡What happens to the spoon?

Thermal Conductors cont. ¡The spoon will become hot! ¡This is because thermal energy from the boiling water was transferred to the metal spoon

Thermal Insulators ¡A material that prevents thermal energy from being transferred ¡Insulation in housing is made up of thermal insulating material to keep your home warm or cold ¡ Prevents warm air from leaving in the winter ¡ Prevents warm air from entering in the summer

Thermal Insulators cont.

Thermal Insulators cont.

Thermal Expansion ¡When an object’s volume increases as a result of the object’s temperature increasing ¡Example: Hot air balloon ¡ As heat is added, the balloon inflates ¡ When it inflates, the volume increases

Thermal Contraction ¡The decrease in an object’s volume as a result of its temperature being decreased ¡A balloon’s volume will decrease when exposed to colder temperatures

Heat ¡The transfer of thermal energy from one object to another

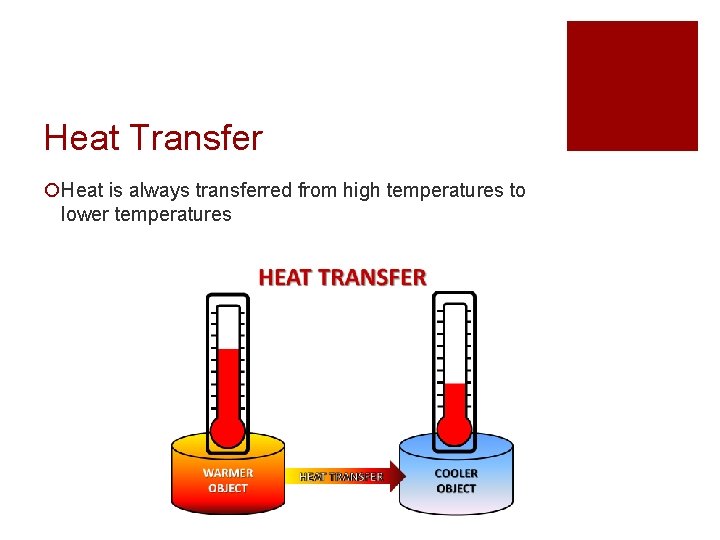
Heat Transfer ¡Heat is always transferred from high temperatures to lower temperatures

Heat transfer ¡Heat can be transferred in three different ways ¡Conduction ¡Convection ¡Radiation

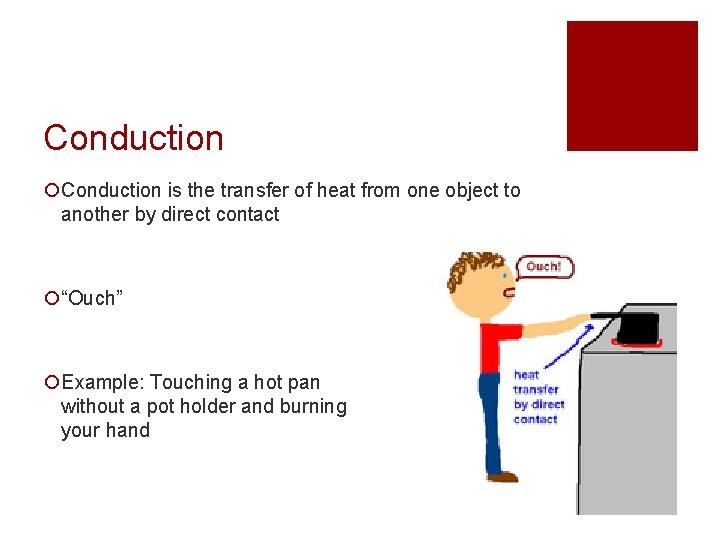
Conduction ¡Conduction is the transfer of heat from one object to another by direct contact ¡“Ouch” ¡Example: Touching a hot pan without a pot holder and burning your hand

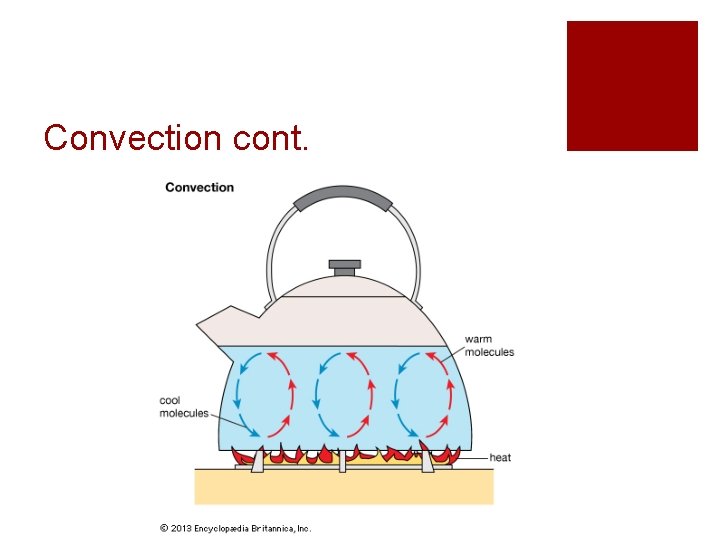
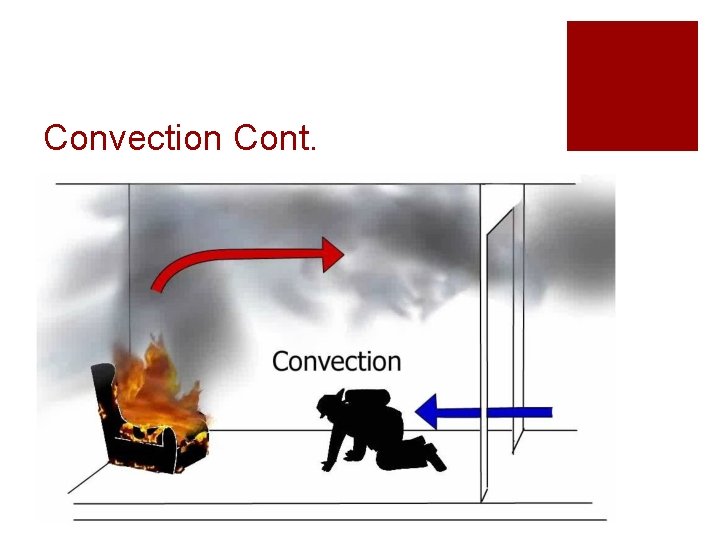
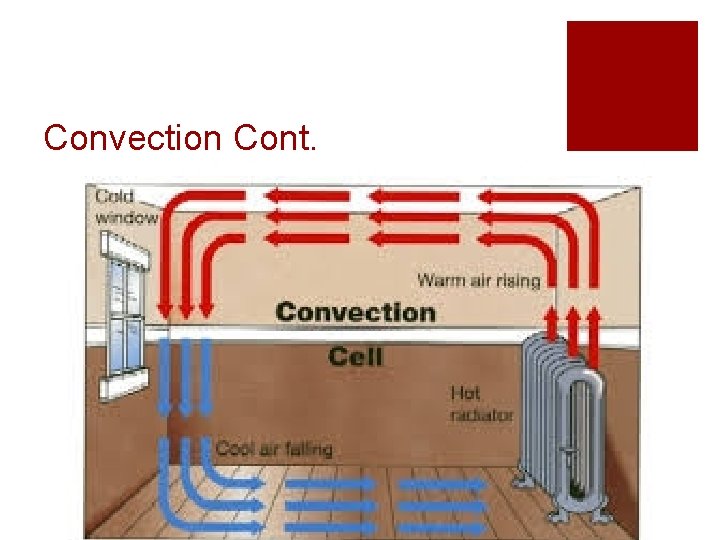
Convection ¡The transfer of heat through movement or circulation ¡Warm air rises and cool air sinks ¡ Creates a “convection” current ¡Remember: Heat moves from warm to cold

Convection cont.

Convection Cont.

Convection Cont.

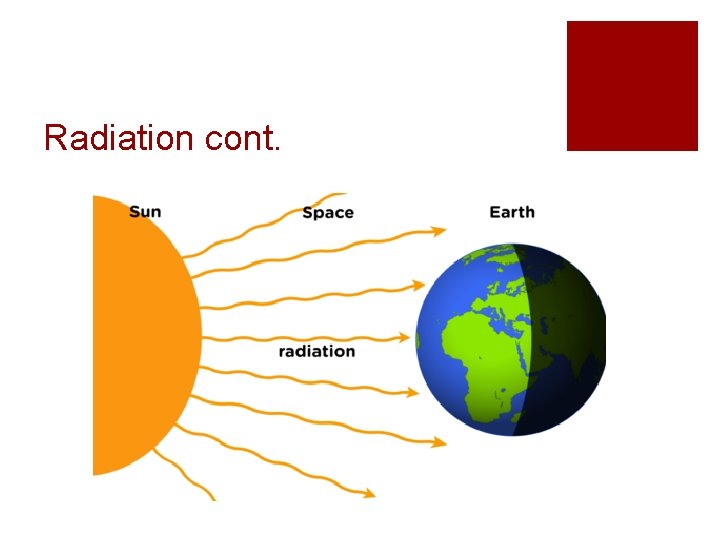
Radiation ¡The transfer of heat by waves or rays

Radiation cont.

Using Radiation ¡We use radiation as a form of alternative energy ¡Solar Radiation


Specific Heat ¡The amount of heat per unit mass required to raise the temperature by 1 degree Celsius ¡The larger a material’s specific heat, the more thermal energy the material can absorb and hold on to

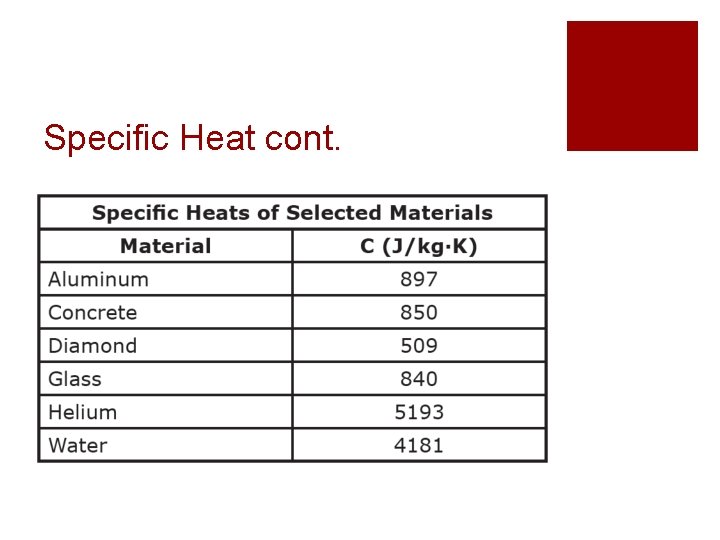
Specific Heat cont.

Specific Heat cont. ¡Water has a higher specific heat than aluminum ¡This means that: ¡Water can hold more thermal energy than aluminum ¡Water stays warmer for much longer than aluminum does

Geothermal Energy ¡We have found ways to use thermal energy as a source of alternative energy ¡Geothermal Energy