States of Matter SOLID LIQUID GAS Thermal Energy




























- Slides: 29

States of Matter SOLID LIQUID GAS

Thermal Energy & Matter § All matter has thermal energy. § Thermal energy is the type of energy that gives matter the ability to move.

Thermal Energy & Matter § The temperature of a substance tells you how much thermal energy it has. LESS MORE

Thermal Energy & Matter Which has more thermal energy? Ice Cream 4 o. C Hot Soup 35 o. C

Thermal Energy & Matter § The amount of thermal energy matter has determines what state (phase) it is in. § There are 3 states of matter:

Thermal Energy & Matter § When the amount of thermal energy that matter has changes, its state changes as well. § Different types of matter change states at different temperatures.

Thermal Energy & Matter § The amount of thermal energy matter has directly determines its state. § This means that the particles of matter have different motions and behaviors at different temperatures.

Thermal Energy & Matter Increasing the amount of thermal energy matter has Decreasing the amount of thermal energy matter has

Physical Change § When matter gains or looses thermal energy, it changes its state. § Even though the matter may be in a different state because of thermal energy amounts, it never stops being the same type of matter - only some extensive properties change.

Physical Change § Changing state (or phase) is only a physical change in matter. No new substances are created. § Water in all three states is consistently H 2 O

Matter in Motion § Every atom that makes every type of matter always has some amount of thermal energy – therefore every atom is always in motion. § The amount of energy changes the motion. § The change in motion of matter changes its physical properties.

SOLIDS: Thermal Energy § Matter in its solid state has the lowest amount of thermal energy (for that type of matter). § Because solids have less thermal energy than liquids or gasses, the atoms in their solid state move very little.

SOLIDS: Particle Motion § Particles (atoms or molecules) of matter in their solid state vibrate in place. § They are packed together and are arranged in a type of pattern.

SOLIDS: Properties § Because matter in its solid state has very little motion, it has specific properties. § Solids have a definite shape. They keep their shape no matter where they are. § Solids have a definite volume – it can be measured (displacement or = x x ) vlwh

SOLIDS: Diagram

LIQUIDS: Thermal Energy § Matter in its liquid state has a medium amount of thermal energy than when it is a solid. § Because liquids have more thermal energy than solids, they move differently than solids.

LIQUIDS: Particle Motion § Particles of matter in their liquid state slide past each other. § They are looser and not arranged in any organized way.

LIQUIDS: Properties § Because matter in its liquid has more energy than solids, it has specific properties. § Liquids have no definite shape; they take the shape of their container. § Liquids have a definite volume – it can be measured (graduated cylinder)

LIQUIDS: Diagram

GASSES: Thermal Energy § Matter in its gas state has the most thermal energy than when it is a solid or a liquid. § Because gasses have more thermal energy than others, they move differently than others.

GASSES: Particle Motion § Particles of matter in their gas state bounce everywhere there is open space. § They are even looser than liquids and not arranged in any organized way.

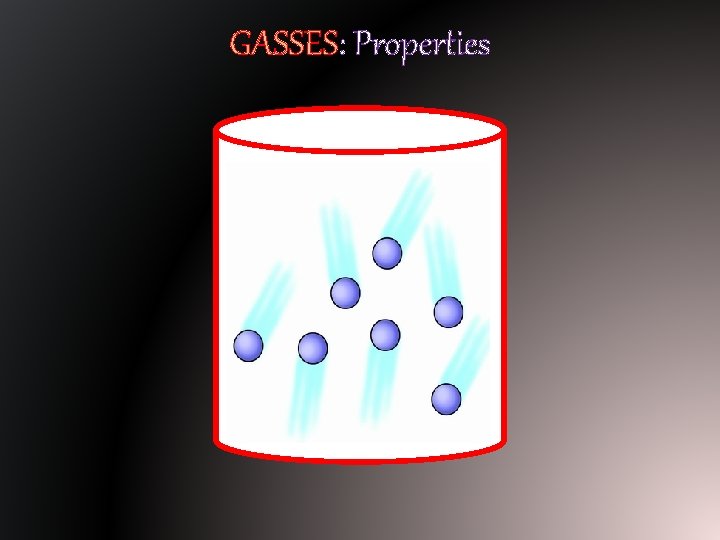
GASSES: Properties § Because matter in its gas state has more energy than solids and liquids, it has specific properties. § Gasses have no definite shape; they take the shape of their container. § Gasses have no definite volume – it can not be directly measured.

GASSES: Properties

Thermal Energy & Particles http: //web. visionlearning. com/custom/chemistry/animations/CHE 1. 1 -an-threestates. shtml

A Fourth State? § If a gas is super heated, then it may change into a 4 th state of matter: Plasma. § The amount of thermal energy required to make matter a plasma is so high, atoms break apart into a mixture of negatively and positively charged particles.

A Fourth State? § Matter in its plasma state cannot exist permanently on earth. § When air (a gas) is struck by lightning, it temporarily turns plasmic while the bolt is coursing through it, but returns to its gas state after the lighting strike.

A Fourth State?

A Fourth State? § The gasses that fuel burning stars are constantly in their plasmic state. § The positive and negative particles shoot out of stars as radiation (gamma rays, radio waves, light, and heat are examples)

A Fourth State?