Chapter 2 Chemical Foundations for Cells Chemical Benefits

Chapter 2 Chemical Foundations for Cells Chemical Benefits and Costs • Chemical pollutants damage ecosystems • Understanding of chemistry provides fertilizers, medicines, etc.

Elements • Fundamental forms of _____ • Can’t be broken apart by normal means • ___ occur naturally on Earth Most Common Elements in Living Organisms • ____ r u • ____ fo ig b • ______ e h T • _____
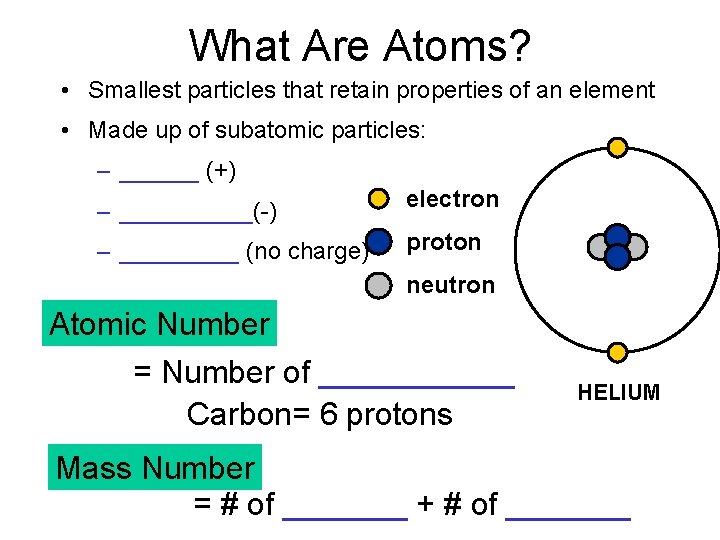
What Are Atoms? • Smallest particles that retain properties of an element • Made up of subatomic particles: – ______ (+) – _____(-) electron – _____ (no charge) proton neutron Atomic Number = Number of ______ Carbon= 6 protons HELIUM Mass Number = # of _______ + # of _______
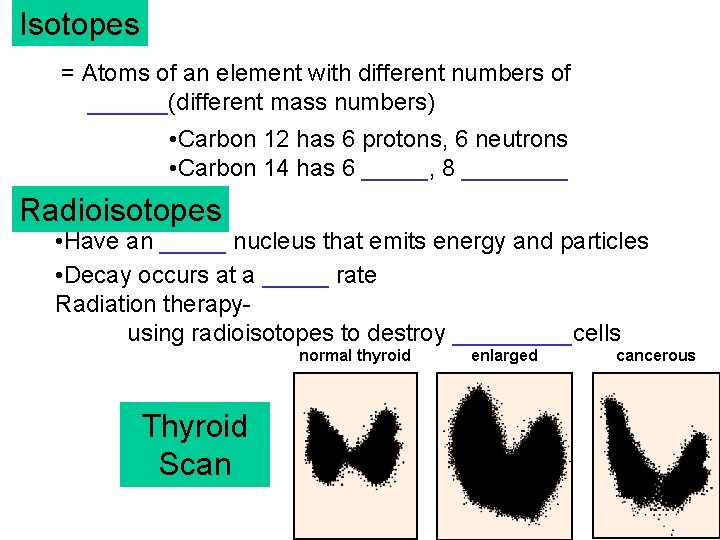
Isotopes = Atoms of an element with different numbers of ______(different mass numbers) • Carbon 12 has 6 protons, 6 neutrons • Carbon 14 has 6 _____, 8 ____ Radioisotopes • Have an _____ nucleus that emits energy and particles • Decay occurs at a _____ rate Radiation therapyusing radioisotopes to destroy _____cells normal thyroid Thyroid Scan enlarged cancerous
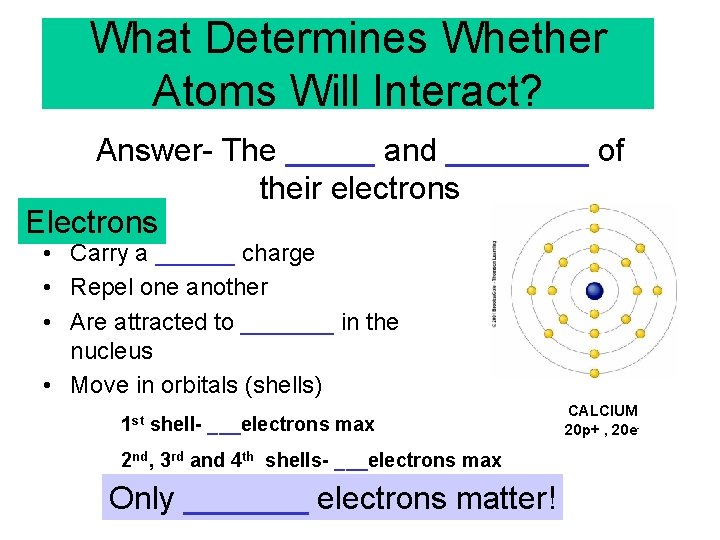
What Determines Whether Atoms Will Interact? Answer- The _____ and ____ of their electrons Electrons • Carry a ______ charge • Repel one another • Are attracted to _______ in the nucleus • Move in orbitals (shells) 1 st shell- ___electrons max 2 nd, 3 rd and 4 th shells- ___electrons max Only _______ electrons matter! CALCIUM 20 p+ , 20 e-
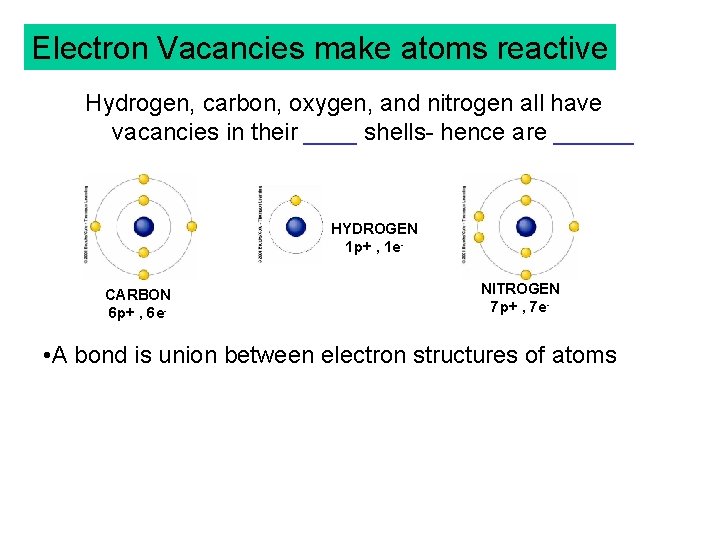
Electron Vacancies make atoms reactive Hydrogen, carbon, oxygen, and nitrogen all have vacancies in their ____ shells- hence are ______ HYDROGEN 1 p+ , 1 e. CARBON 6 p+ , 6 e- NITROGEN 7 p+ , 7 e- • A bond is union between electron structures of atoms
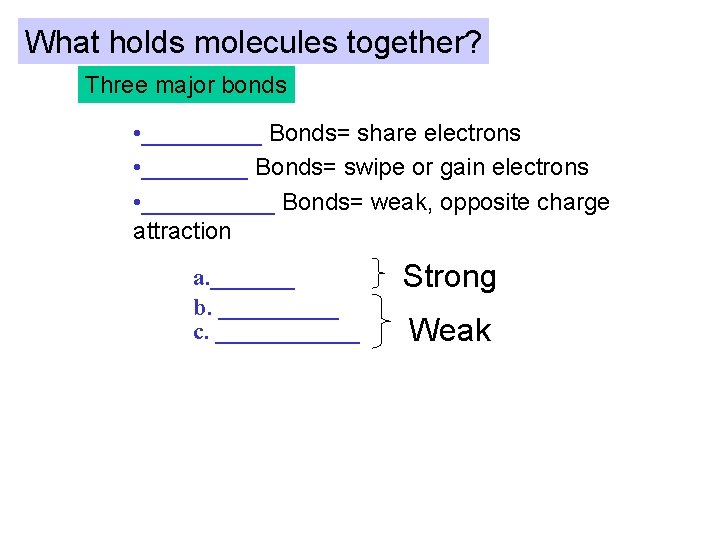
What holds molecules together? Three major bonds • _____ Bonds= share electrons • ____ Bonds= swipe or gain electrons • _____ Bonds= weak, opposite charge attraction a. _______ b. _____ c. ______ Strong Weak
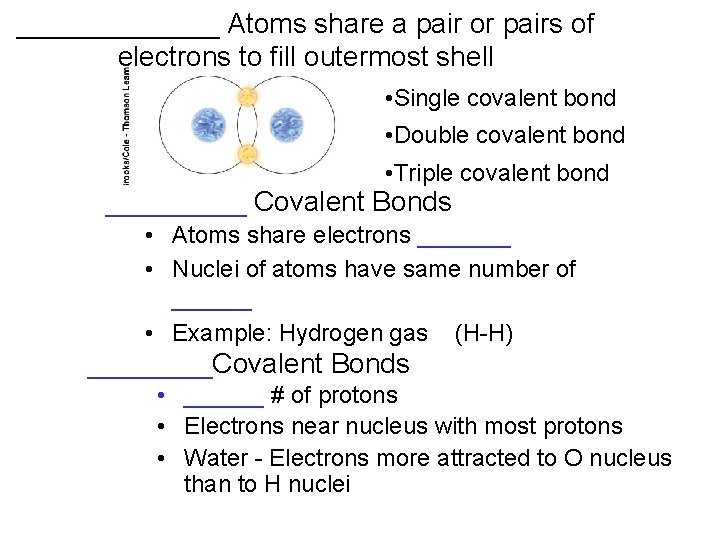
_______ Atoms share a pair or pairs of electrons to fill outermost shell • Single covalent bond • Double covalent bond • Triple covalent bond _____ Covalent Bonds • Atoms share electrons _______ • Nuclei of atoms have same number of ______ • Example: Hydrogen gas (H-H) ____Covalent Bonds • ______ # of protons • Electrons near nucleus with most protons • Water - Electrons more attracted to O nucleus than to H nuclei
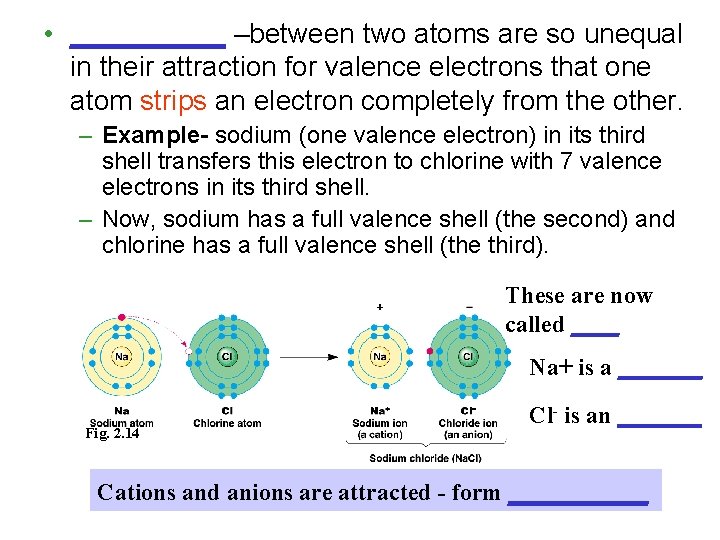
• _____ –between two atoms are so unequal in their attraction for valence electrons that one atom strips an electron completely from the other. – Example- sodium (one valence electron) in its third shell transfers this electron to chlorine with 7 valence electrons in its third shell. – Now, sodium has a full valence shell (the second) and chlorine has a full valence shell (the third). These are now called ____ Na+ is a _______ Fig. 2. 14 Cl- is an _______ Cations and anions are attracted - form _____
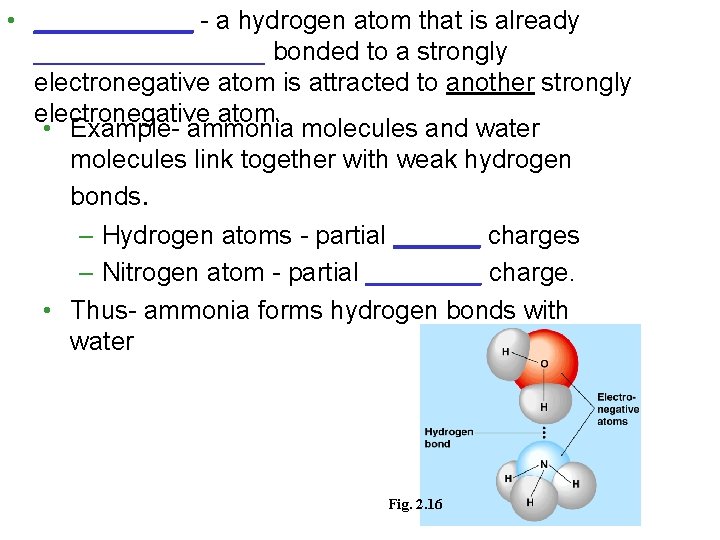
• ______ - a hydrogen atom that is already ________ bonded to a strongly electronegative atom is attracted to another strongly electronegative atom. • Example- ammonia molecules and water molecules link together with weak hydrogen bonds. – Hydrogen atoms - partial ______ charges – Nitrogen atom - partial ____ charge. • Thus- ammonia forms hydrogen bonds with water Fig. 2. 16
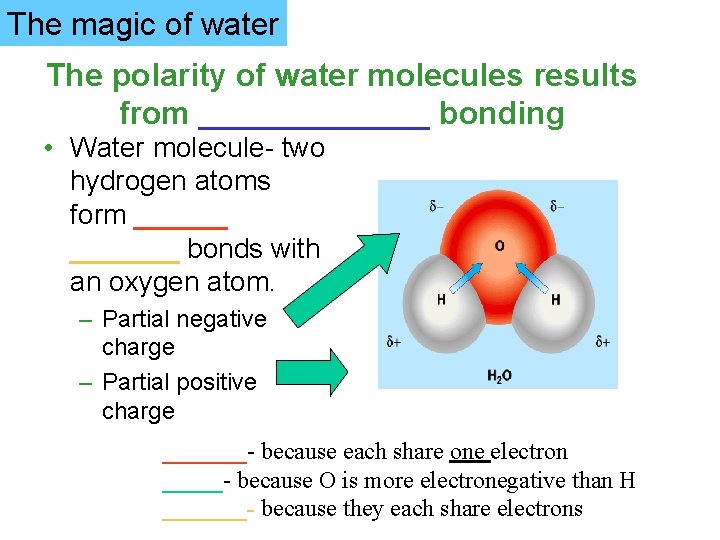
The magic of water The polarity of water molecules results from _______ bonding • Water molecule- two hydrogen atoms form _______ bonds with an oxygen atom. – Partial negative charge – Partial positive charge _______- because each share one electron _____- because O is more electronegative than H _______- because they each share electrons
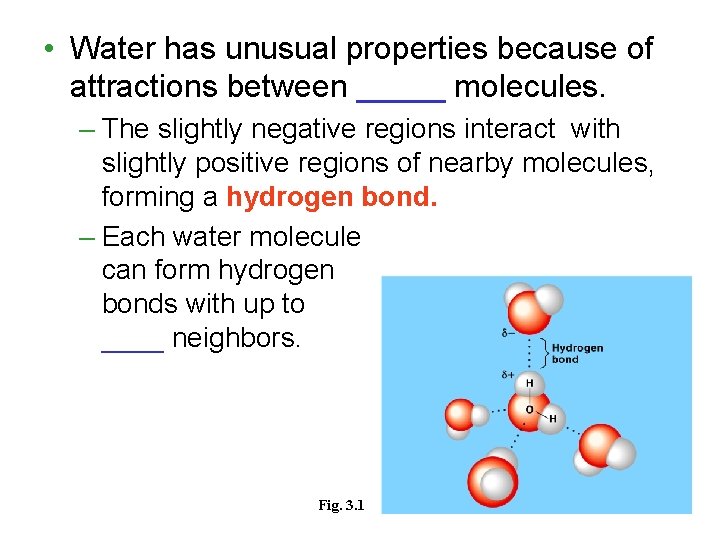
• Water has unusual properties because of attractions between _____ molecules. – The slightly negative regions interact with slightly positive regions of nearby molecules, forming a hydrogen bond. – Each water molecule can form hydrogen bonds with up to ____ neighbors. Fig. 3. 1
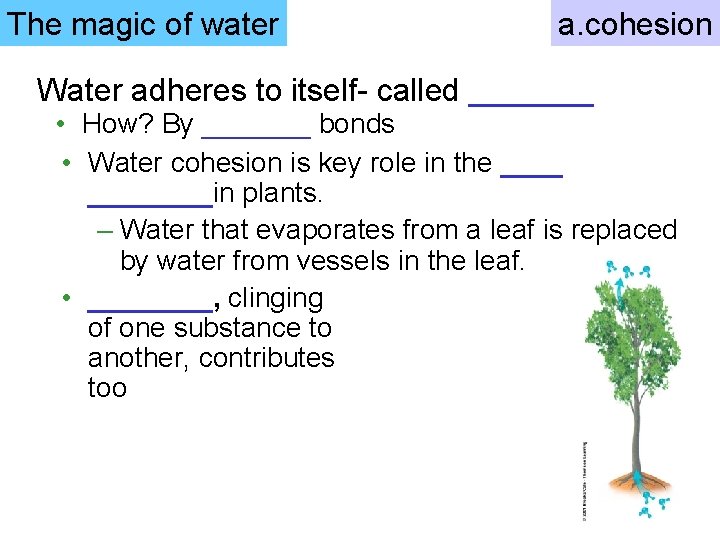
The magic of water a. cohesion Water adheres to itself- called _______ • How? By _______ bonds • Water cohesion is key role in the ________in plants. – Water that evaporates from a leaf is replaced by water from vessels in the leaf. • ____, clinging of one substance to another, contributes too

The magic of water b. Surface tension • _______, a measure of the force necessary to stretch or break the surface of a liquid, is related to cohesion. – Water has h igh ________ – Why? _____ bonds resist breaking the surface. – Water behaves as if covered by an invisible ______.

The magic of water c. Temperature stabilization Water moderates temperatures on Earth • Water _______ air temperatures by ______ heat from warmer air and releasing heat to cooler air. • Water can absorb or release relatively _____ of heat with only a slight change in its own temperature. • Water resists changes in temperature – Why? ________. – Heat must be absorbed to break _________ and is released when hydrogen bonds form.

The magic of water d. evaporative cooling • As a liquid evaporates, the surface of the liquid that remains behind cools - __________. – The most energetic molecules evaporate, leaving the lower kinetic energy molecules behind. • Evaporative cooling moderates __________ in lakes and ponds and prevents terrestrial organisms from overheating. – Evaporation of water from the leaves of plants or the skin of humans removes excess heat.

The magic of water e. Prevent lake freezing • Water (unlike most liquids) is less dense as a solid than as a liquid. – At 0 o. C-water becomes a ___________ with each molecule bonded to the maximum of ______ partners. – Ice is about ______ dense than water at 4 o. C, thus ice _____. If ice sank, eventually all ponds, lakes, and even the ocean would freeze solid.

The magic of water f. Powerful solvent Water dissolves almost anything polar or ionic • ____- A completely ________ mixture of substances. • ______ = dissolving agent • _______ = agent being dissolved. – Example-water is a solvent and sugar the solute.

Hydrophobic vs. hydrophilic • Affinity for water = _____. – Substances with ________ bonds. • no affinity for water = ________. – substances with __________ covalent bonds. • Example-Oils, such as vegetable oil, are non-polar • Hydrophobic molecules are major ingredients of __________.

• How do we measure acidity? – Answer: In ______. • A water molecule dissociates into a hydrogen ion and a hydroxide ion: • H 2 O <=> H+ + OH • At equilibrium the concentration of H+ or OH- is 10 -7 M (25°C). • Hydrogen and hydroxide ions are very reactive, and thus drastically affect the proteins and other molecules of a cell.

• How do we measure acidity? – Answer: In _______. The p. H Scale • Measures ___ concentration of fluid • Change of 1 on scale means ____ change in ____ concentration Highest H+ Lowest H+ 0 -----------7 ----------14 ______ Neutral _____

Examples of p. H • Pure water is neutral with p. H of ____ • Acidic- p. H= < 7. 0 – Stomach acid: p. H ______ – Lemon juice: p. H ____ • Basic- p. H= > 7. 0 – Seawater: p. H 7. 8 - 8. 3 – Baking soda: p. H _____ • Weak acids- Reluctant H+ donors – _____ acid (H 2 CO 3) • Strong acids- Completely give up H+ when dissolved – HCl (hydrochloric acid)
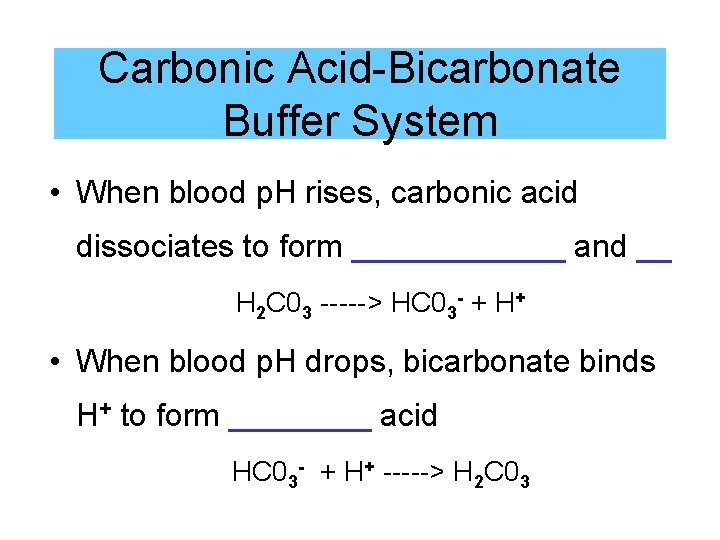
Carbonic Acid-Bicarbonate Buffer System • When blood p. H rises, carbonic acid dissociates to form ______ and __ H 2 C 03 -----> HC 03 - + H+ • When blood p. H drops, bicarbonate binds H+ to form ____ acid HC 03 - + H+ -----> H 2 C 03

• _______ (a product of the formation of carbonic acid from carbon dioxide and water) occurs when rain, snow, or fog has a p. H that is more acidic than 5. 6. • caused primarily by _______ and ______ from burning of fossil fuels Rain in the Adirondack Mountains of upstate New York averages a p. H of ____ Great impact on eggs and early developmental stages of _____ organisms This has done major damage to forests in Europe and substantial damage of forests in North America
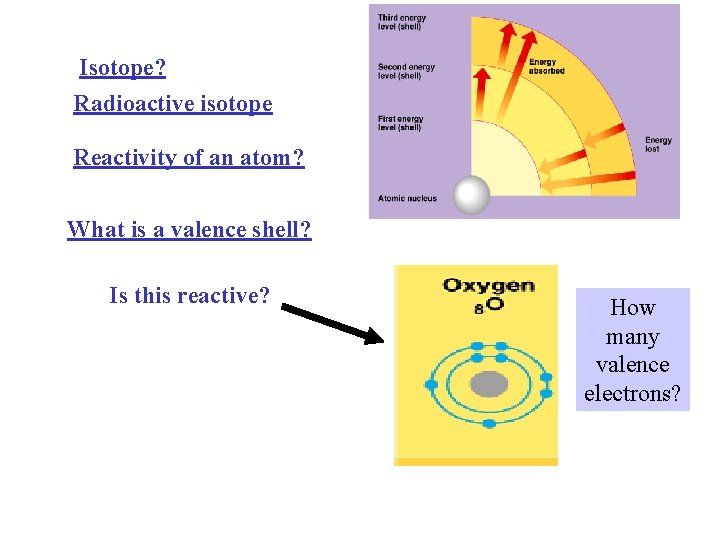
Isotope? Radioactive isotope Reactivity of an atom? What is a valence shell? Is this reactive? How many valence electrons?
- Slides: 25