4 3 CHEMICAL EQUATIONS Pages 202 211 CHEMICAL









- Slides: 12

4. 3 CHEMICAL EQUATIONS Pages 202 -211

CHEMICAL EQUATIONS Chemical Change: a change in the arrangement and connections between ions and atoms (color or energy change, production of gases or precipitate). Chemical Reaction: when one or more changes occur at the same time. Chemical equation. reactions can be represented by an

CHEMICAL EQUATIONS Reactants: pure substances that react in a chemical change (written to the left). Products: pure substances formed in a chemical change that have different properties than those of the reactants (written to the right). Reactants Products Word Equation: nitrogen monoxide + oxygen nitrogen dioxide Symbolic Equation: 2 NO + O 2 2 NO 2

COEFFICIENTS Integers are numbers such as 1, 2, 3… etc. Integers are placed in front of a formula or chemical symbol of an element. Shows the ratio of various compounds in a chemical reaction. Example: 4 K + 02 2 K 20 In other words: 4 K atoms will combine with 1 molecule of O 2 to produce potassium oxide.

LAW OF CONSERVATION OF MASS “Mass is conserved in a chemical reaction; the total mass of products is always equal to the total mass of the reactants in any chemical equation. ” Atoms are conserved; never made or destroyed. WHO SAID THIS? ? French chemist, Antoine Lavoisier and his wife Marie. Anne (1789), measured the masses of reactants and their products of many chemical reactions. Discovered that the total mass never changes during a chemical reaction. This law is true for all chemical equations.


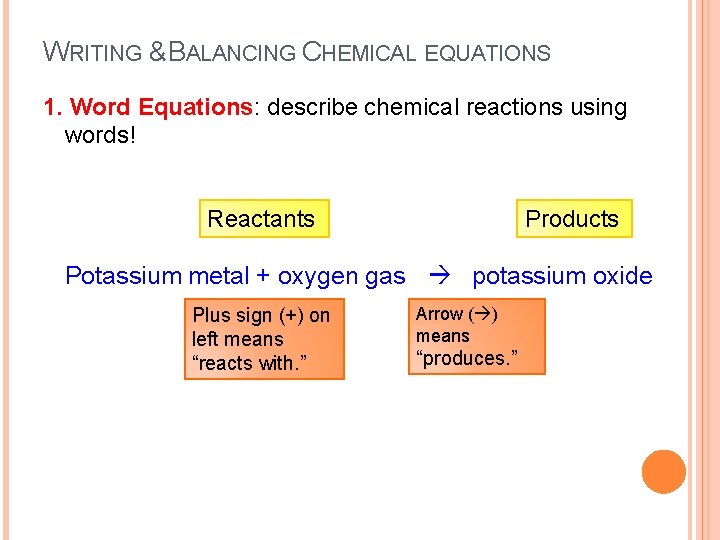
WRITING & BALANCING CHEMICAL EQUATIONS 1. Word Equations: describe chemical reactions using words! Reactants Products Potassium metal + oxygen gas potassium oxide Plus sign (+) on left means “reacts with. ” Arrow ( ) means “produces. ”

2. Skeleton Equations: shows the chemical formulas of the reactants and products. K + 02 K 20 Shows the elements involved but NOT the correct proportion or ratio in which they combine. Recall: Conservation of Mass

3. Balanced Chemical Equation: shows the identities of each pure substance and the equal number (aka coefficient) of elements on each side of the equation (conservation of mass). Skeleton equation: K + 02 K 20 Balanced Equation: 4 K + 02 2 K 2 O Ratio of Reaction = 4: 1: 2 Recall: Coefficient of 1 is not shown

TIPS FOR WRITING EQUATIONS Common compounds: methane (CH 4), ammonia (NH 3) and water (H 20) (need to memorize). Diatomic elements: elements that exist on their own in nature as 2 atom molecules: H 2, N 2, 02, F 2, Cl 2, Br 2, and I 2 Written this way when on their own and NOT part of a compound.

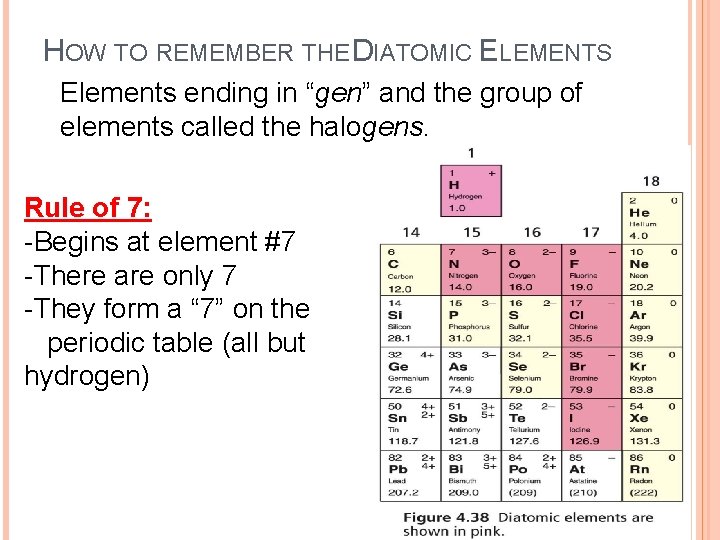
HOW TO REMEMBER THE DIATOMIC ELEMENTS Elements ending in “gen” and the group of elements called the halogens. Rule of 7: -Begins at element #7 -There are only 7 -They form a “ 7” on the periodic table (all but hydrogen)

PRACTICE Eg: Write the balanced equation for the following: hydrogen + nitrogen ammonia Skeleton equation: H 2 + N 2 NH 3 Balanced equation: 3 H 2 + N 2 2 NH 3