Chapter 6 Chemical Bonding Chemical Bonding Chemical Bond

Chapter 6 Chemical Bonding

Chemical Bonding � Chemical Bond – mutual attraction between 2 atoms or ions that holds them together. 1. Ionic Bond – bond resulting from the attraction between ions of opposite charge. � Usually occur between metals and nonmetals. 2. Covalent Bond – bond that occurs when 2 atoms share pairs of valence electrons. � Usually occurs between nonmetal atoms.

Ionic vs. Covalent Bonding � video

Covalent Bonding 1. Nonpolar Covalent Bond – electrons are shared equally between 2 atoms, creating a balanced distribution of charge. 2. Polar Covalent Bond – bond in which electrons are shared unequally because they are pulled to the more electronegative atom. This creates an uneven distribution of charge, or a dipole. �

Predicting Bond Type � The electronegativity difference between 2 atoms can be used to predict the type of bond that exists between them: � 0 – 0. 4 Nonpolar � 0. 5 – 2. 0 Polar � 2. 1 and up Ionic

Covalent Bonding Terms � � Molecule – a neutral group of atoms held together by covalent bonds. Molecular Compound – compound composed of molecules; not ions. Molecular Element – element made up of molecules; not individual atoms. There are 7 diatomic elements:

Electron Dot Symbols � Electron Dot Symbol (Lewis Dot Symbol) – uses dots around an element’s chemical symbol to show many valence electrons it has. � Valence Electrons – electrons in the highest occupied energy level of an atom that participate in chemical bonding.
Valence Electrons

Draw Lewis dot symbols for the following atoms � calcium � iodine � 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 2 � neon � aluminum

Intro to Chemical Bonding � Read section 6. 2 and write 10 questions and answers, pages 164 -175.
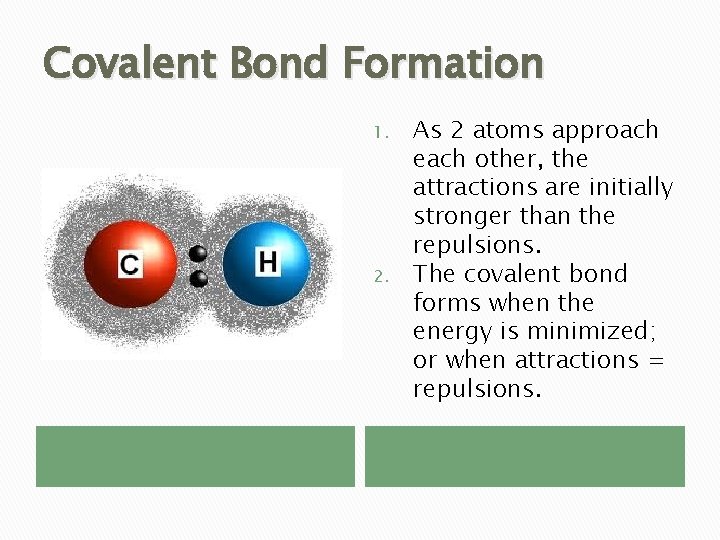
Covalent Bond Formation 1. 2. As 2 atoms approach each other, the attractions are initially stronger than the repulsions. The covalent bond forms when the energy is minimized; or when attractions = repulsions.

Covalent Bond Formation � Bond Length – average distance between 2 atoms covalently bonded. � Bond Energy – energy needed to break a covalent bond. � High bond energy = short bond length

Assignment � #1 -4 page 163, #33, 37 page 196

Lewis Structures � Lewis Structure – diagram that shows the structure of a covalently bonded molecule using only valence electrons. It includes lone pairs and bond pairs of atoms bonded together. � Lone Pair – unshared pair of electrons not involved in a covalent bond. � Bond Pair – pair of electrons shared by 2 atoms to form a covalent bond. � Lone pairs are represented with dots; bond pairs with dashes (-).

Lewis Structures � Single Bond – bond in which one pair of electrons is shared between atoms. � Double Bond – bond in which 2 pairs of electrons are shared. � Triple Bond – bond in which 3 pairs of electrons are shared. � Triple bonds tend to have the highest bond energy and shortest bond length.

How to draw Lewis structures 1. 2. � 3. Determine the total number of valence electrons from all atoms in the molecule. Choose a central atom and arrange all atoms to show they are bonded. C is ALWAYS a central atom. Otherwise, the first atom in the formula is probably the central atom. Fill in lone pairs until all atoms have an octet. H should have just 2 valence electrons.

How to draw Lewis Structures 4. Check to make sure the total number of electrons is correct. If not, change the bond types.

Lewis Structures of Polyatomic Ions � Anions – ADD electrons to the total due to the negative charge. � Cations – must SUBTRACT electrons from the total due to the positive charge.

Resonance Structures – equivalent Lewis structures that can be drawn to represent molecules or ions that cannot be depicted with just one structure.
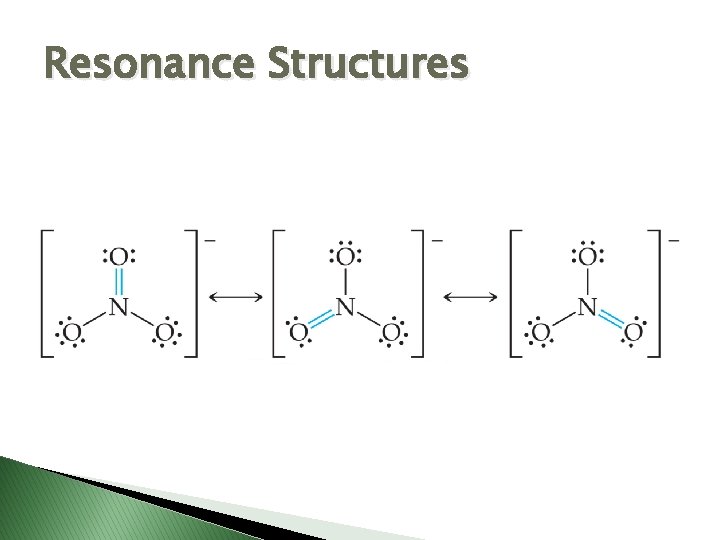
Resonance Structures

Exceptions to the Octet Rule 1. � � 2. � � 3. Less than an Octet Hydrogen will have 2 valence electrons Boron is stable having 6 valence electrons Odd Number of Valence Electrons Causes the molecule or ion to be very unstable and reactive The central atom usually will be lacking an octet because it is lower in electronegativity. Expanded Octet – elements in the 3 rd period of the PT and below can be stable having more than 8 valence electrons as central atoms.

Assignment � #41 -42 page 197 – Draw Lewis Structures and any Resonance structures that exist.

Molecular Geometry � VSEPR Theory – Valence Shell Electron Pair Repulsion theory � Says electron pairs will orient themselves as far apart as possible to minimize repulsions. � Shapes of molecules can be determined by looking at how many electron pairs are around the central atom.
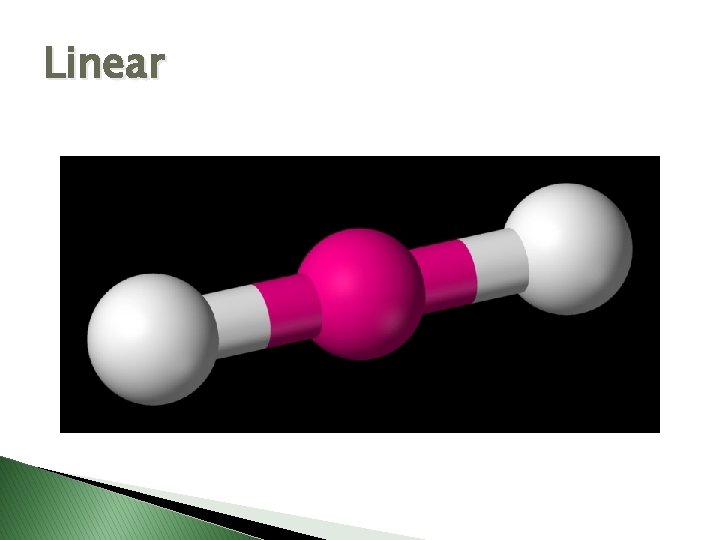
Linear
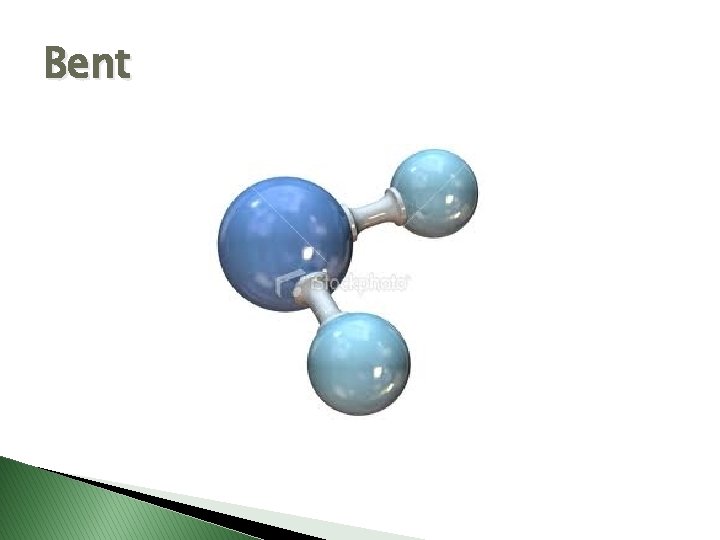
Bent
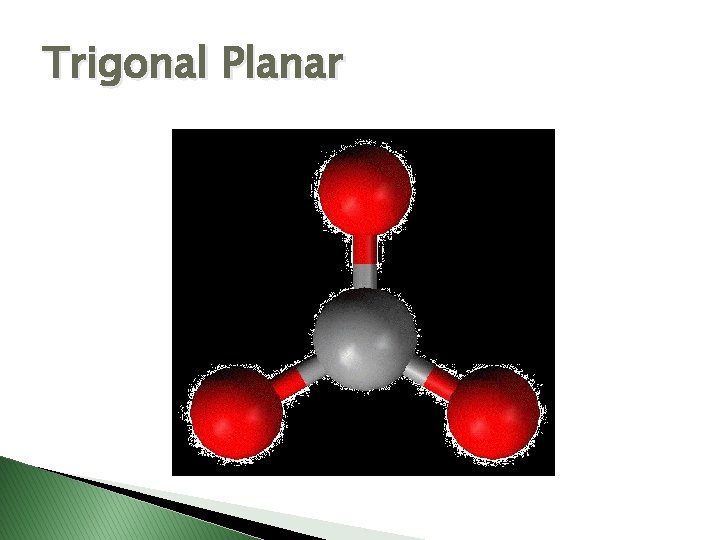
Trigonal Planar
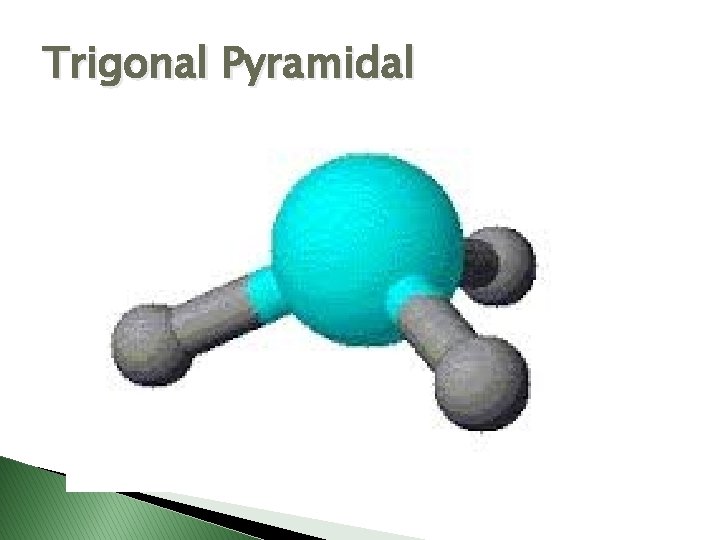
Trigonal Pyramidal
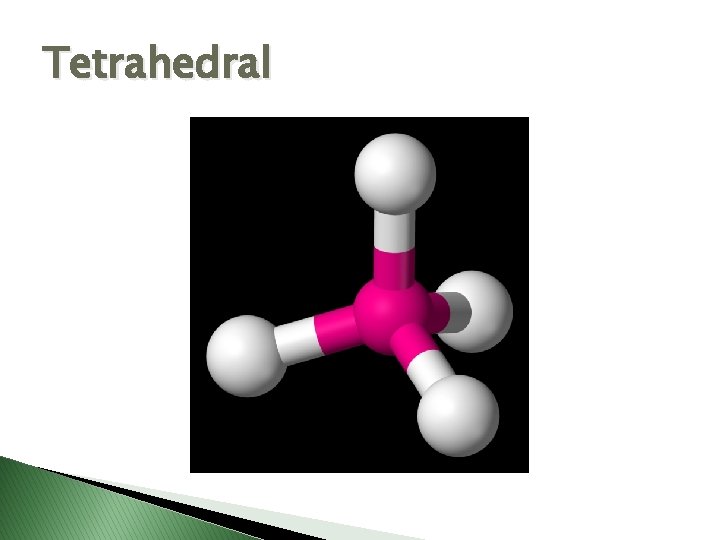
Tetrahedral

Trigonal Bipyramidal
Octahedral

Molecular Polarity �A 1. 2. � � molecule is POLAR if: At least one bond is POLAR. AND…All dipole moment arrows point in the same general direction (left, right, up, down), which means electrons are pulled to same side of molecule. Dipole moment – arrow drawn to show the more electronegative atom in a polar bond. Lewis structures must be drawn in the correct shape to predict polarity.
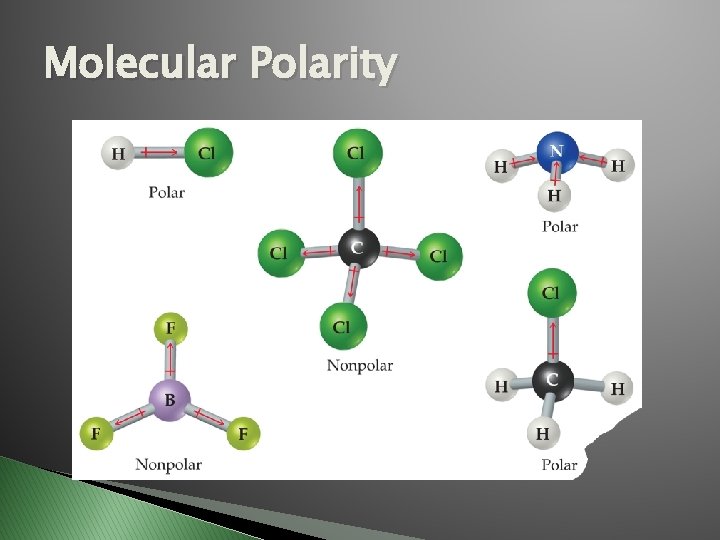
Molecular Polarity

Assignment: #47 -48 page 197 1. 2. 3. Draw Lewis Structures showing the correct shape. Add dipole moment arrows (if there any) using electronegativity chart. Polar or Nonpolar?

Ionic vs. Covalent Bonding 1. 2. 3. 4. 5. Explain the difference between ionic and covalent bonds. Describe the difference between the strength of covalent and ionic bonds. Compare the melting and boiling points of ionic and molecular compounds. Describe the typical physical state of ionic and molecular compounds. Explain how ionic compounds are able to conduct electricity.

Ionic vs. Covalent Bonding � Ionic Bonds occur between ions of opposite charge, Covalent Bonds occur between atoms that share electrons. � Ionic bonds are STRONGER than covalent bonds. � Ionic compounds have higher melting and boiling points due to stronger attractions. � Ionic compounds are brittle solids, covalent molecules are often gases. � Ionic compounds can conduct electricity when dissolved in water.

Metallic Bonding-Section 6. 4 � Read the section, pages 181 -182. � Describe the main idea of each of the three topics in bold.

Metallic Bonding � Metallic Bond – bond that results from the attraction between metal atoms and a surrounding “sea of valence electrons. ” � Electrons are delocalized in metals; they are able to move from atom to atom through a network of empty d orbitals. � Metallic bond animation � Metal Properties: � High conductivity, malleable, ductile, high heat of vaporization

What Type of Bond is Present? � CO 2 � Fe 2 O 3 � Fe solid � C 12 H 22 O 11 � Ca. CO 3 � Aluminum can

Intermolecular Forces � Attractive forces that exist between molecules � Much weaker than the 3 types of chemical bonding � 3 Types: Dipole forces, hydrogen bonds, London-dispersion forces � Video Explanation
- Slides: 39