Organic Organic Chemistry and AP Organic is not

















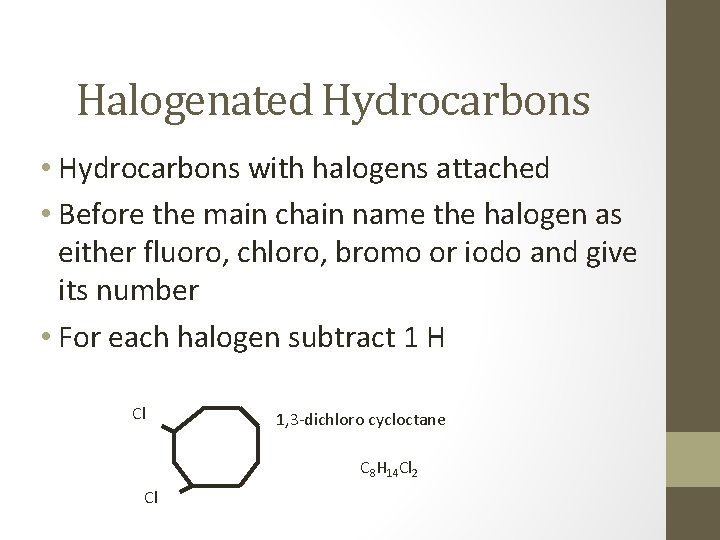













- Slides: 52

Organic

Organic Chemistry and AP • Organic is not really on the AP test. • In several cases, they have used organic molecules to go over different points (Lewis Dot, hybridization, intermolecular forces). • I am just zipping through this as a refresher because there is always a weird question on the test.

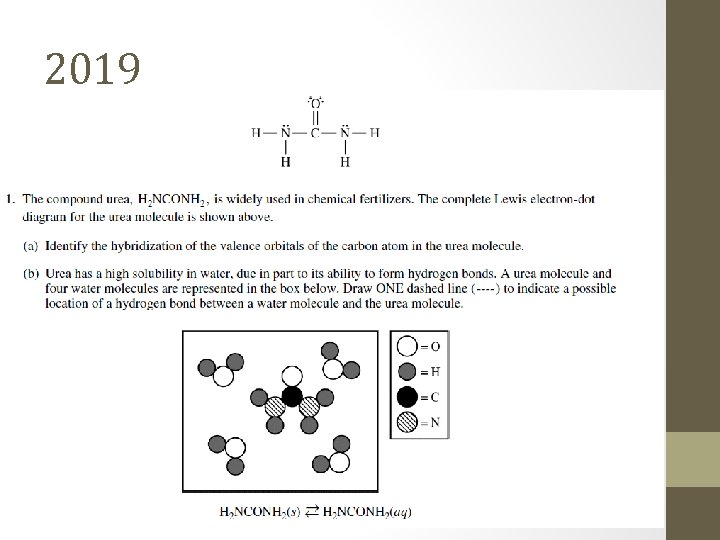
2019

Carbon • Carbon has the unusual ability of bonding to itself to form long chains or rings of carbon atoms. • Carbon forms strong bonds to other nonmetals such as hydrogen, nitrogen, oxygen, sulfur, and the halogens. • Several million (11 million-plus) are known, and the number continues to grow rapidly. • Carbon is the most important compound to the biological world.

Organic Chemistry • Organic Chemistry is the study of carboncontaining compounds and their properties. • Oxides and carbonates that contain carbon are not considered to be organic, they are inorganic. • The original distinction between organic and inorganic was based on whether a compound was produced by living things. • Organic chemistry plays a vital role in our quest to understand living systems.

• Industrial organic chemistry produces synthetic fibers (nylon, rayon), plastics, rubber (latex) explosives, artificial sweeteners, vinegar and pharmaceuticals that are such an important part of modern life. • The energy on which we rely so heavily on to power our civilization is based mostly on organic materials found in coal and petroleum.

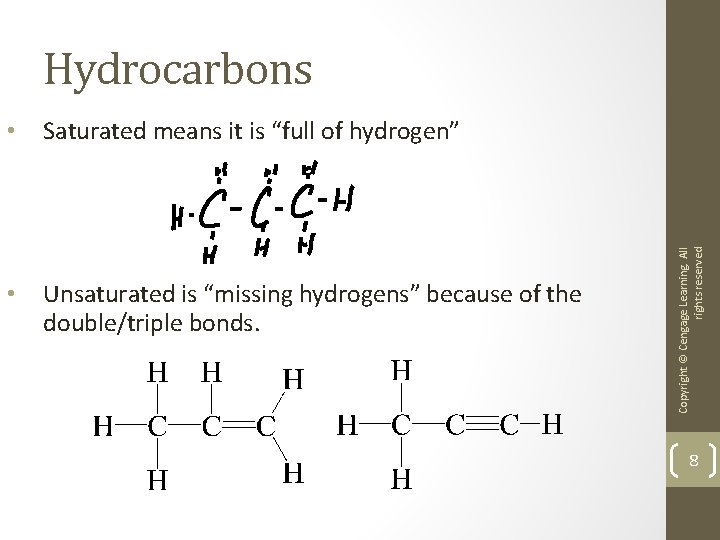
Saturated and Unsaturated Hydrocarbons • Hydrocarbons are compounds composed of carbon and hydrogen. • Saturated hydrocarbons contain all carbon-carbon single bonds (each carbon is bonded to four atoms). • Unsaturated hydrocarbons contain carbon-carbon double or triple bonds.

• Saturated means it is “full of hydrogen” • Unsaturated is “missing hydrogens” because of the double/triple bonds. Copyright © Cengage Learning. All rights reserved Hydrocarbons 8

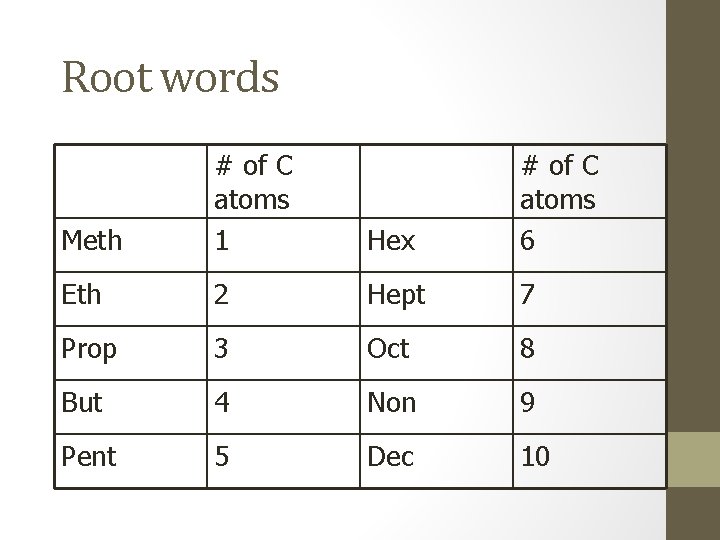
Root words Meth # of C atoms 1 Hex # of C atoms 6 Eth 2 Hept 7 Prop 3 Oct 8 But 4 Non 9 Pent 5 Dec 10

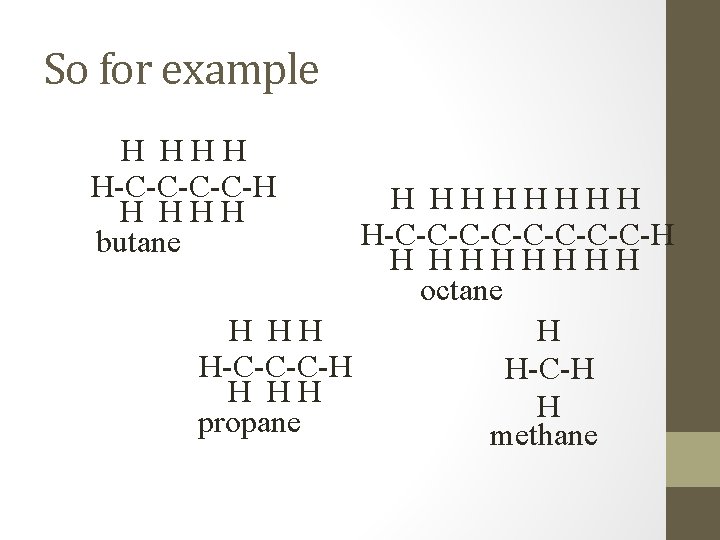
So for example H HHH H-C-C-H H HHH butane H HHHHHHH H-C-C-C-C-H H HHHHHHH octane H HH H H-C-C-C-H H HH H propane methane

Molecular Formulas • Alkanes always have the molecular formula of: • Cx. H 2 x+2 • 2 H on every C except the end, they get 3 • Hexane • C 6 H 14 molecular formula Lewis Dot, or Structural Formula HHHHHH H-C-C-C-H HHHHHH

Skeleton Formulas • Drawing Lewis Dot structural formulas for long organic compounds can get rather tedious. • Skeleton structures are just a jagged line. At each corner there is a carbon • They don’t write the C’s or the H’s, but all other elements are written • Assume all extra spaces are filled with H

Condensed Structural Formulas • These show all the each carbon atom bonded together with everything bonded to carbon as you would write in a molecular formula. • Acetate • CH 3 COO

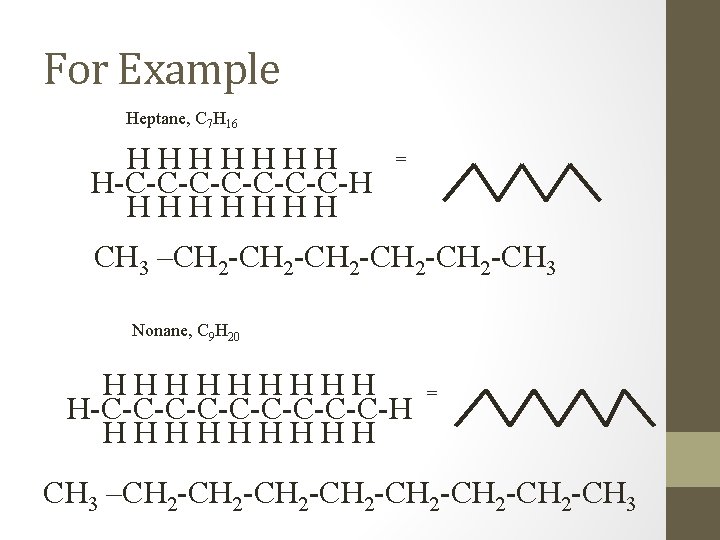
For Example Heptane, C 7 H 16 HHHHHHH H-C-C-C-C-H HHHHHHH = CH 3 –CH 2 -CH 2 -CH 3 Nonane, C 9 H 20 HHHHH H-C-C-C-C-C-H HHHHH = CH 3 –CH 2 -CH 2 -CH 3

Isomers • Isomers- compounds with the same molecular formula but different structural formulas • Different structural formulas mean it has different properties • Butane is the first alkane with a possible isomer HHHH = H-C-C-H HHHH H or H-C- C - C-H Both are C H H HCH H H 4 10

Copyright © Cengage Learning. All rights reserved Butane 16

Naming Isomers • Name the longest chain possible. • As a prefix name the chain attached with – yl on the end and give the number of the carbon atom it is attached to 2 1 6 4 3 Longest Chain 5 7 3 ethyl heptane • It could also be 5 ethyl heptane if you started numbering from the other side, when given an option always go with the Lower number!!!

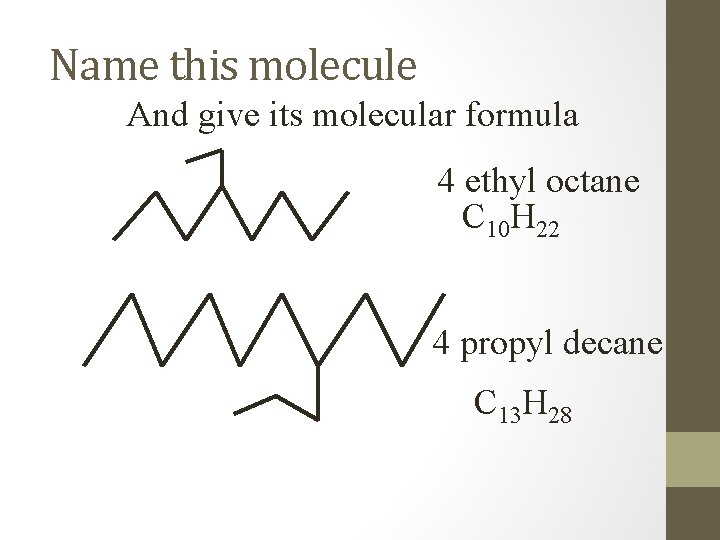
Name this molecule And give its molecular formula 4 ethyl octane C 10 H 22 4 propyl decane C 13 H 28

Cyclic Hydrocarbons • A hydrocarbon that is a ring instead of a chain. To name it, give it the prefix “cyclo” • Molecular Formula • Subtract 2 H from Cx. H 2 x+2 • Cx. H 2 X • cyclobutane HH H-C-C-H HH C 4 H 8

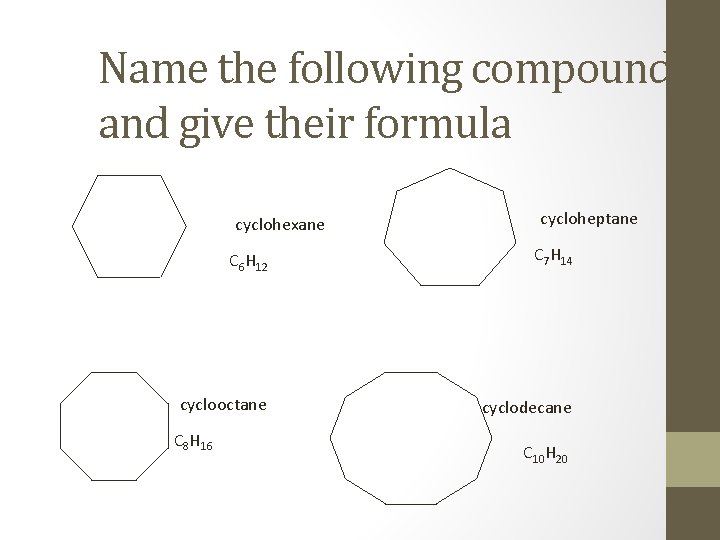
Name the following compounds and give their formula cyclohexane cycloheptane C 6 H 12 C 7 H 14 cyclooctane cyclodecane C 8 H 16 C 10 H 20

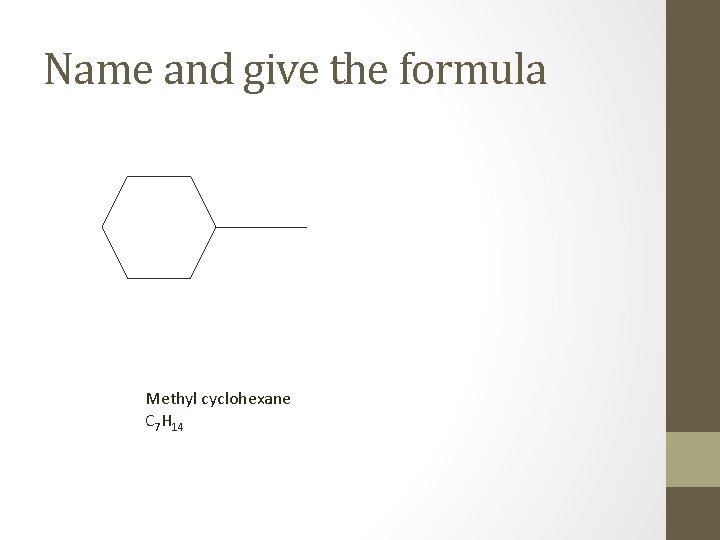
Name and give the formula Methyl cyclohexane C 7 H 14

Alkenes • Contain a double bond • They get the suffix “-ene” and the number of the carbon atom the double bond is on (lowest number) • Molecular formula • Subtract 2 H for each double bond from Skeleton fomula Cx. H 2 x+2 1 butene H H-C=C-C-C-H H C 4 H 8

Alkynes • Contain a triple bond • They get the suffix “-yne” and the number of the carbon atom the triple bond is on. Molecular formula • subtract 4 H for each triple bond from Cx. H 2 x+2 Skeleton fomula H H H 2 pentyne H-C-C=C-C-C-H C 5 H 8

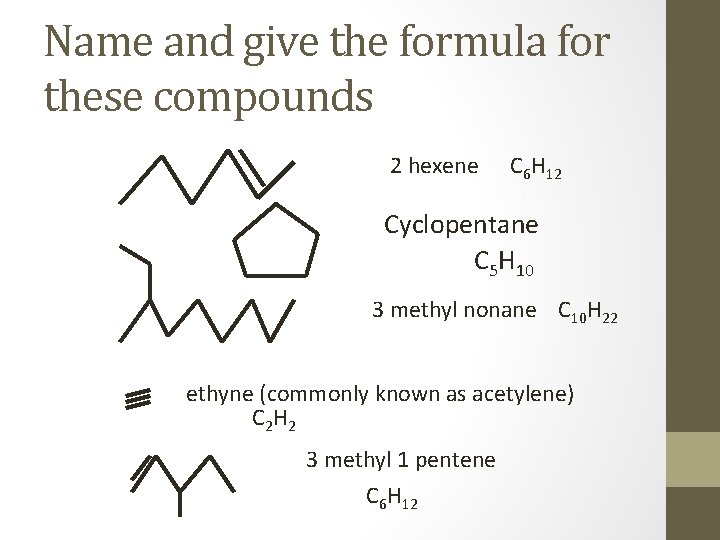
Name and give the formula for these compounds 2 hexene C 6 H 12 Cyclopentane C 5 H 10 3 methyl nonane C 10 H 22 ethyne (commonly known as acetylene) C 2 H 2 3 methyl 1 pentene C 6 H 12

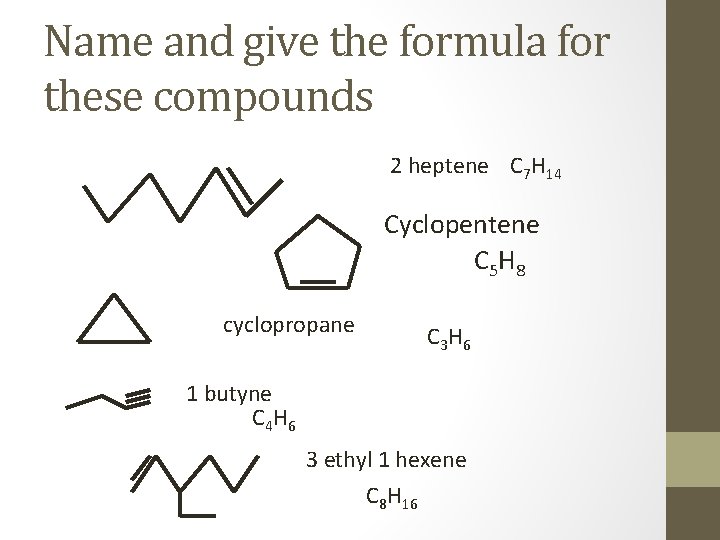
Name and give the formula for these compounds 2 heptene C 7 H 14 Cyclopentene C 5 H 8 cyclopropane C 3 H 6 1 butyne C 4 H 6 3 ethyl 1 hexene C 8 H 16

Doubles and triples • If you have two of the same thing put “di” in front of it • If you have three of the same thing put “tri” in front of it

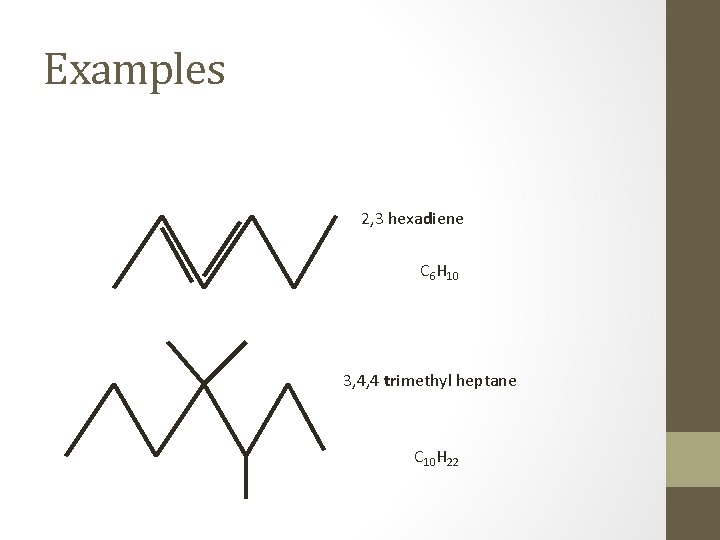
Examples 2, 3 hexadiene C 6 H 10 3, 4, 4 trimethyl heptane C 10 H 22

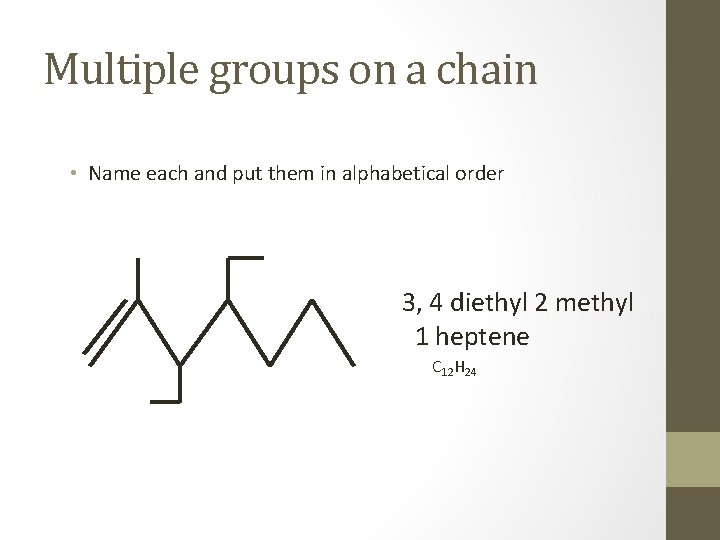
Multiple groups on a chain • Name each and put them in alphabetical order 3, 4 diethyl 2 methyl 1 heptene C 12 H 24

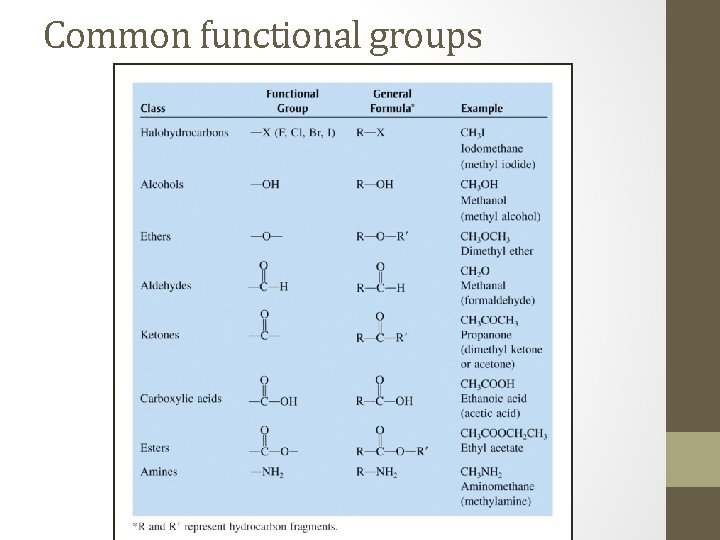
Common functional groups

Functional Groups • Atoms other than hydrogen or carbon covalently bonded to a carbon atom in an organic molecule. • Most commonly oxygen, nitrogen, or the halogens. • The presence of a functional group drastically changes the chemical properties of a molecule.

Different Functional groups with a 2 carbon chain • Ethane- gas (found in natural gas) • Ethanol- grain alcohol (drinkable) • Ethanoic acid- vinegar • Diethyl ether- starting fluid • Chloro fluoro ethane (CFC’s used as refrigerants) • Ethanal- foul smelling liquid (similar to formaldehyde)
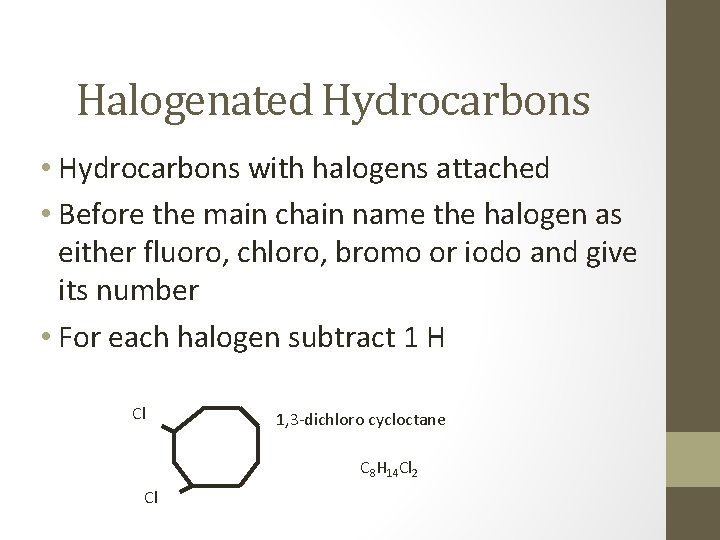
Halogenated Hydrocarbons • Hydrocarbons with halogens attached • Before the main chain name the halogen as either fluoro, chloro, bromo or iodo and give its number • For each halogen subtract 1 H Cl 1, 3 -dichloro cycloctane C 8 H 14 Cl 2 Cl

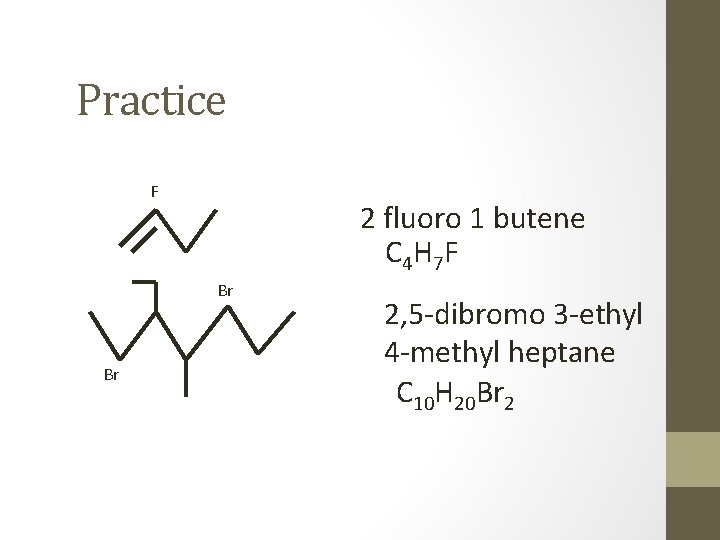
Practice F 2 fluoro 1 butene C 4 H 7 F Br Br 2, 5 -dibromo 3 -ethyl 4 -methyl heptane C 10 H 20 Br 2

Alcohols • Hydrocarbons with an –OH attached • To name it, give it the suffix –(an)ol and the number the OH is attached to • Normally you subtract one H from the main group and put an OH on the end (to signify it is an alcohol) H O C 2 H 5 OH Ethanol OH C 3 H 7 OH 2 propanol Commonly Isopropanol or Rubbing alcohol

Aldehydes • Hydrocarbons with a =O on the outer edge of the chain • (most have a foul stench, like formaldehyde or methanal) • To name it add the suffix “–al” • For the formula subtract 2 H and add O =O O= hexanal C 6 H 12 O octanal C 8 H 16 O

Ketones • Hydrocarbons with a =O not on the edge of the compound • To name it add the suffix “–one” • For the formula subtract 2 H and add O O= O= cyclopropanone C 3 H 4 O 3 -nonanone C 9 H 18 O

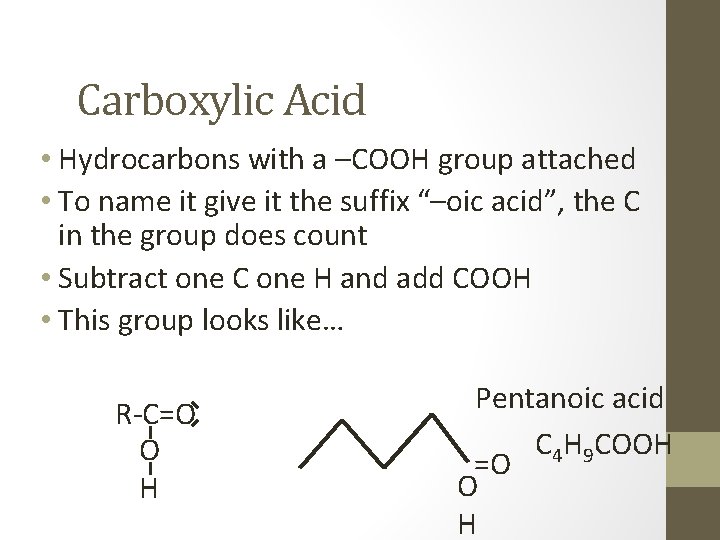
Carboxylic Acid • Hydrocarbons with a –COOH group attached • To name it give it the suffix “–oic acid”, the C in the group does count • Subtract one C one H and add COOH • This group looks like… R-C=O O H Pentanoic acid C 4 H 9 COOH =O O H

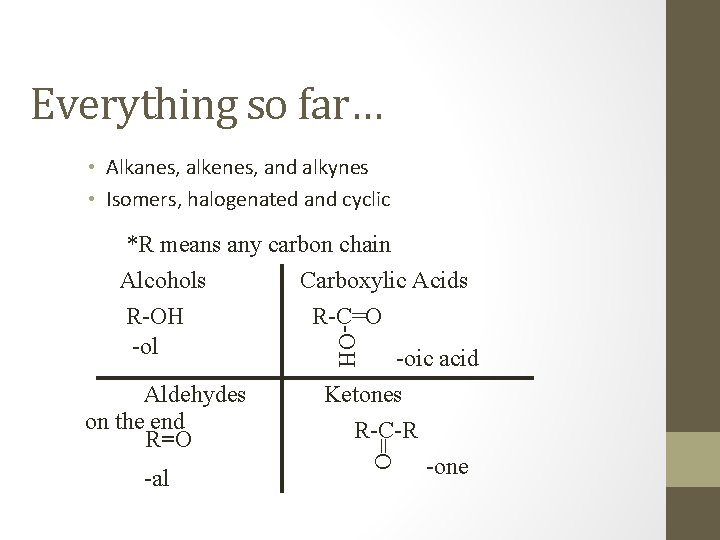
Everything so far… • Alkanes, alkenes, and alkynes • Isomers, halogenated and cyclic -OH *R means any carbon chain Alcohols Carboxylic Acids R-OH R-C=O -ol -oic acid -al Ketones R-C-R =O Aldehydes on the end R=O -one

Predicting organic reactions • Combustion reactions occur when an organic compound is burned in oxygen. • The products of a complete combustion are water vapor and carbon dioxide. • C 6 H 12 O 6 + 6 O 2 6 H 2 O+ 6 CO 2 • Incomplete combustion is when oxygen is in short supply. Carbon monoxide and carbon are produced! • Carbon monoxide is a poisonous gas. • Carbon will make things black

What is petroleum? • Also known as crude oil • It is a thick black sludge • It comes from ancient plant and animal life long since buried and kept under extreme pressure for millions of years. • It is composed of countless different organic compounds.

What is made from petroleum • Gasoline, kerosene, and rocket fuel • Most plastics and other polymers (elastomers and fibers) • Synthetic rubbers and fabrics • Most pharmaceutical drugs • And several other things • If we run out of petroleum it would have a devastating effect on us

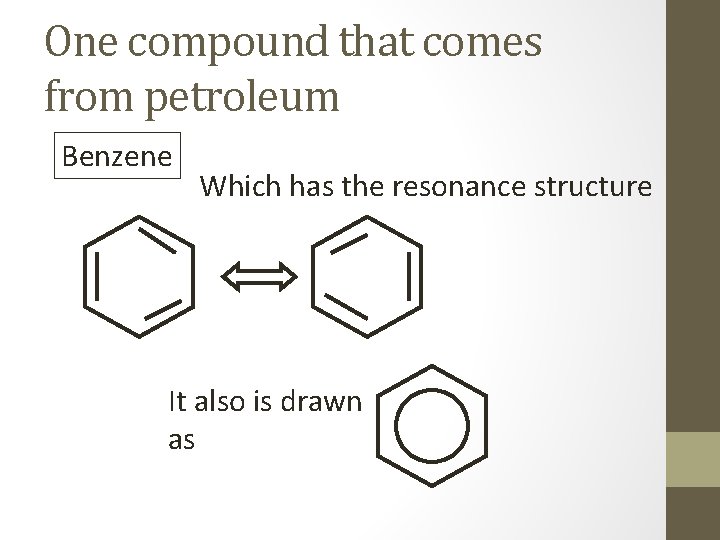
One compound that comes from petroleum Benzene Which has the resonance structure It also is drawn as

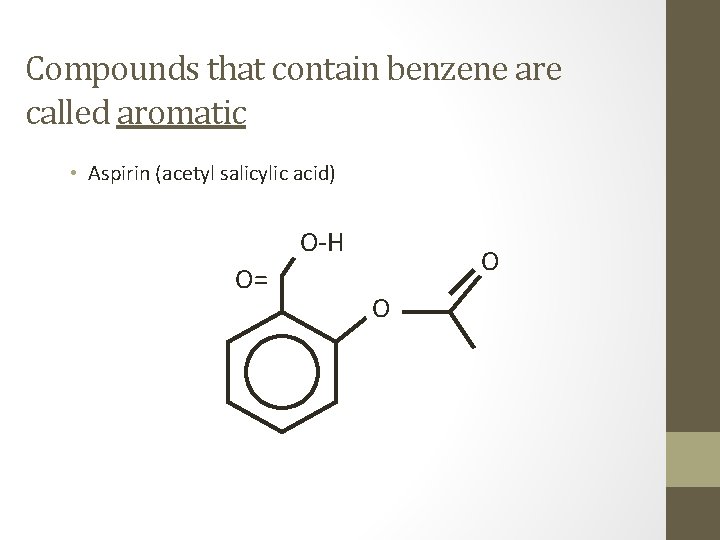
Compounds that contain benzene are called aromatic • Aspirin (acetyl salicylic acid) O-H O= O O

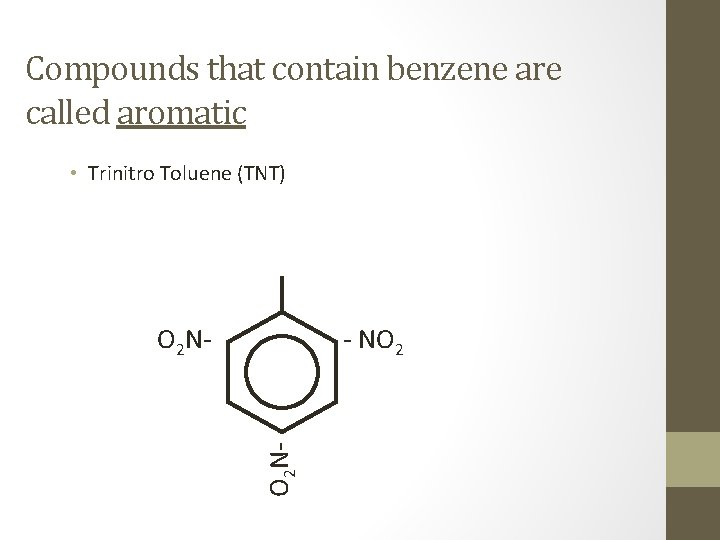
Compounds that contain benzene are called aromatic • Trinitro Toluene (TNT) - NO 2 O 2 N-

A few other aromatics • Vinyl, napthalene (found in moth balls), acetaminophen, penacillin • Benzene is an extremely common organic compound • The fact that the double bonds flip back and forth (called resonance) give it a very stable structure

Polymerization

Esters O= • Esters- a functional group in the middle of a carbon chain; R-COO-R • It gets the suffix –oate It is very similar to carboxylic acids ~In fact, it is formed by a carboxylic acid an alcohol R-C-O-R + H-O-R R-C-O-R the water came from… O= O= R-C-O-H + H-O-H

Now if you have a few compounds that have both a O= + H-O-H H-O-R-C-O-H O= O= O= Carboxylic acid end +an alcohol end H-O-R-C-O-H + They could form an ester that looks like… But the compound still has a… And an alcohol end Carboxylic acid end So it could repeat this process thousands even millions of times and make a whole bunch of… poly esters Of course, the scientific prefix for “whole bunch of” is

This is the basis for a polymer • Polymer-A large chain-like molecule composed of smaller molecules linked together • The smaller units it is made up of are called monomers • monomers need to have ends that can join together (or stack on top of one another) • Like an extension cord or markers • So you could (infinitely) join them together to make a large polymer

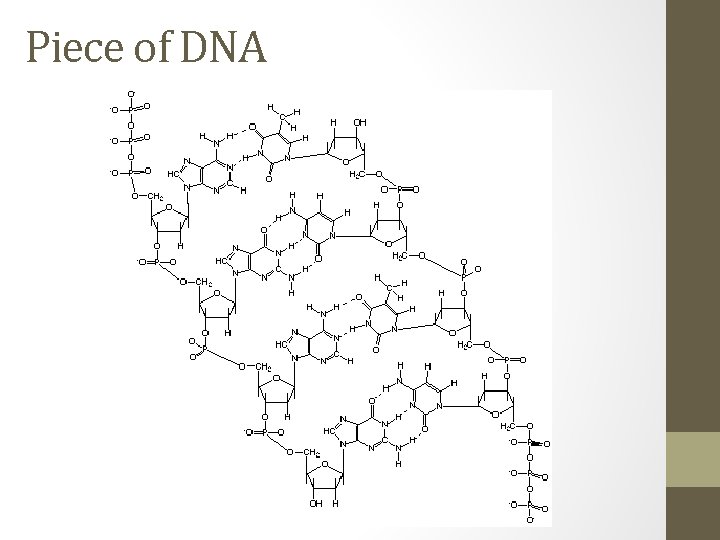
Polymers can get very large • common polymers have a molecular mass of around 50, 000 g/mol • The first molecules seen under a microscope were polymer chains • Common polymers include things like… • Nylon, Kevlar©, latex, PVC, rubber, acrylic, vinyl, Deoxyribonucleic acid (DNA) and carbohydrates

Piece of DNA

Polymers are put into three classes Plastics Elastomers. Polymers that can be stretched to 10 x their normal size and return to their original shape Elastic Fibers Polymers that cannot stretch and flex or be reshaped more than fibers once formed but less than Elastomers Nylon and Acrylic Polypropylene polystyrene and PVC (polyvinyl chloride)