FischerRosanoff Convention Before 1951 only relative configurations could

Fischer-Rosanoff Convention • Before 1951, only relative configurations could be known. • Sugars and amino acids with same relative configuration as (+)-glyceraldehyde were assigned D and same as (-)glyceraldehyde were assigned L. • With X-ray crystallography, now know absolute configurations: D is (R) and L is (S). • No relationship to dextro- or levorotatory. =>
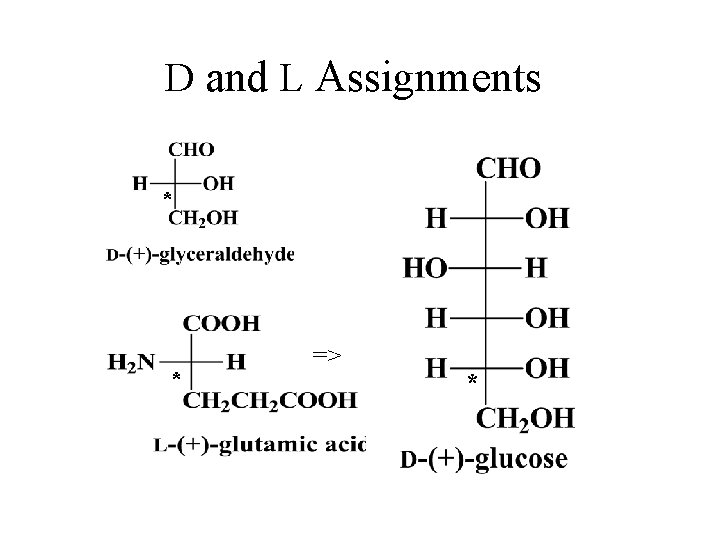
D and L Assignments * * => *

Properties of Diastereomers • Diastereomers have different physical properties: m. p. , b. p. • They can be separated easily. • Enantiomers differ only in reaction with other chiral molecules and the direction in which polarized light is rotated. • Enantiomers are difficult to separate. =>

Resolution of Enantiomers React a racemic mixture with a chiral compound to form diastereomers, which can be separated. =>

Chromatographic Resolution of Enantiomers =>


Organic Chemistry, 5 th Edition L. G. Wade, Jr. Chapter 6 Alkyl Halides: Nucleophilic Substitution and Elimination
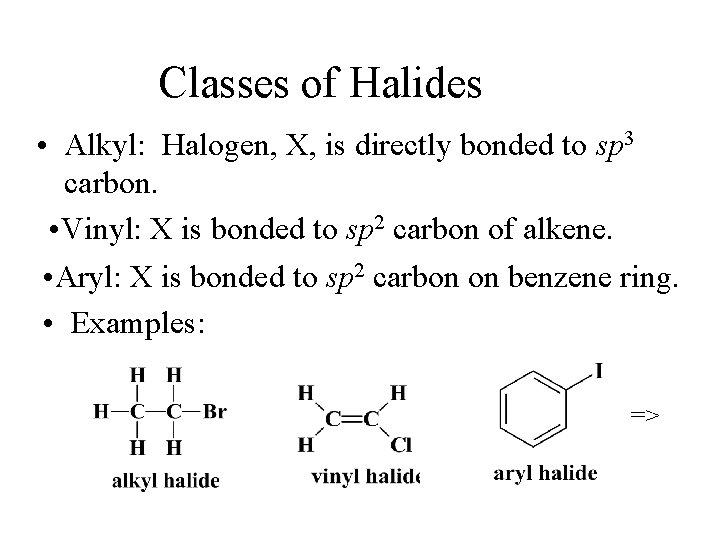
Classes of Halides • Alkyl: Halogen, X, is directly bonded to sp 3 carbon. • Vinyl: X is bonded to sp 2 carbon of alkene. • Aryl: X is bonded to sp 2 carbon on benzene ring. • Examples: =>

Polarity and Reactivity • Halogens are more electronegative than C. • Carbon-halogen bond is polar, so carbon has partial positive charge. • Carbon can be attacked by a nucleophile. • Halogen can leave with the electron pair. =>

Classes of Alkyl Halides • Methyl halides: only one C, CH 3 X • Primary: C to which X is bonded has only one C-C bond. • Secondary: C to which X is bonded has two C-C bonds. • Tertiary: C to which X is bonded has three C-C bonds. =>
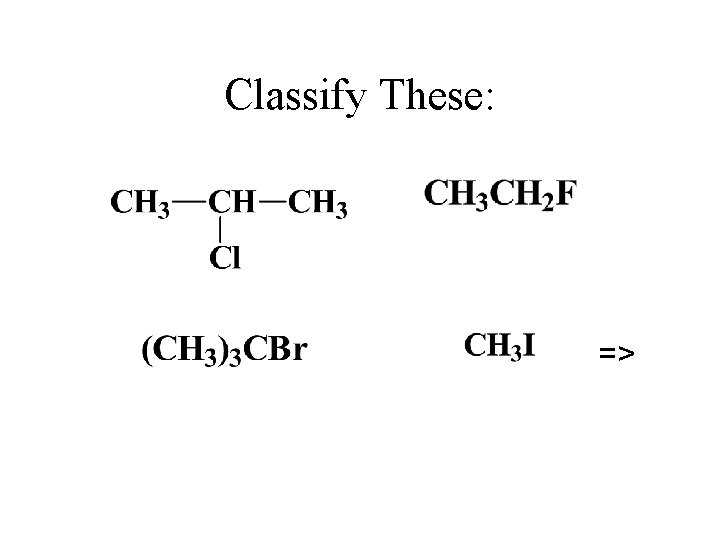
Classify These: =>

Dihalides • Geminal dihalide: two halogen atoms are bonded to the same carbon • Vicinal dihalide: two halogen atoms are bonded to adjacent carbons. =>

IUPAC Nomenclature • Name as haloalkane. • Choose the longest carbon chain, even if the halogen is not bonded to any of those C’s. • Use lowest possible numbers for position. =>

Systematic Common Names • Name as alkyl halide. • Useful only for small alkyl groups. • Name these: =>

“Trivial” Names • CH 2 X 2 called methylene halide. . • CHX is a haloform. 3 • CX 4 is carbon tetrahalide. • Examples: –CH 2 Cl 2 is methylene chloride –CHCl 3 is chloroform -CHI 3 is iodoform –CCl 4 is carbon tetrachloride

Preparation of RX • Free radical halogenation (Chapter 4) REVIEW • Free radical allylic halogenation – produces alkyl halide with double bond on the neighboring carbon. LATER =>

Substitution Reactions • The halogen atom on the alkyl halide is replaced with another group. • Since the halogen is more electronegative than carbon, the C-X bond breaks heterolytically and X- leaves. The group replacing X- is a nucleophile. =>
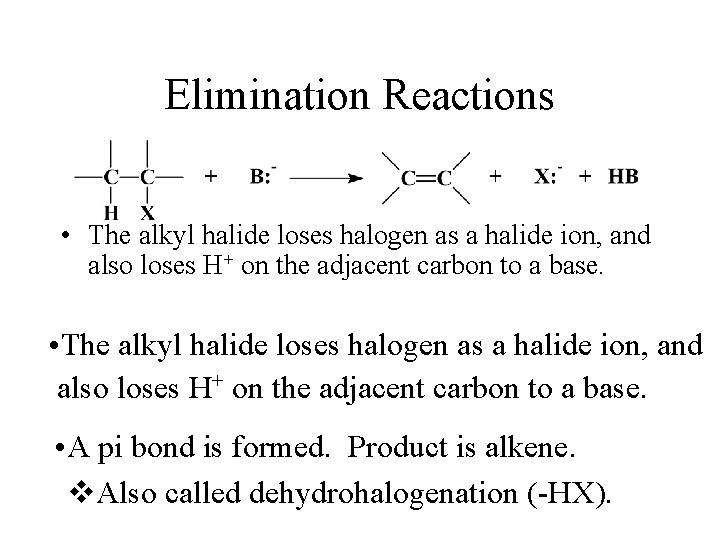
Elimination Reactions • The alkyl halide loses halogen as a halide ion, and also loses H+ on the adjacent carbon to a base. • A pi bond is formed. Product is alkene. v. Also called dehydrohalogenation (-HX).

Ingold

Ingold Sir Christopher Father of Physical Organic Chemistry Coined such names and symbols as: SN 1, SN 2, E 1, E 2, nucleophile, electrophile resonance effect, inductive effect/ In print, he often attacked enemies vigorously and sometimes in vitrolic manner.
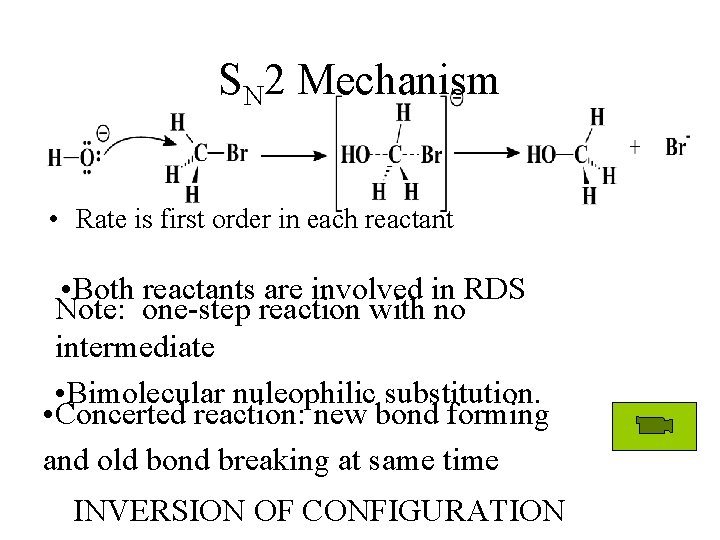
SN 2 Mechanism • Rate is first order in each reactant • Both reactants are involved in RDS Note: one-step reaction with no intermediate • Bimolecular nuleophilic substitution. • Concerted reaction: new bond forming and old bond breaking at same time INVERSION OF CONFIGURATION
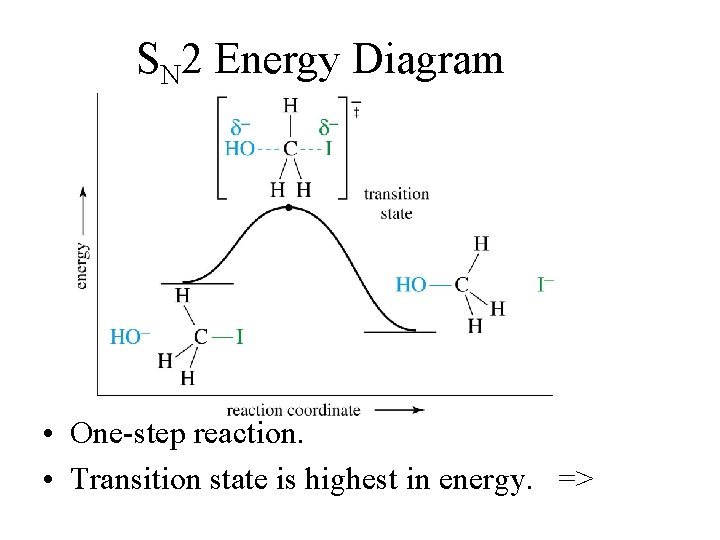
SN 2 Energy Diagram • One-step reaction. • Transition state is highest in energy. =>

Uses for SN 2 Reactions • Synthesis of other classes of compounds. • Halogen exchange reaction. =>

SN 2: Nucleophilic Strength • Stronger nucleophiles react faster. • Strong bases are strong nucleophiles, but not all strong nucleophiles are basic. =>

Trends in Nuc. Strength • Of a conjugate acid-base pair, the base is stronger: OH- > H 2 O, NH 2 - > NH 3 • Decreases left to right on Periodic Table. More electronegative atoms less likely to form new bond: OH- > F-, NH 3 > H 2 O ØIncreases down Periodic Table, as size and polarizability increase: I- > Br- > Cl-
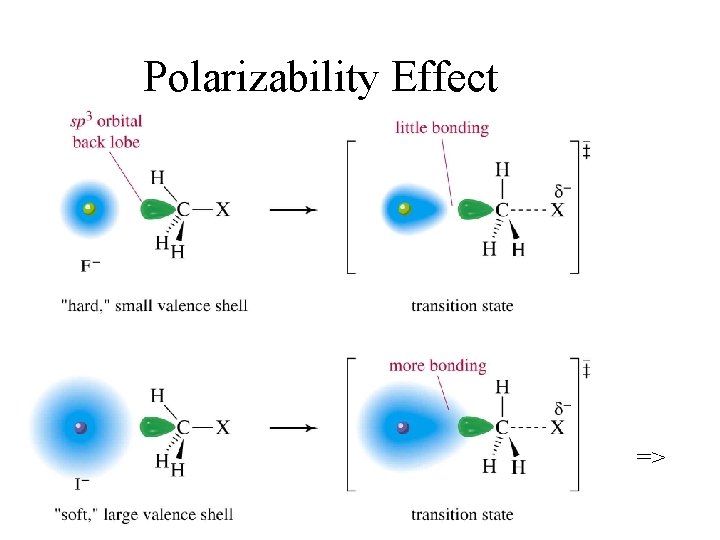
Polarizability Effect =>

Bulky Nucleophiles Sterically hindered for attack on carbon, so weaker nucleophiles. =>
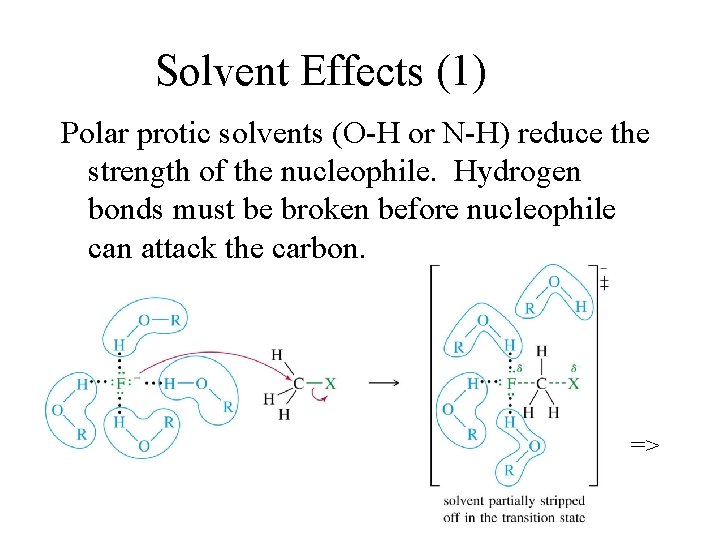
Solvent Effects (1) Polar protic solvents (O-H or N-H) reduce the strength of the nucleophile. Hydrogen bonds must be broken before nucleophile can attack the carbon. =>
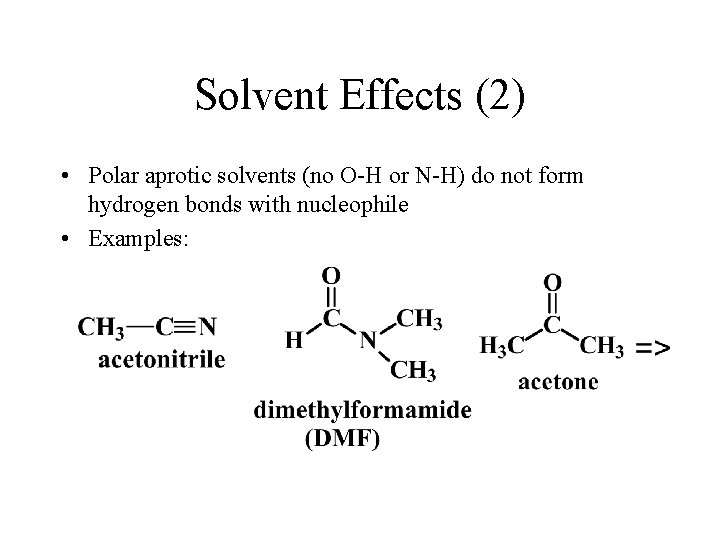
Solvent Effects (2) • Polar aprotic solvents (no O-H or N-H) do not form hydrogen bonds with nucleophile • Examples:
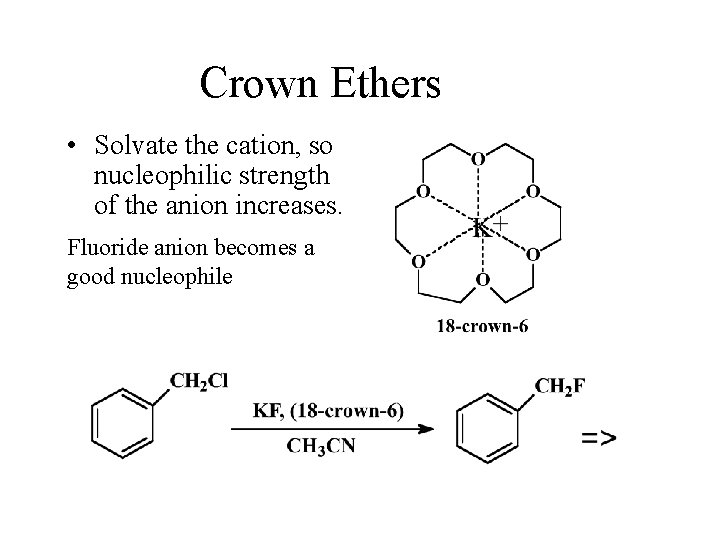
Crown Ethers • Solvate the cation, so nucleophilic strength of the anion increases. Fluoride anion becomes a good nucleophile

SN 2: Reactivity of Substrate ü Carbon must be partially positive. üMust have a good leaving group üCarbon must not be sterically hindered. =>
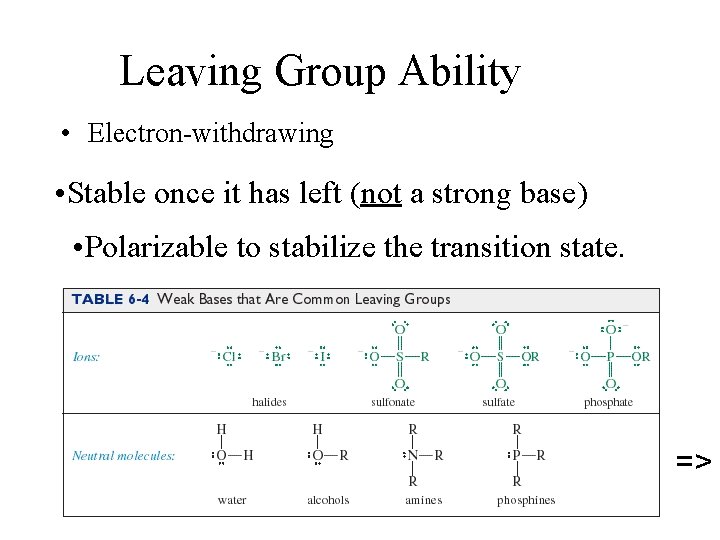
Leaving Group Ability • Electron-withdrawing • Stable once it has left (not a strong base) • Polarizable to stabilize the transition state. =>

Structure of Substrate • Relative rates for SN 2: CH 3 X > 1° > 2° >> 3° • Tertiary halides do not react via the SN 2 mechanism, due to steric hindrance. =>
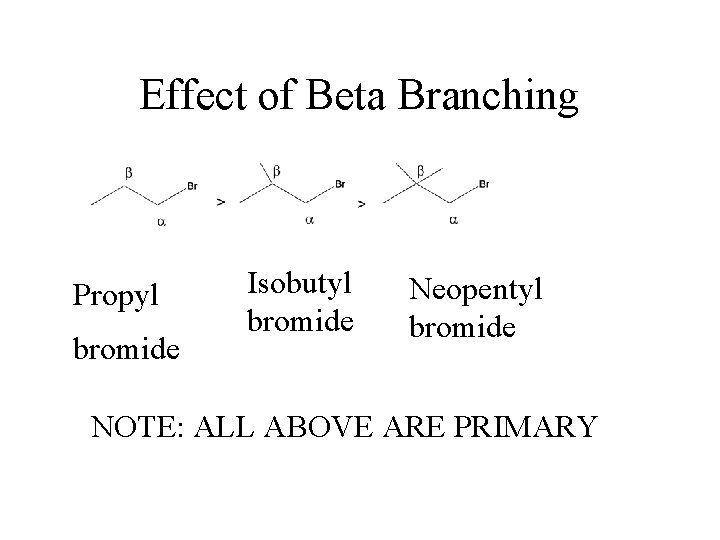
Effect of Beta Branching Propyl bromide Isobutyl bromide Neopentyl bromide NOTE: ALL ABOVE ARE PRIMARY

Miscellaneous Substrate All have sterically hindered backsides - No SN 2 reactivity
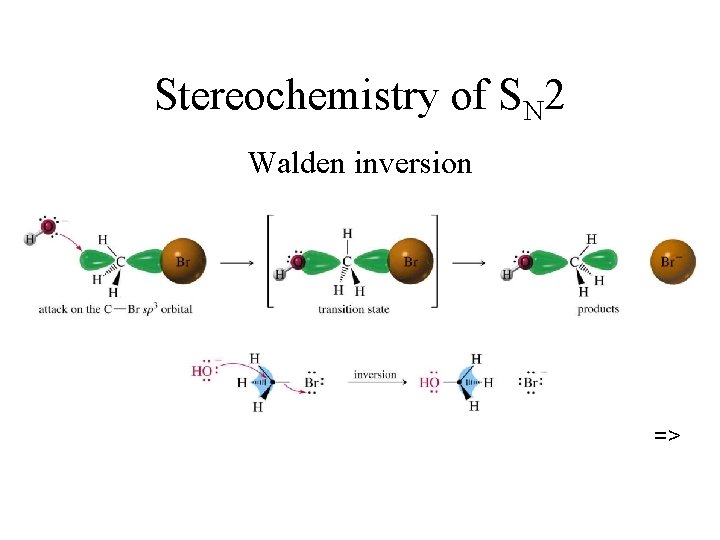
Stereochemistry of SN 2 Walden inversion =>
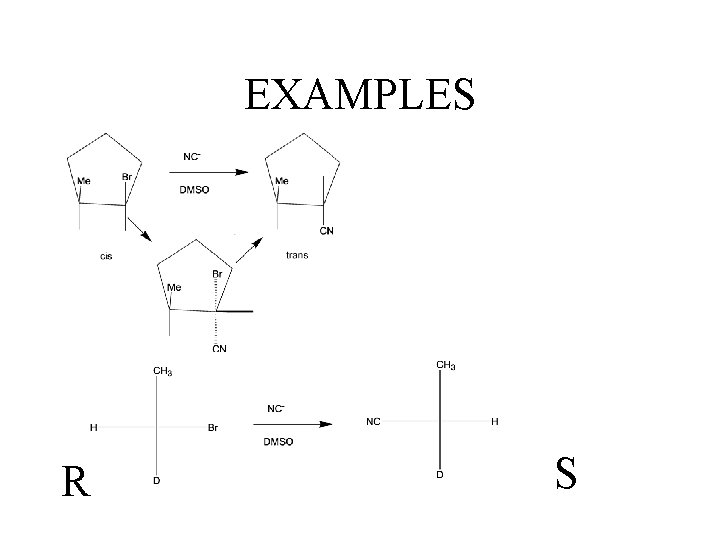
EXAMPLES R S
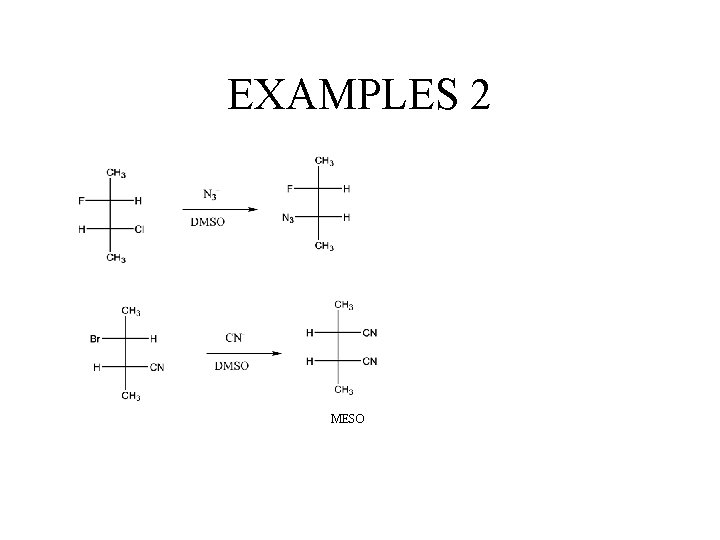
EXAMPLES 2 MESO
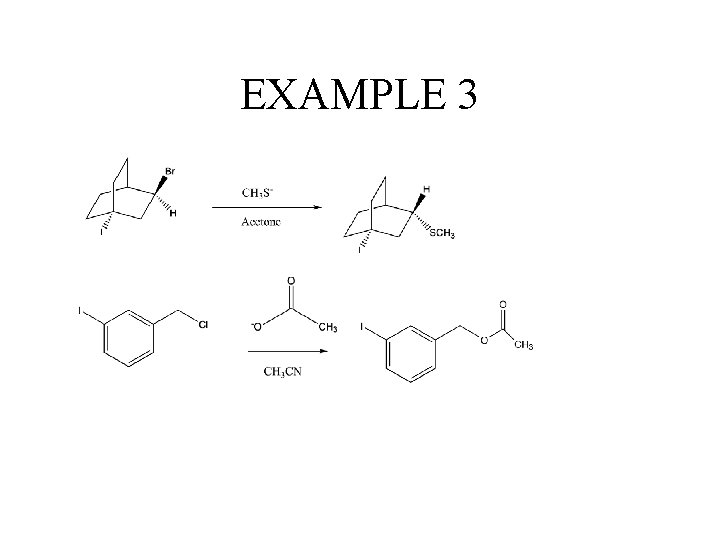
EXAMPLE 3

EXAMPLE 4 How would you prepare the following from an alkyl halide? retrosynthetic analysis

SN 1 Reaction • Unimolecular nucleophilic substitution. • Two step reaction with carbocation intermediate. • Rate is first order in the alkyl halide, zero order in the nucleophile. • Racemization occurs.
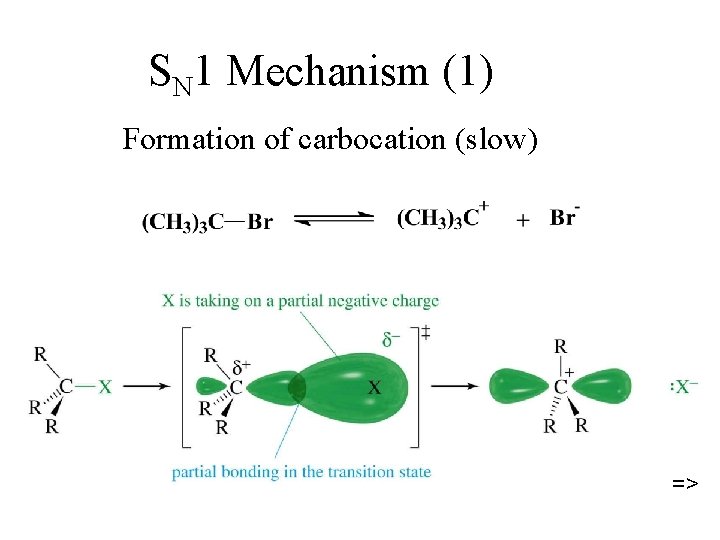
SN 1 Mechanism (1) Formation of carbocation (slow) =>
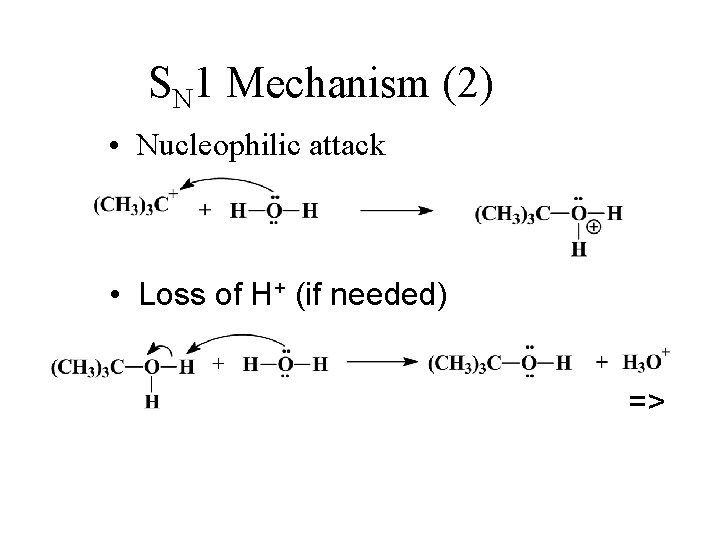
SN 1 Mechanism (2) • Nucleophilic attack • Loss of H+ (if needed) =>
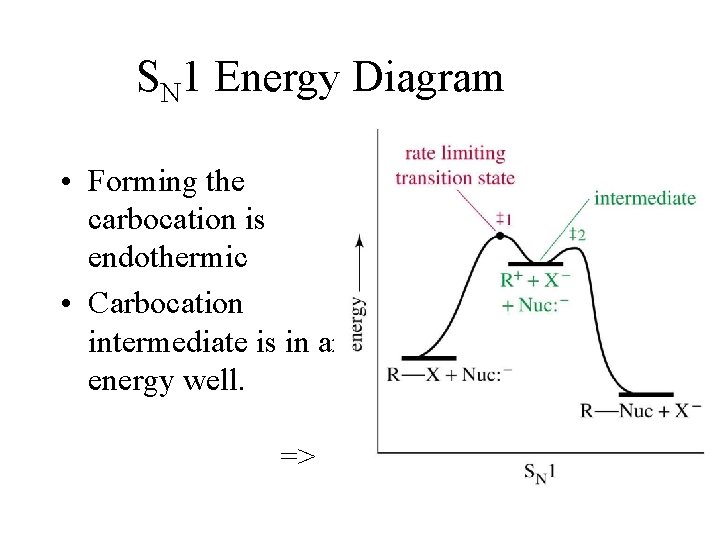
SN 1 Energy Diagram • Forming the carbocation is endothermic • Carbocation intermediate is in an energy well. =>

Rates of SN 1 Reactions • 3° > 2° > 1° >> CH 3 X –Order follows stability of carbocations (opposite to SN 2) –More stable ion requires less energy to form • Better leaving group, faster reaction (like SN 2) Polar protic solvent best: It solvates ions strongly with hydrogen bonding
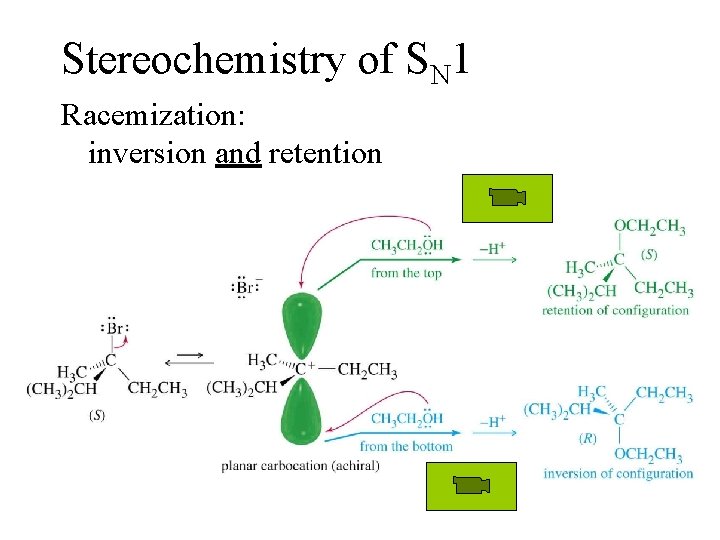
Stereochemistry of SN 1 Racemization: inversion and retention =>

Rearrangements • Carbocations can rearrange to form a more stable carbocation. Hydride shift: H- on adjacent carbon bonds with C+. • Methyl shift: CH 3 - moves from adjacent carbon if no H’s are available.
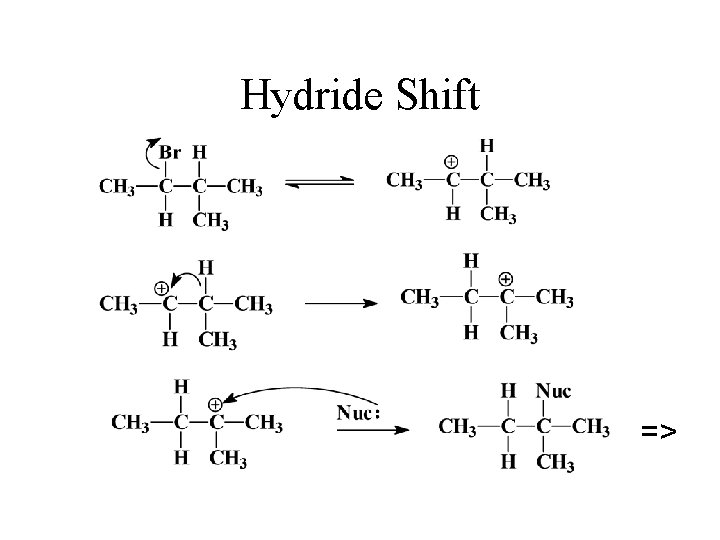
Hydride Shift =>
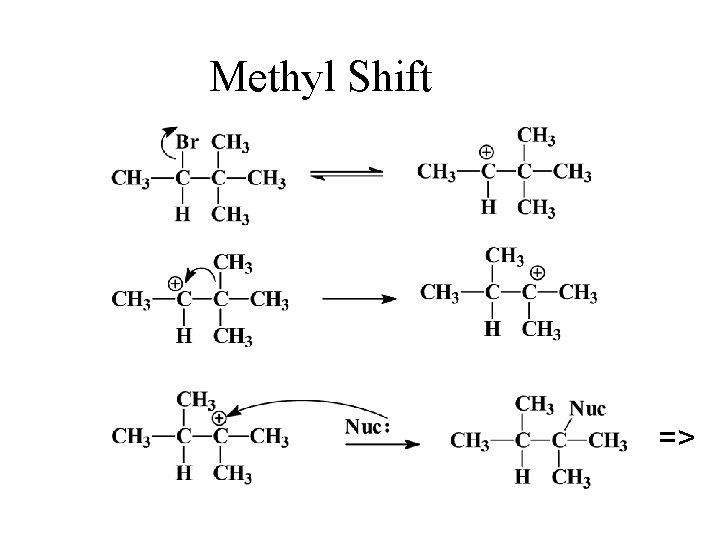
Methyl Shift =>
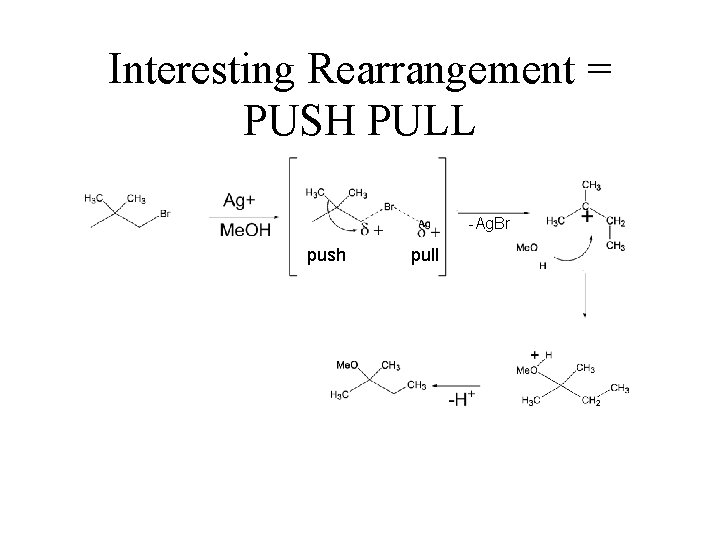
Interesting Rearrangement = PUSH PULL -Ag. Br push pull

S N 2 or SN 1? • Primary or methyl • Tertiary • Strong nucleophile • Weak nucleophile (may also be solvent) • Polar protic solvent, Ag+ • Polar aprotic solvent • Rate = k[halide][Nuc] • Rate = k[halide] • Inversion at chiral carbon • Racemization of optically active compound • No rearrangements ØRearranged products

E 1 Reaction • Unimolecular elimination • Two groups lost (usually X- and H+) • Nucleophile acts as base) v. Also have SN 1 products (mixture SN 1 and E 1 have common first step.
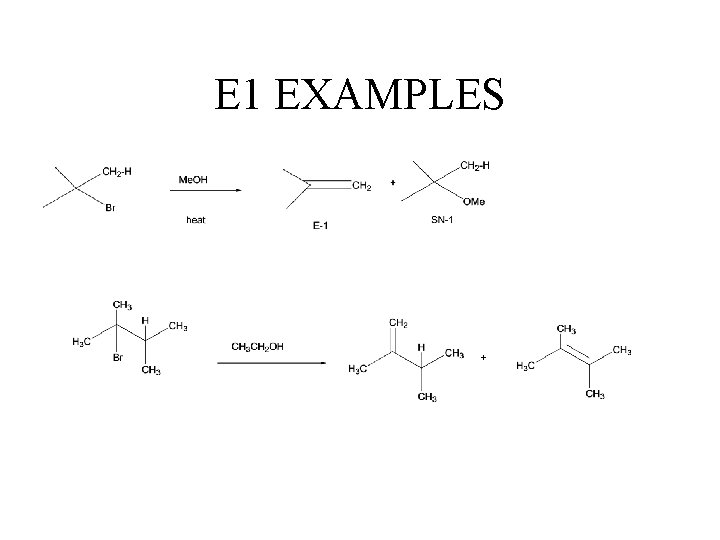
E 1 EXAMPLES +
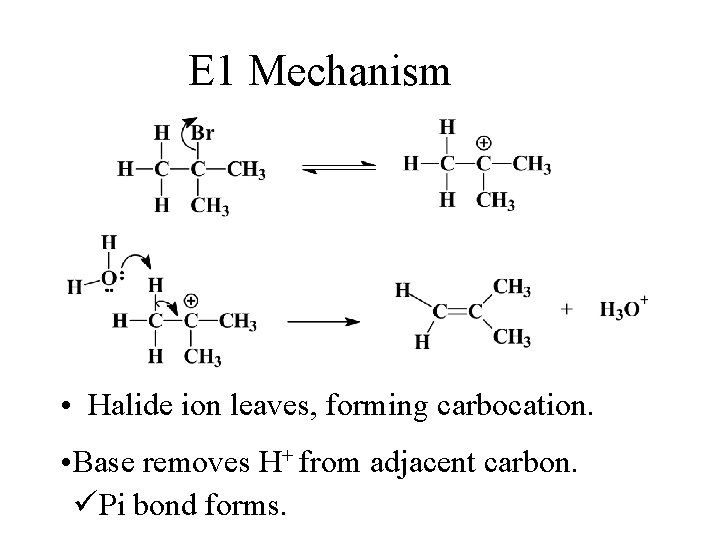
E 1 Mechanism • Halide ion leaves, forming carbocation. • Base removes H+ from adjacent carbon. üPi bond forms.
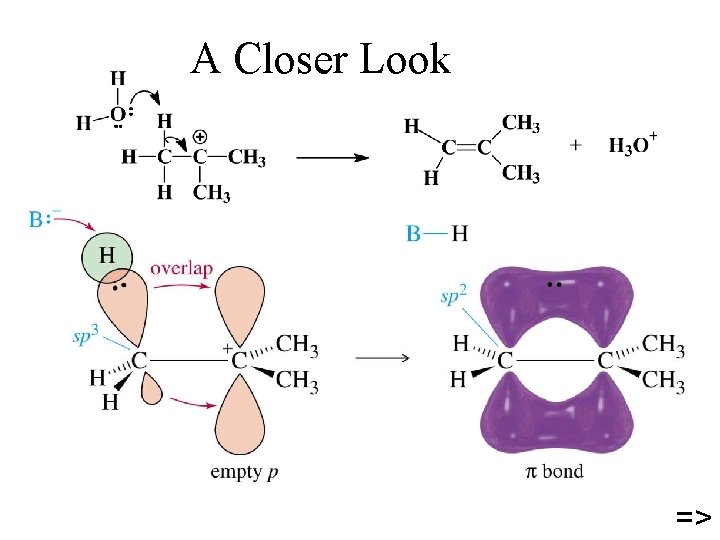
A Closer Look =>
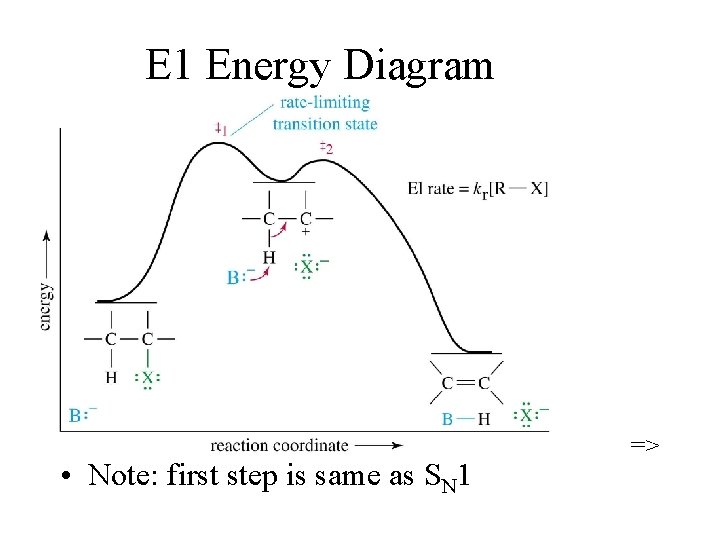
E 1 Energy Diagram • Note: first step is same as SN 1 =>

E 2 Reaction • Bimolecular elimination • Requires a strong base • Halide leaving and proton abstraction happens simultaneously - no intermediate

E 2 Examples
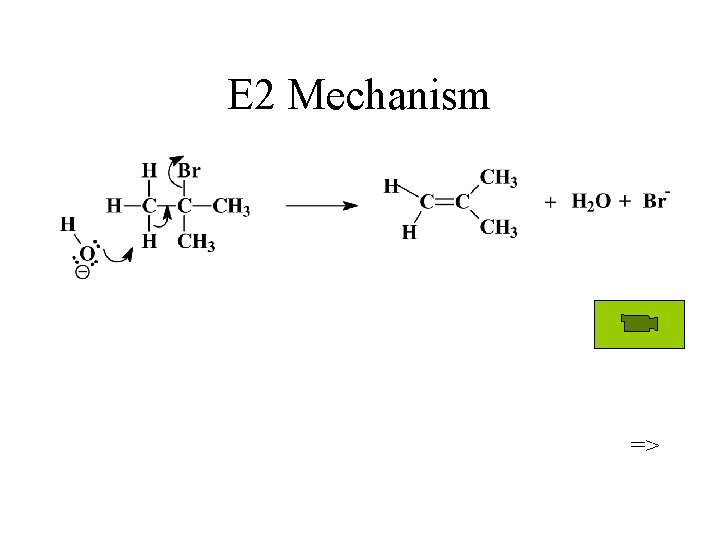
E 2 Mechanism =>
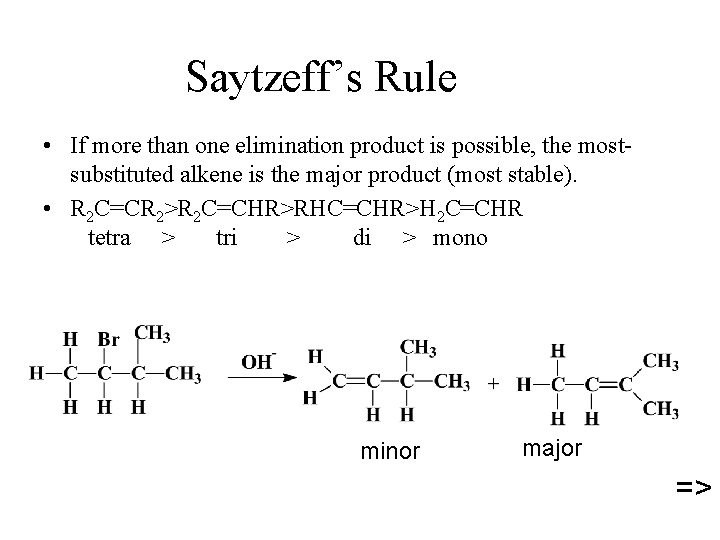
Saytzeff’s Rule • If more than one elimination product is possible, the mostsubstituted alkene is the major product (most stable). • R 2 C=CR 2>R 2 C=CHR>RHC=CHR>H 2 C=CHR tetra > tri > di > mono minor major =>
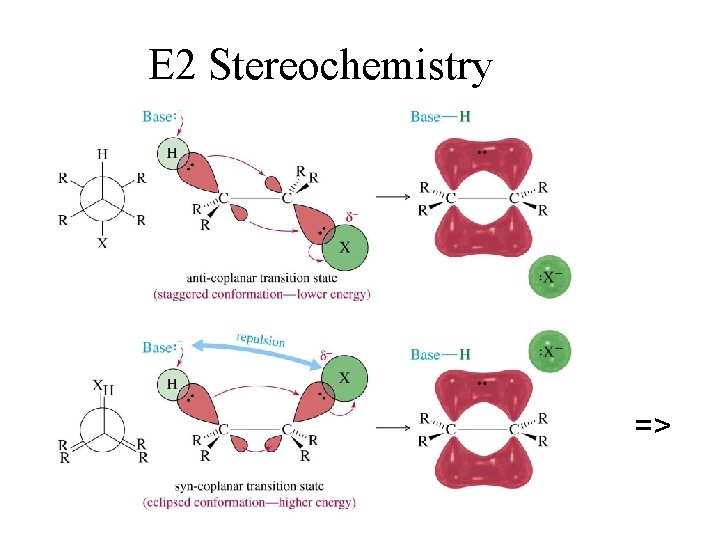
E 2 Stereochemistry =>

E 1 or • Tertiary > Secondary • Weak base • Good ionizing solvent • Rate = k[halide] • Saytzeff product • No required geometry • Rearranged products E 2? • • • Tertiary > Secondary Strong base required Solvent polarity not important Rate = k[halide][base] Saytzeff product Coplanar leaving groups (usually anti) • No rearrangements =>

End of Chapter 6
- Slides: 63