Lecture 4 Ionic and Molecular Compounds Course Lecturer



















































- Slides: 65

Lecture 4: Ionic and Molecular Compounds Course Lecturer: Assist. Prof. Dr. Altijana Hromić-Jahjefendić

�Book chapter: 6 �Pages: 165 -211

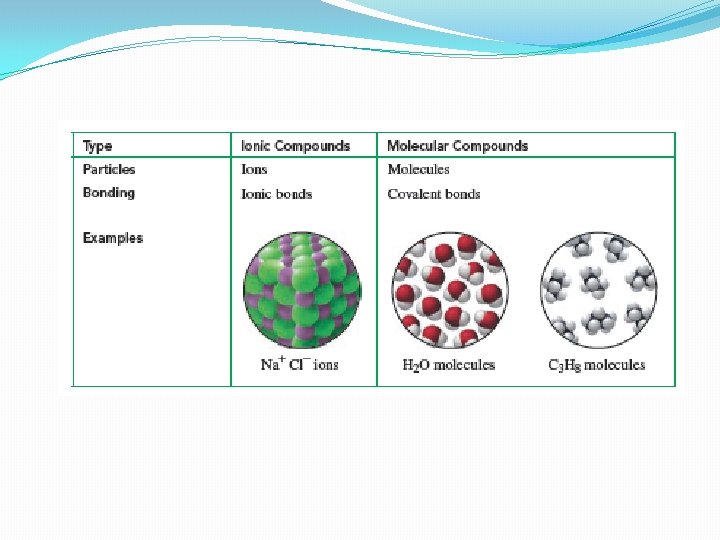
�In nature almost all elements are found in combination �Only noble gases do not combine (very stable) �A compound is composed of two or more elements �Can be ionic and molecular

Ionic compound �One or more electrons are transferred from metals to nonmetals �Forms positive and negative ions �The attraction between these ions is ionic bond q. Everyday examples: kitchen salt Na. Cl, baking soda Na. HCO 3, mineral supplements etc. �Precious and semiprecious gemstones (minerals) are also ionic compounds �Sapphire and rubies are made of Al 2 O 3 with impurities

Molecular compounds �Consists of two or more nonmetals that share one or more valence electrons �Resulting molecules are held by covalent bonds q. Examples: water H 2 O, carbon dioxide CO 2, alcohol, antibiotics etc.

Ions: Transfer of Electrons �Compounds form when electrons are transferred or shared to give stable electron arrangement to the atoms! �Ionic or covalent bond �Gain, lose or share valence electrons to acquire an octet (8 valence electrons) �Tendency to be in stable electron arrangement is octet rule


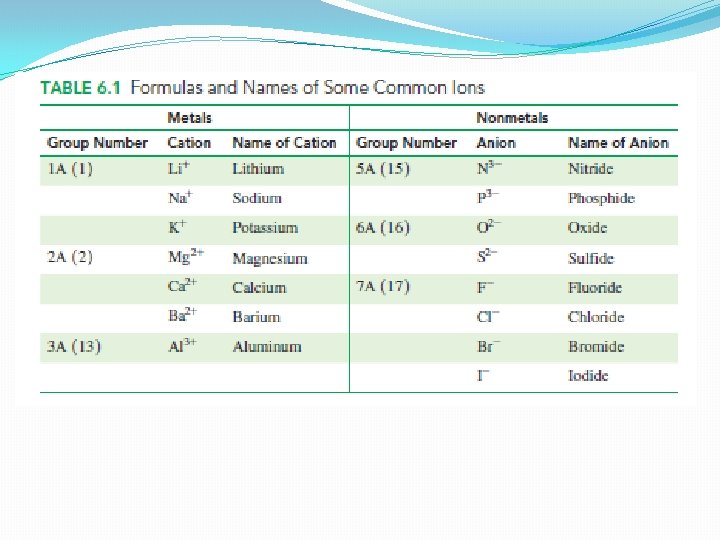
Positive Ions: Loss of Electrons �Ions – electrical charges �Form when atoms lose or gain electrons �Metals �Positively charged ions of metals – cations �A metal ion is named by its element name �Examples: Na+, Mg 2+, etc.

Negative Ions: Gain of Electrons �By gaining electrons, a nonmetal atom forms a negatively charged ion �Anion �The name is formed by using the first syllable of its element name followed by ide q. Example: Chloride


Ionic Charges from Group Numbers �Using group numbers in the periodic table to determine the charges for the ions of the representative elements �Group 1 A lose one electron to form ions with 1+ charge q. Example: Na+ �Group 2 A lose two electrons to form ions with 2+ charge q. Example: Mg 2+ �Group 3 A lose 3 electrons to form ions with 3+ charge �Example: Al 3+

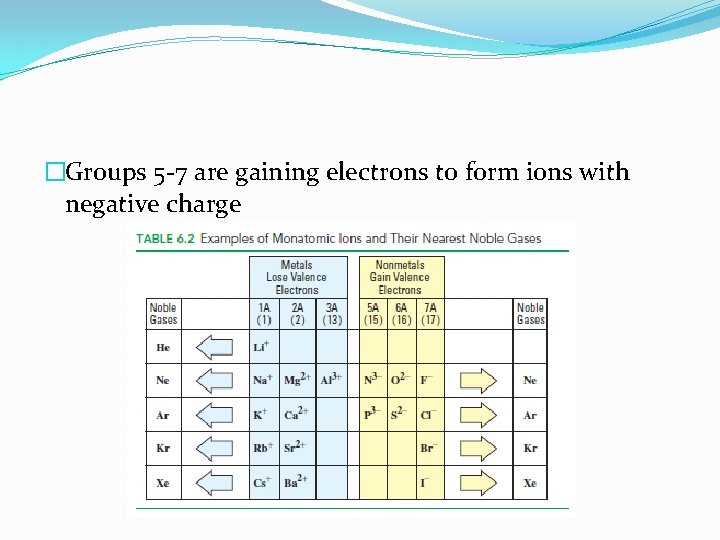
�Groups 5 -7 are gaining electrons to form ions with negative charge

Three ways to find the valence electrons: http: //www. wikihow. com/Find-Valence-Electrons

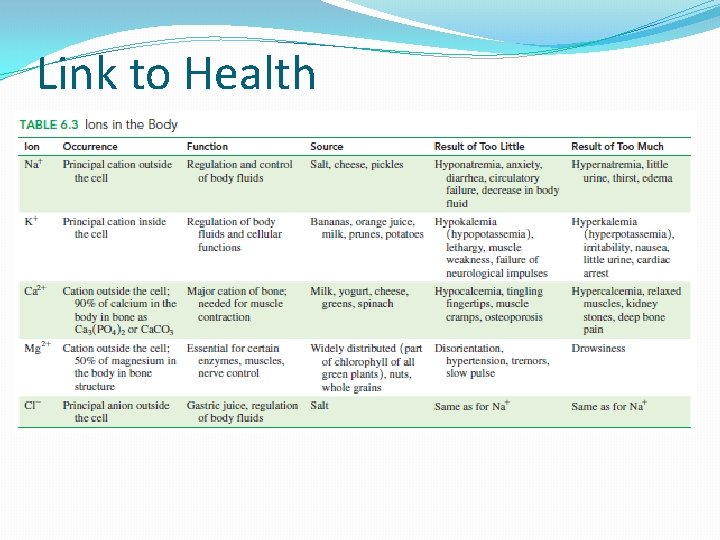
Link to Health �Some ions are important in the body �Physiological and metabolic function �Food provides body with ions important in regulating body functions

Link to Health

Ionic compounds - Review �Consist of positive and negative ions �Held together by ionic bons �Positive ions – formed by metals �Negative ions – formed by nonmetals

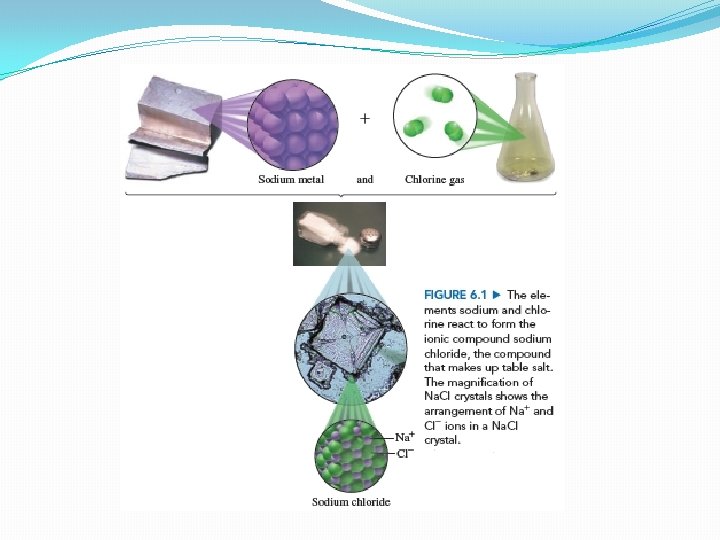
Properties of ionic compounds �Properties of an compound are very different from those of the original elements q. Example: Na. Cl Ø Sodium (Na) is soft, shiny, very reactive metal Ø Chlorine (Cl) is yellow-green poisonous gas Ø Upon reaction they produce Na. Cl – ordinary table salt Ø Hard, white, crystalline substance


�Cl- ions are larger �Na+ ions are smaller �Arranged in 3 D structure in which Na+ ions occupy the space between the Cl- ions �Every Na+ ion is surrounded by six Cl- ions and vice versa �Many strong attractions between the positive and negative ions �Accounts for the high melting points

Chemical formulas of ionic compounds �The chemical formula of a compound represents the symbols and subscripts in the lowest whole-number ratio of the atoms or ions. �Thus, the total amount of positive charge is equal to the total amount of negative charge. �Na atom loses its one valence electron to form Na+, and one Cl atom gains one electron to form a Cl- ion �Formula indicates that the compound has charge balance

Subscripts in Formulas �Example: Mg and Cl �Mg loses 2 electrons (Mg 2+) �Two Cl atoms gain one electron to form two Cl- ions �Two Cl- ions are needed to balance positive charge of Mg 2+ �Gives formula Mg. Cl 2 �Subscript 2 indicates that 2 Cl- ions are needed for charge balance

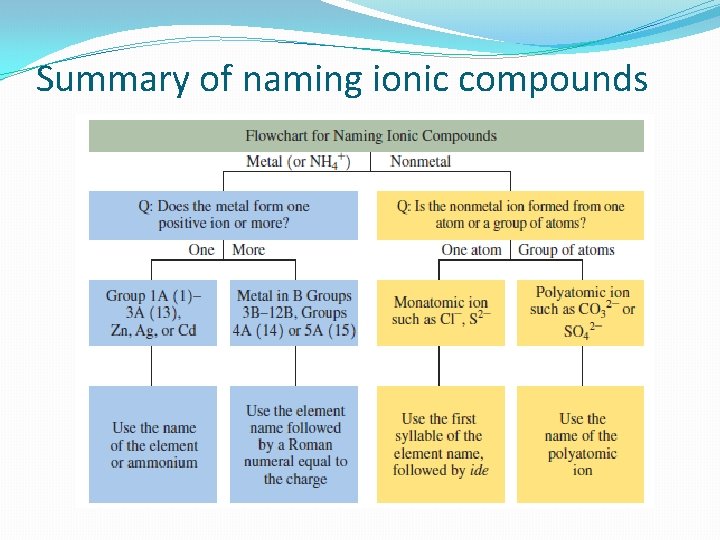
Naming Ionic Compounds �Name of metal ion is the same as its element name �The name of nonmetal ion is first syllable + ide �Subscripts are not used, they are understood

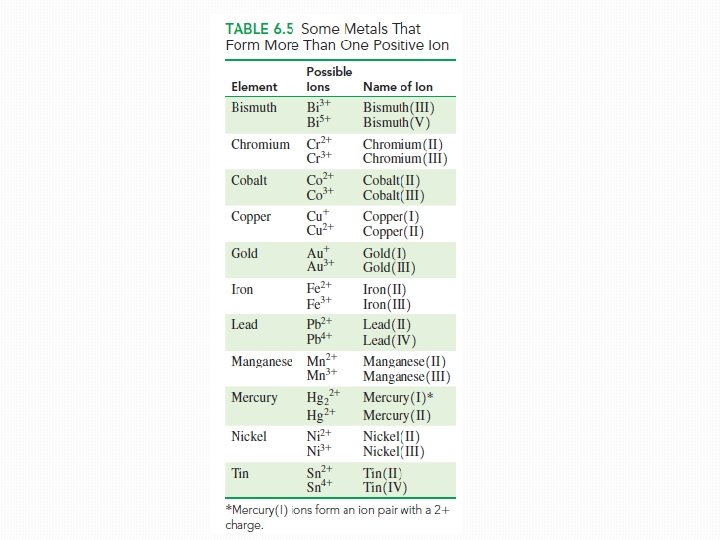
Metals with Variable charges �Charge detrmined from its group number �Cannot determine the charge of a transition element �Typically forms two or more positive ions �Variable charges �Then we cannot predict the ionic charge from a group number �For metals forming two or more ions – naming system


Determination of Variable Charges �You need to determine if the metal is a representative or a transition element �If transition element (except Zn, Cd and Ag) we need to use its ionic charge �The calculation of ionic charge depends on the negative charge of the anions in the formula q Example: Cu. Cl 2 �Two chloride ions (each with 1 - charge; total negative charge is 2 -) �To balance this 2 - charge the copper ion must have a charge of 2+

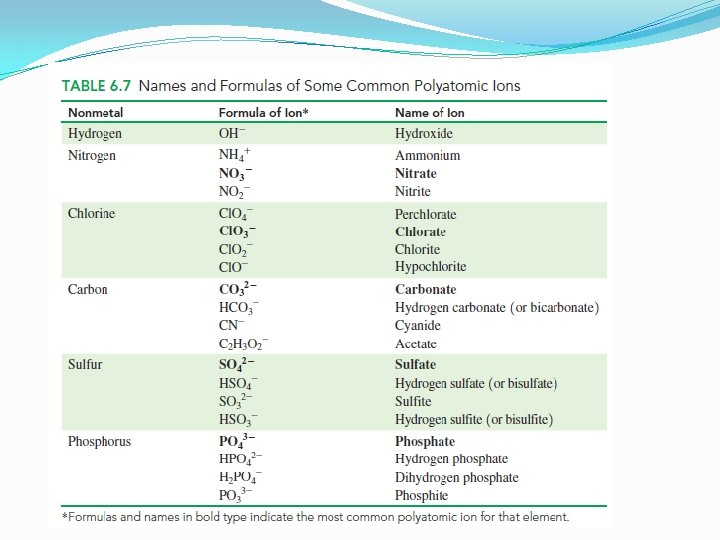
Polyatomic ions � An ionic compound may also contain a polyatomic ion as one of its cations or anions. � A polyatomic ion is a group of covalently bonded atoms that has an overall ionic charge. � Most polyatomic ions consist of a nonmetal such as phosphorus, sulfur, carbon, or nitrogen covalently bonded to oxygen atoms. � Almost all the polyatomic ions are anions with charges of 1 -, 2 -, or 3 -. Only one common polyatomic ion, NH 4 +, has a positive charge. .

Names of Polyatomic Ions �Names of most of them end in ate (nitrate, sulfate) �When a related ion has one less oxygen atom – ending ite (nitrite, sulfite) �Hydroxide ion (OH-) and cyanide ion (CN-) are exceptions to this naming pattern


Writing formulas for compounds containing polyatomic ions �No polyatomic ion exists by itself �It must be associated with ions of opposite charge �To write correct formula – same rules as for writing simple ionic compounds �The total negative and positive charge must equal zero

Naming Ionic Compounds Containing Polyatomic Ions �First we write the positive ion (usually a metal) �Then we write the name for the polyatomic ion �No prefixes are used

Summary of naming ionic compounds

Summary �Ions �Octet/ octet rule �Cations �Anions �Ionic charges from group numbers �Properties of ionic compounds �Chemical formulas of ionic compounds �Naming ionic compounds �Variable charges �Polyatomic ions

Molecular compounds �Contains two or more nonmetals that form covalent bons �Valence electrons are shared – covalent bond �Atoms sharing electrons – form a molecule

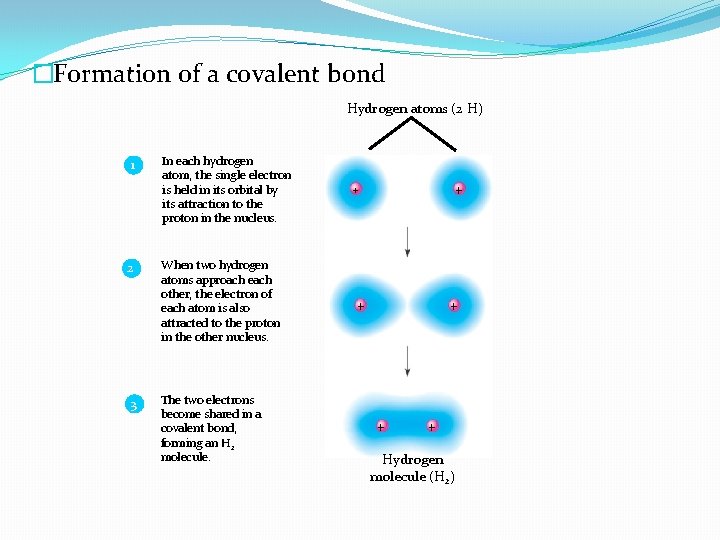
�Formation of a covalent bond Hydrogen atoms (2 H) 1 2 3 In each hydrogen atom, the single electron is held in its orbital by its attraction to the proton in the nucleus. When two hydrogen atoms approach each other, the electron of each atom is also attracted to the proton in the other nucleus. The two electrons become shared in a covalent bond, forming an H 2 molecule. + + + Hydrogen molecule (H 2)

Covalent bond �Sharing a pair of valence electrons by two atoms �Two or more atoms held together by covalent bonds constitute a molecule �Occurs in non-metals �Can be single, double and triple bond

Difference between single, double and triple bond �Single bond= a pair of shared electrons (hydrogen H 2) �Double bond= sharing a two pairs of valence electrons (oxygen O 2) �Triple bond= sharing three pairs of valence electrons (nitrogen)

Names and formulas of Molecular Compounds �The first nonmetal in the formula is named by its element name �Second nonmetal is named using first syllable of its element name + ide �When a subscript indicates two or more atoms of an element, a prefix is shown in front of its name

�Prefixes are needed �Different compounds can be formed from the same two nonmetals �Example: carbon and oxygen �Carbon monoxide (CO) and carbon dioxide (CO 2) �Prefix indicates the number of oxygen atoms

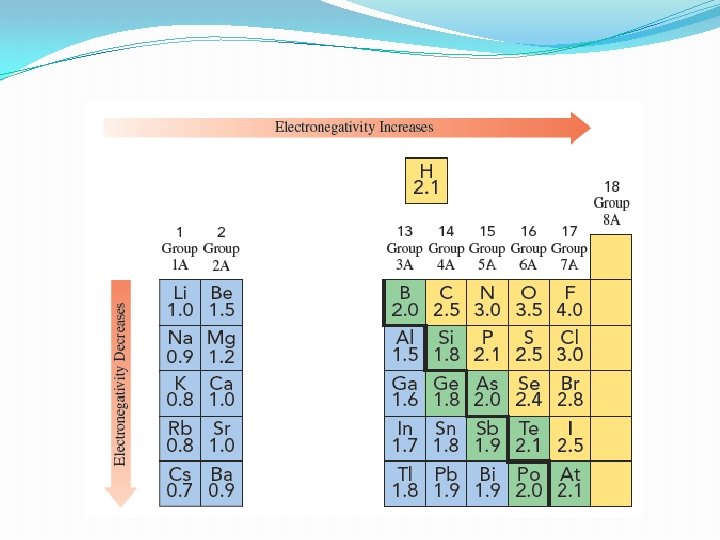
Electronegativity and Bond Polarity �Electronegativity- the attraction of a particular kind of atom for the electrons in a covalent bond �The more electronegative an atom the more strongly it pulls shared electrons toward itself �Covalent bond between two atoms of the same element are equally electronegative


�In some cases, more electronegative atoms strip electrons away from their bonding partners �Typical example is Na and Cl �Na has 1 electron in valence shell and Cl has 7 �Lone valence electron of Na is transferred to the chlorine atom and both atoms have their valence shell completed

�Bonding capacity corresponds to the number of covalent bonds thé atom can form �Also called valence �Equal to the number of unpaired electrons in the valence shell

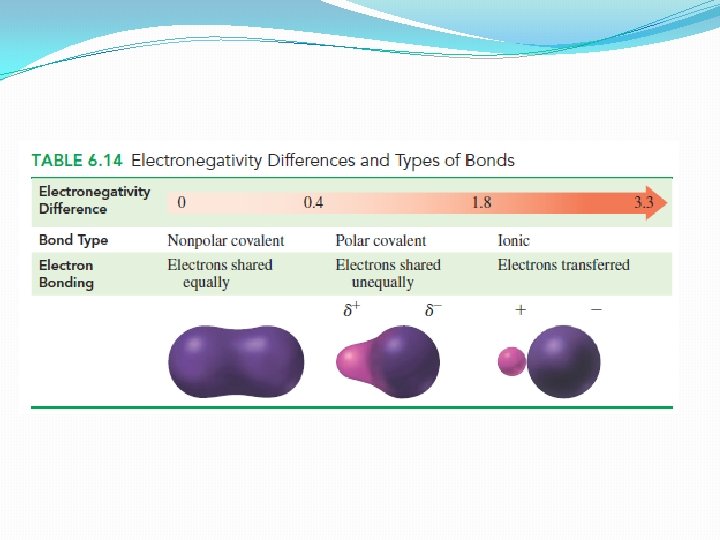
Polarity of Bonds �The difference in electronegativity values of two atoms can be used to predict the type of bond � 2 types: 1. polar covalent bond 2. non-polar covalent bond

�In a polar covalent bond �The atoms have different electronegativities �Share the electrons unequally �Vary in polarity

�In a nonpolar covalent bond �The atoms have same electronegativities �Share the electron equally �H 2 or O 2

Dipoles and Bond Polarity �The polarity of a bond depends on the difference in the electronegativity values of its atoms �In polar bond – shared electrons are attracted to the more electronegative atom; makes them partially negative �Atom with the lower electronegativity becomes partially positive; lack of the electrons at that atom

�A bond becomes more polar as the electronegativity difference increases �Polar covalent bond with separation of charges is called a dipole �The positive and negative ends of the dipole are indicated by the lowercase Greek letter delta with a positive or negative sign, δ+ and δ-


Shapes of Molecules �Using electron-dot formula we can predict 3 D shapes of many molecules �Shape is important to understand of how molecules interact with enzymes or certain antibiotics �Identify the number of electron groups around central atom �Counting lone pairs of electrons and bonds as one electron group �Once we know the number of electron groups we can determine its specific shape

Lewis representation of electrons

Central Atoms with Two Electron groups �CO 2 molecule �Minimal repulsion occurs when two electron groups are on opposite sides of the central C atom �Linear electron-group geometry �Bond angle of 180°

Central Atoms with Three Electron groups �H 2 CO �Central carbon attached to two H atoms by single bond and to the O by a double bond �Minimal repulsion – three groups are far apart as possible around the central C atom �Geometry: trigonal planar; 120°

Central Atoms with Four Electron groups �CH 4 (methane) �Central C bonded to four H atoms �Minimal repulsion is tetrahedral; bond angles 109°

Polarity of molecules � 2 types of covalent bonds � 2 types of molecule polarities: 1. polar 2. nonpolar

Nonpolar molecules �All the bonds are nonpolar �Or the polar bonds cancel each other out because of symmetrical arrangement �H 2, Cl 2, CH 4 contain only nonpolar bonds �CO 2 is linear, has two equal polar covalent bonds whose dipoles point in opposite directions �Dipoles cancel out making CO 2 nonpolar molecule

Polar molecules �One end of the molecule is more negatively charged than the other end �Dipoles from the individual polar bonds do not cancel each other �HCl – has one covalent bond that is polar so the molecule is polar

�Molecules with two or more electron-groups the shape determines whether or not the dipoles cancel �H 2 O has a bent shape �It is polar because the individual dipoles do not cancel �In CH 3 F, the C-F bond is polar covalent � 3 C-H bonds are nonpolar covalent �Because there is only one dipole which does not cancel, this is polar molecule

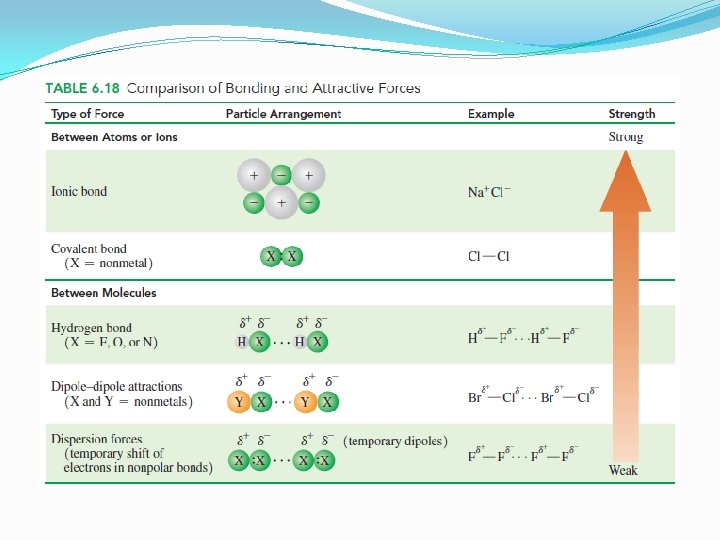
Attractive forces in compounds �In gases the interaction between particles is minimal �In solids and liquids there are sufficient interactions between particles to hold them close together Ø Dipole-dipole attractions Ø Hydrogen bonding Ø Dispersion forces

Dipole-dipole Attractions �Between the positive end of one molecule and negative end of another �Partially positive atom of one molecule attracts partially negative atom in another molecule �HCl

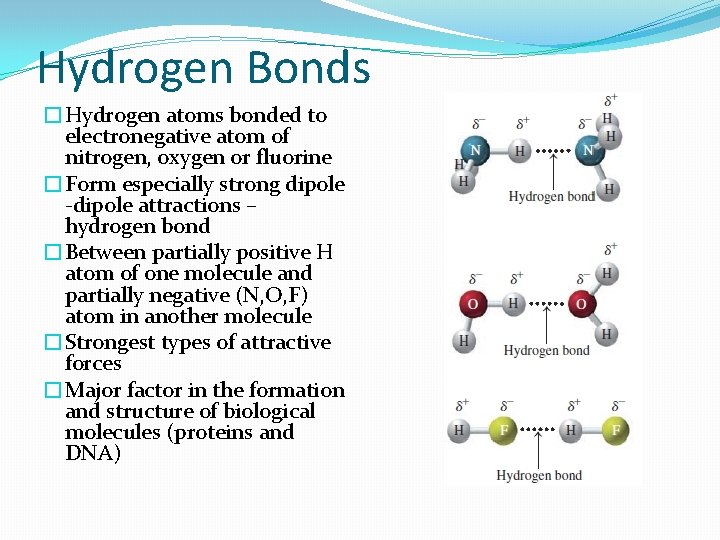
Hydrogen Bonds �Hydrogen atoms bonded to electronegative atom of nitrogen, oxygen or fluorine �Form especially strong dipole -dipole attractions – hydrogen bond �Between partially positive H atom of one molecule and partially negative (N, O, F) atom in another molecule �Strongest types of attractive forces �Major factor in the formation and structure of biological molecules (proteins and DNA)

Dispersion Forces �Very weak attractions between nonpolar molecules �Make it possible for nonpolar molecules to form liquids and solids �Electrons are distributed symmetrically �However, they can accumulate more in one part of the molecule than another – forms temporary dipole


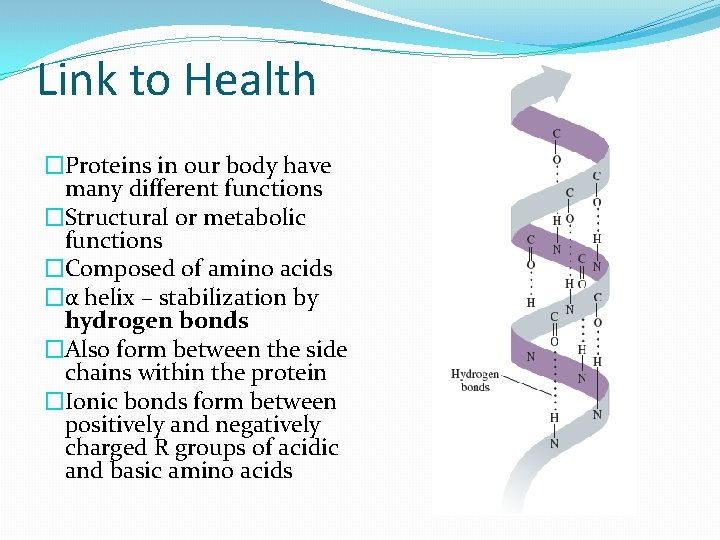
Link to Health �Proteins in our body have many different functions �Structural or metabolic functions �Composed of amino acids �α helix – stabilization by hydrogen bonds �Also form between the side chains within the protein �Ionic bonds form between positively and negatively charged R groups of acidic and basic amino acids

Summary �Molecular compounds �Covalent bond �Electronegativity and bond polarity �Polar vs. non-polar �Dipoles �Shape of molecules �Attraction forces

�Book pages: 165 -211 �Homework 2: Describe attractive forces in compounds! �Send the homework per e-mail till 4. 11. 2018 �Bring the hard copy next time to the lecture (6. 11. 2018) �Plagiarism!!!!! No copy-paste from the book or any internet sources!!!!! Use your own sentences!!!!! �Otherwise you loose points!!!!!