Covalent Bonding Notes Ionic vs Covalent Bonding Ionic

Covalent Bonding Notes

Ionic vs Covalent Bonding • Ionic: electron(s) leave one atom & gained by another atom to satisfy both atoms’ octets, this results in the formation of ions. The resulting opposite charges attract each other. • Covalent: electrons are shared by two or more atoms to satisfy their octets.

How can you tell if a bond is IONIC or COVALENT? • Subtract the two electronegativity values (look at an electronegativity chart p. 263). >1. 7 to 4. 0: Ionic >0. 4 to 1. 7: Polar Covalent 0. 0 to 0. 4: Non-Polar Covalent • Electronegativity: atom’s ability to attract electrons in a chemical bond. (higher electronegativity means the atom wants electrons more)

How can you tell if a bond is IONIC or COVALENT? • Easy way: All metals = metallic bond Nonmetals and Metals = ionic bond All nonmetals = covalent bond

Yes, there are 2 kinds of covalent bonds! • Polar covalent: the electrons are shared, but one atom is pulling on the electrons a lot more. The electrons spend more time around that atom. • Nonpolar covalent: the electrons are evenly shared between the two atoms.
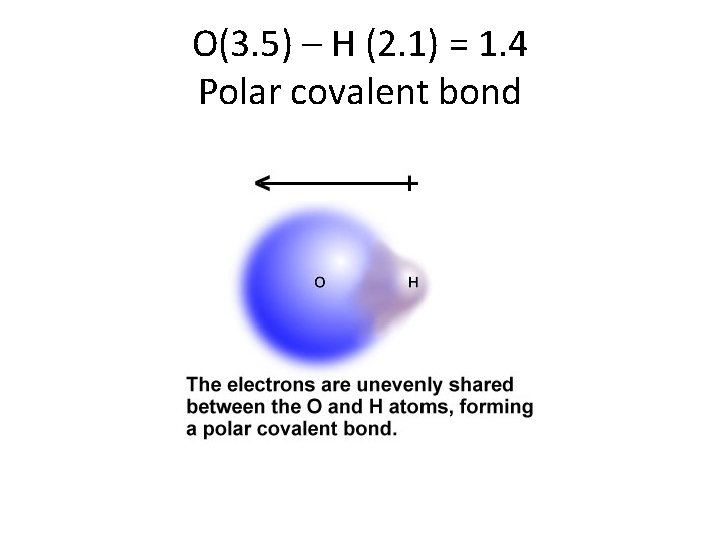
O(3. 5) – H (2. 1) = 1. 4 Polar covalent bond

• The closer the elements are on the periodic table, their electronegativities are more similar… more likely to form covalent bonds • Farther away… greater difference in electronegativity… more likely to form ionic bonds. • Metal + nonmetal = usually ionic • Nonmetal + nonmetal = usually covalent.

Properties of Covalent Compounds 1) Covalent compounds generally have much lower melting and boiling points than ionic compounds. 2) Covalent compounds are soft and squishy (compared to ionic compounds, anyway).

Properties, Cont’d • 3) Covalent compounds tend to be more flammable than ionic compounds. – There are exceptions to this rule! • 4) Covalent compounds don't conduct electricity in water.

Bond Dissociation Energy • Bond dissociation energy = energy required to break a covalent bond. Highest bond energy Hardest to break Triple Bond Shortest bond length Strongest Lowest bond energy Easiest to break Double Bond Single Bond Longest bond length Weakest

How to Name Covalent Compounds • 1. Name the first element (normal name) • 2. Name the second element (root name + “-ide”) • 3. Add prefixes to indicate how many atoms of each element are in the molecule (Subscripts) • Exception: no need to use “mono” for first element. Example: CO 2 = carbon dioxide
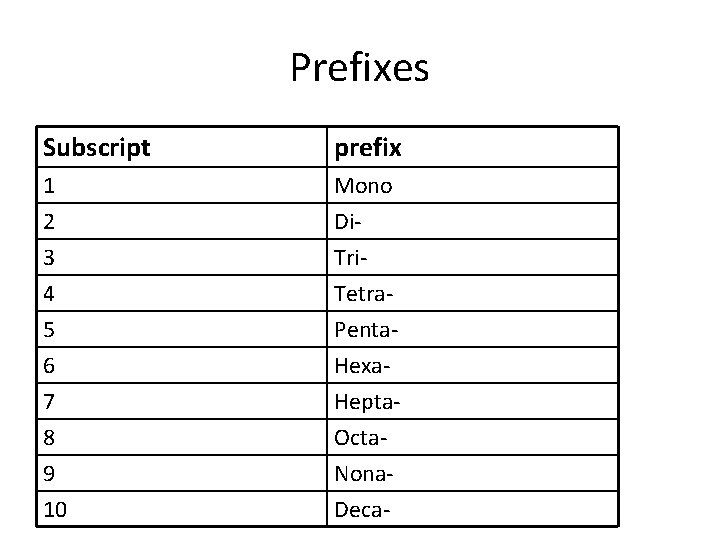
Prefixes Subscript prefix 1 2 3 Mono Di. Tri- 4 5 6 7 8 9 10 Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca-

Memorize these exceptions Chemical formula name H 2 O water CH 4 Methane NH 3 ammonia
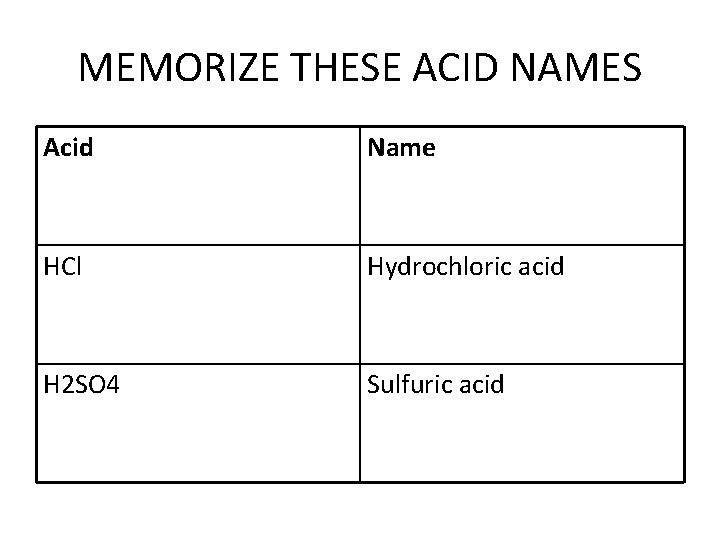
MEMORIZE THESE ACID NAMES Acid Name HCl Hydrochloric acid H 2 SO 4 Sulfuric acid

Diatomics • Diatomic elements are elements that do not exist singularly in nature because they are highly reactive. “Which elements are the diatomics? ” “HON, it’s the halogens!” • H 2, O 2, N 2, F 2, Cl 2, Br 2, I 2
- Slides: 15