Chemical Nomenclature 1 Binary Ionic Compounds Type I

Chemical Nomenclature 1. Binary Ionic Compounds - Type I 2. Binary Ionic Compounds - Type II 3. Ionic Compounds & Polyatomic (Complex) Ions 4. Hydrated Ionic Compounds 5. Binary Covalent Compounds 6. Binary Acids 7. Oxy-Acids

Definitions n n n An IONIC COMPOUND consists of a metal cation bonded to a nonmetal anion. Electrostatic attraction holds them together. A COVALENT COMPOUND consists of two nonmetal atoms sharing valence electrons. A BINARY compound is one that is made of just two elements. There can be one of each element such as in Na. Cl or KF. There can also be several of each element such as Na 2 O or Al. Br 3.

Type I Binary Ionic Compounds n The metal cations in these compounds have only ONE possible charge. Na+ sodium Zn 2+ zinc Al 3+ aluminum Ca 2+ calcium The charges are memorized or predicted using a periodic table! n The cations are bonded to nonmetal anions: O 2 oxide N 3 nitride Ffluoride Br bromide Notice that simple anions are always named with the suffix “ide”
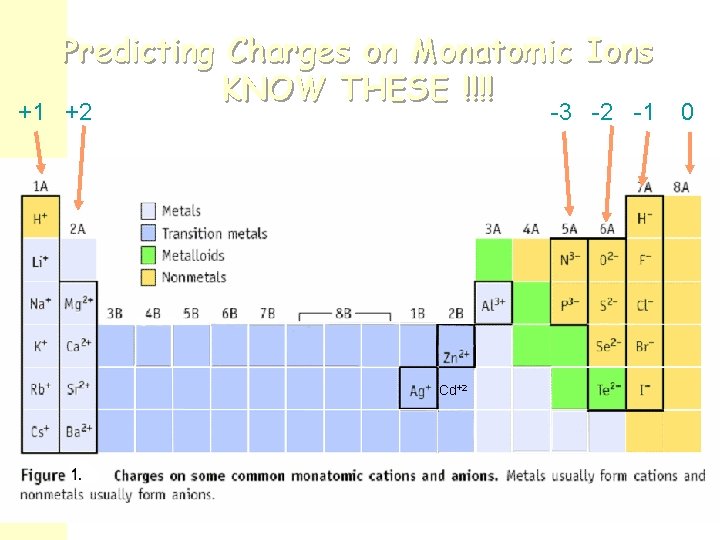
Predicting Charges on Monatomic Ions KNOW THESE !!!! +1 +2 -3 -2 -1 Cd+2 0

COMPOUNDS FORMED FROM IONS CATION + ANION ---> COMPOUND Na+ + Cl- --> Na. Cl A neutral compound requires equal number of + and - charges.

Naming Compounds Binary Ionic Compounds: n n n 1. Cation first, then anion 2. Monatomic cation = name of the element 2+ = calcium ion n Ca 3. Monatomic anion = root + -ide - = chloride n Cl n Ca. Cl 2 = calcium chloride

Naming Binary Ionic Compounds l Examples: Na. Cl Zn. I 2 Al 2 O 3

Learning Check Complete the names of the following binary compounds: Na 3 N sodium ________ KBr potassium ________ Al 2 O 3 aluminum ________ Mg. S _____________

Formulas for Binary Ionic Compounds n A binary compound is one made of two different elements. There can be one of each element such as in sodium bromide or potassium iodide. There can also be several of each element such as lithium oxide or aluminum bromide.

Writing Formulas Formula Unit – lowest whole number ratio of ions in an ionic compound n 1. 2. 3. 4. 5. Points to remember about writing the formula from the name The order in a formula is first the cation, then the anion. You must know the charges associated with each cation and anion. The sum of the positive charge and the sum of the negative charges MUST add up to zero. You MAY NOT adjust the charges of the cations or anions to get a total charge of zero. You MAY adjust the subscripts to get a total charge of zero.

Example #1 Suppose you must write the formula for sodium chloride. Write down the Na+ and Cl¯ right next to each other, as in this image: Move the positive charge (dropping the sign) to the subscript position of the anion: Move the negative charge (dropping the sign) to the subscript position of the cation: The result of all this moving is: Since subscripts of one are not written, but understood to be present, the final answer is:

Example #2 n Write the formula for aluminum oxide. n n Write down the Al 3+ and O 2¯ right next to each other : n n Move the positive charge (dropping the sign) to the subscript position of the anion: n n Move the negative charge (dropping the sign) to the subscript position of the cation: n n The result of all this moving is:



Learning Check Write the correct formula for the compounds containing the following ions: 1. Sodium sulfide a) Na. S b) Na 2 S c) Na. S 2 2. Aluminum chloride a) Al. Cl 3 b) Al. Cl c) Al 3 Cl 3. Magnesium nitride a) Mg. N b) Mg 2 N 3 c) Mg 3 N 2

Solution 1. Na+ S 2 b) Na 2 S 2. Al 3+ Cla) Al. Cl 3 3. Mg 2+ N 3 c) Mg 3 N 2

Examples: Type I Binary Ionic Compounds Write the formulas: Write the names: • potassium oxide • K 3 N • zinc chloride • Ag. I • silver sulfide • Zn. Br 2 • aluminum nitride • Al 2 O 3 • gallium oxide • Ba 3 P 2 • calcium iodide • Li. H

Transition Metals Elements that can have more than one possible charge MUST have a Roman Numeral to indicate the charge on the individual ion. 1+ or 2+ Cu+, Cu 2+ copper(I) ion copper (II) ion 2+ or 3+ Fe 2+, Fe 3+ iron(II) ion iron(III) ion

Names of Variable Ions These elements REQUIRE Roman Numerals because they can have more than one possible charge: anything except Group 1 A, 2 A, Ag, Zn, Cd, and Al (You should already know the charges on these!) Or another way to say it is: Transition metals and the metals in groups 4 A and 5 A (except Ag, Zn, Cd, and Al) require a Roman Numeral. Fe. Cl 3 Cu. Cl Sn. F 4 Pb. Cl 2 Fe 2 S 3 (Fe 3+) (Cu+ ) (Sn 4+) (Pb 2+) (Fe 3+) iron (III) chloride copper (I) chloride tin (IV) fluoride lead (II) chloride iron (III) sulfide

Learning Check Complete the names of the following binary compounds with variable metal ions: Fe. Br 2 iron (_____) bromide Cu. Cl copper (_____) chloride Sn. O 2 ___(_____ ) _______ Fe 2 O 3 ____________ Hg 2 S ____________

Examples: Type II Binary Ionic Compounds Write the formulas: Write the names: • iron (II) oxide • Fe 2 O 3 • copper (II) chloride • Sn. S • tin (IV) sulfide • Ni. Br 2 • cupric nitride • Cu. S • nickel (III) oxide • Pb 3 P 2 • ferrous iodide • Cu. Br • cobalt (III) selenide • Fe. Cl 3

Formulas of Ionic Compounds Formulas of ionic compounds are determined from the charges on the ions atoms ions Na + F : sodium + fluorine Charge balance: Na+ – : F : Na. F sodium fluoride 1+ 1 - formula = 0
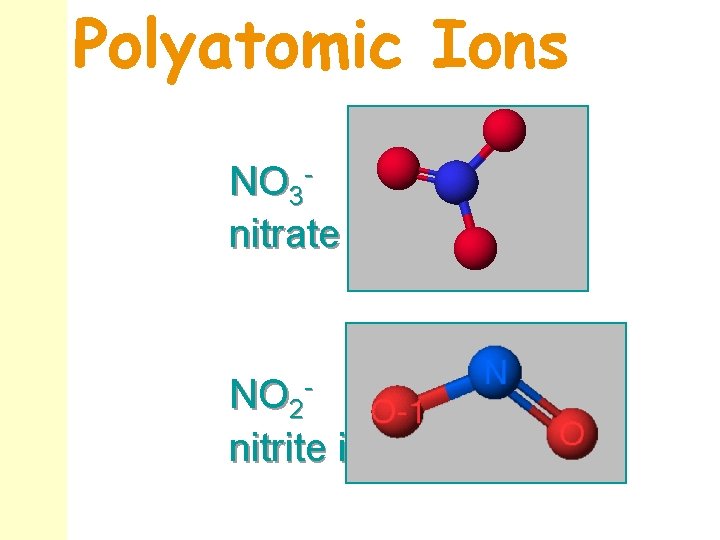
Polyatomic Ions NO 3 nitrate ion NO 2 nitrite ion

Polyatomic (Complex) Ions n n All of the cations and anions so far have been simple ions - single atoms that have lost or gained electrons. Complex polyatomic ions may be cations or anions. examples: nitrate: NO 3 - sulfate: SO 42 OH- hydroxide:
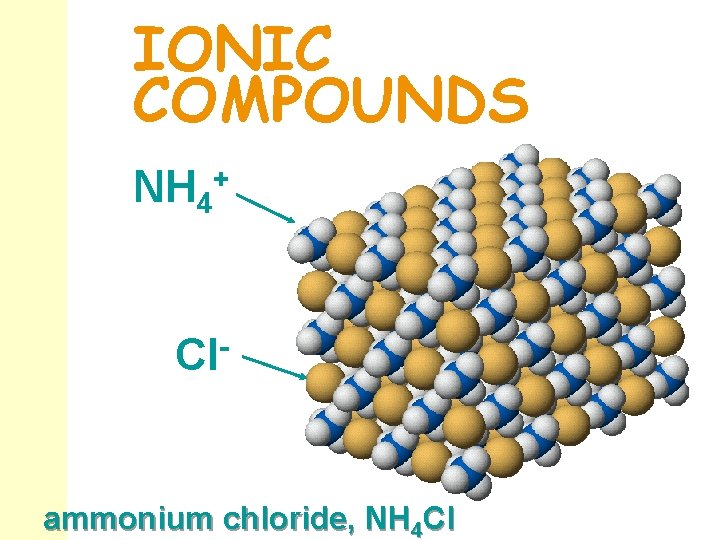
IONIC COMPOUNDS NH 4+ Cl- ammonium chloride, NH 4 Cl

Things to Notice n n Most complex ions are anions. Ammonium, NH 4+, is the most common complex cation. Several complex ions form a short series of ions. The ions differ only in the number of oxygen atoms: perchorate Cl. O 4 - sulfate SO 42 - chlorate Cl. O 3 - sulfite SO 32 - chlorite Cl. O 2 - hypochlorite Cl. O- nitrate NO 3 - nitrite NO 2 -

n If an ion carries a charge like “-2” or “-3”, a series of related ions can be formed by adding hydrogen cations (H+) while still leaving a net charge: examples: Sulfide: S 2 - hydrogen sulfide: HS- (bisulfide) Sulfate: SO 42 - hydrogen sulfate: (bisulfate) Carbonate: CO 32 - hydrogen carbonate: Phosphate: PO 43 - hydrogen phosphate: (bicarbonate) HSO 4 HCO 3 HPO 42 - dihydrogen phosphate: H 2 PO 4 -

Ionic Compounds containing Polyatomic Ions Writing Formulas n n Write each ion, cation first. Don’t show charges in the final formula. Overall charge must equal zero. u. If charges cancel, just write symbols. u. If not, use subscripts to balance charges. n n Use parentheses to show more than one of a particular polyatomic ion. Use Roman numerals to indicate the ion’s charge when needed (stock system)


Learning Check 1. aluminum nitrate a) Al. NO 3 b) Al(NO)3 c) Al(NO 3)3 2. copper(II) nitrate a) Cu. NO 3 b) Cu(NO 3)2 c) Cu 2(NO 3) 3. Iron (III) hydroxide a) Fe. OH b) Fe 3 OH 4. Tin(IV) hydroxide a) Sn(OH)4 b) Sn(OH)2 c) Fe(OH)3 c) Sn 4(OH)

Writing formulas with Complex Ions a) Ammonium chloride b) Silver sulfate c) Aluminum hydroxide d) Calcium phosphate e) Iron (III) nitrite f) Copper(II) permanganate g) Ammonium dichromate h) Zinc acetate

More Formulas with Complex Ions a) Na. HCO 3 b) Ni(HS)2 c) Al(Cl. O 4)3 d) Ba(H 2 PO 4)2 e) Fe 2(SO 2)3 f) Cu. HSO 4 g) Zn(IO 4)2

Mixed Practice! Name the following: 1. Na 2 O 2. Ca. CO 3 3. Pb. S 2 4. Sn 3 N 2 5. Cu 3 PO 4 6. Hg. F 2

Mixed Up… The Other Way Write the formula: 1. Copper (II) chlorate 2. Calcium nitride 3. Aluminum carbonate 4. Potassium bromide 5. Barium fluoride 6. Cesium hydroxide
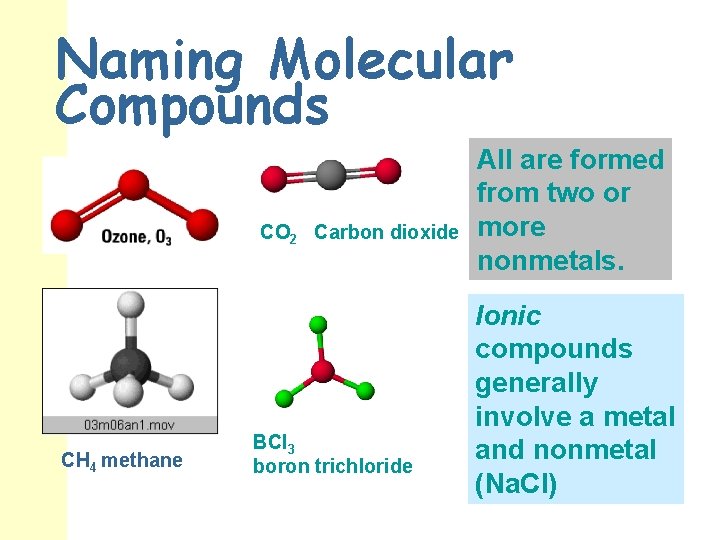
Naming Molecular Compounds CO 2 Carbon dioxide CH 4 methane BCl 3 boron trichloride All are formed from two or more nonmetals. Ionic compounds generally involve a metal and nonmetal (Na. Cl)

Binary Covalent Compounds n n Covalent compounds are made of two NONMETAL elements sharing valence electrons. There are no ions involved!! Because there are no charges to help us write the formulas of covalent compounds, prefixes are used to indicate the number of each atom present in the formula. The first element receives a prefix if the subscript is greater than one. The second element ALWAYS gets a prefix. Change the ending of the second element to ide. The prefix, “mono” is never used for the first element in the formula!

Molecular (Covalent) Nomenclature for two nonmetals Prefix System (binary compounds) CO 2 is named “carbon dioxide” CO is named “carbon monoxide” N 2 O is named “dinitrogen monoxide” SO 3 is named “sulfur trioxide” n .
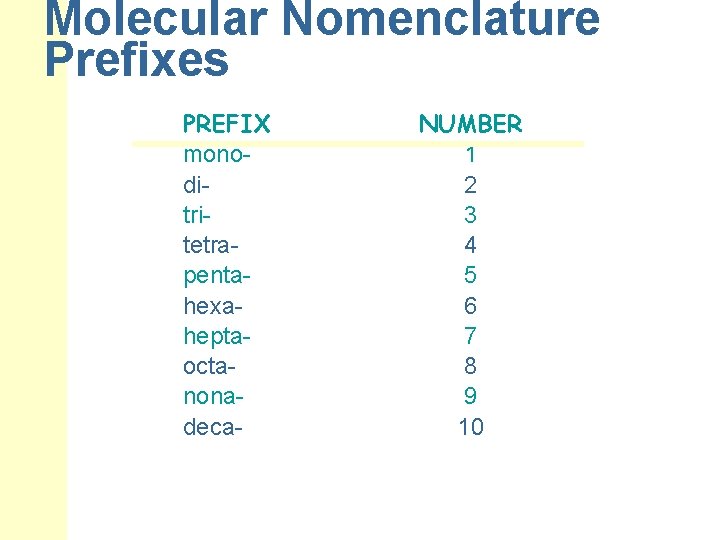
Molecular Nomenclature Prefixes PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

Molecular Nomenclature: Examples n CCl 4 n N 2 O n SF 6

More Molecular Examples n arsenic trichloride n dinitrogen pentoxide n tetraphosphorus decoxide

Learning Check Fill in the blanks to complete the following names of covalent compounds. CO carbon ______oxide CO 2 carbon ________ PCl 3 phosphorus _______chloride CCl 4 carbon ____chloride N 2 O _____nitrogen _____oxide

Learning Check 1. P 2 O 5 a) phosphorus oxide b) phosphorus pentoxide c) diphosphorus pentoxide 2. Cl 2 O 7 a) dichlorine heptoxide b) dichlorine oxide c) chlorine heptoxide 3. Cl 2 a) chlorine b) dichlorine c) dichloride

Binary Acids n n n A simple definition of an “acid” is a substance which produces H+ ions in water. Most acids have hydrogen ions in their formulas. A binary acid composed of a hydrogen cation bonded to one other element: HCl hydrochloric acid HBr hydrobromic acid H 2 S hydrosulfuric acid Binary acids are always named: hydro____ic acid

Oxy-Acids n An acid can also be made of a hydrogen ion bonded to a complex ion. Complex ions whose names end in “ate”: NO 3 - CO 32 - SO 42 - nitric acid: HNO 3 sulfuric acid: H 2 SO 4 carbonic acid: H 2 CO 3 phosphoric acid: H 3 PO 43 - Can you propose a rule for naming oxy-acids with complex ions ending in “ate”?

More Oxy-Acids n Complex ions ending in “ite”: NO 2 - SO 32 - Cl. O- nitrous acid: HNO 2 sulfurous acid: H 2 SO 3 hypochlorous acid: HCl. O chlorous acid HCl. O 2 - Can you propose a rule for naming oxy-acids with complex ions ending in “ite”?

Hydrated Ionic Compounds n n A HYDRATE is an salt that has water molecules trapped within its crystals. Every hydrate has a certain number of water molecules associated with each formula unit of the ionic compound. The number of water molecules is indicated by using prefixes. mono di tri tetra penta hexa hepta octa nona deca Cu. SO 4 · 5 H 2 O copper (II) sulfate pentahydrate Mg. CO 3 · 10 H 2 O magnesium carbonate decahydrate

Examples of hydrates: n n Write the formulas: copper(II) fluoride tetrahydrate Cu. F 2 · 4 H 2 O calcium nitrate trihydrate Ca(NO 3)2 · 3 H 2 O Write the names: Mg. SO 4 · 7 H 2 O magnesium sulfate heptahydrate Fe. Cl 2 · H 2 O iron (II) chloride monohydrate
- Slides: 47