How to Write Formulas for Binary Ionic Compounds

How to Write Formulas for Binary Ionic Compounds and Name them.

What is an Ionic Compound? • Ionic Compounds form when positively charged metal ions bond to negatively charged nonmetal ions. • Metal + Nonmetal = Ionic Compound • Example: Li+1 + S-2 = Li 2 S
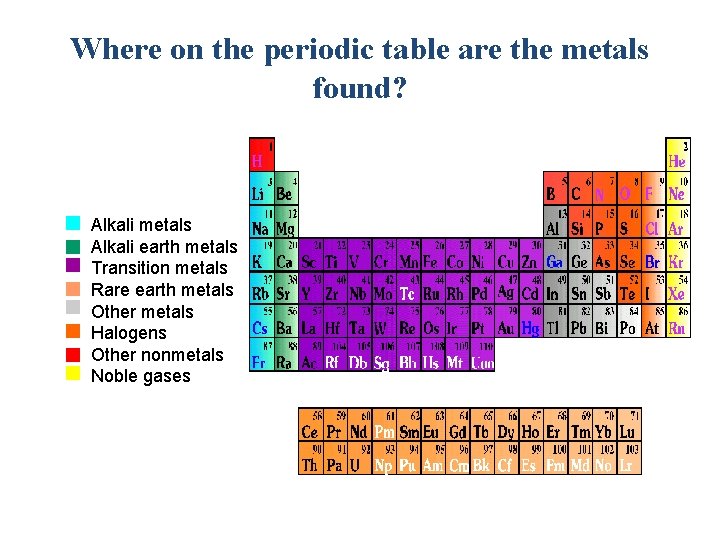
Where on the periodic table are the metals found? Alkali metals Alkali earth metals Transition metals Rare earth metals Other metals Halogens Other nonmetals Noble gases

Binary Compounds n n n A binary compound is composed of two elements. bi = 2 Examples: n Na. Cl n H 2 O n Mg. S

Which of the following are Ionic compounds? 1) 2) 3) 4) 5) 6) K 2 O CH 4 SO 3 Na 2 O Mg. Br 2 C 6 H 12 O 6

Some elements have more than one oxidation number. • These elements will combine differently depending on their oxidation number. • The different oxidation numbers are indicated by Roman Numerals • Ex: Copper (I) Cu+1 Copper (II) Cu+2 Iron (II) Fe+2 Iron (III) Fe+3

Rules for Writing the Formulas of Binary Ionic Compounds Example: Al+3+Br-1 = Al Br 3 1. Write the symbol of the element with the positive oxidation number. 2. Write the symbol of the element with the negative oxidation number. 3. Cross and drop the oxidation numbers 4. If both subscripts are divisible by the same number, they must be reduced **Note: The Sum of the oxidation numbers should add up to zero to have a stable bond.
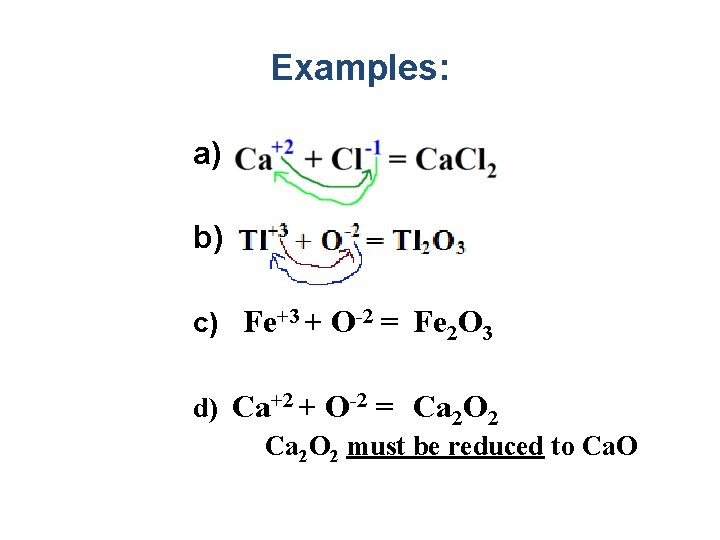
Examples: a) b) c) Fe+3 + O-2 = Fe 2 O 3 d) Ca+2 + O-2 = Ca 2 O 2 must be reduced to Ca. O

Naming Binary Ionic Compounds

Rules for Naming Ionic Binary Compounds Element Root Chlorine Fluorine Nitrogen Oxygen Sulfur Phosphorus Chlor. Fluor. Nitr. Ox. Sulf. Phosph - Ø Write the name of the first element listed in the chemical formula Ø Write the root name of the second element Ø Add the ending -ide to the root

Examples: • Na. Cl = Sodium Chloride • Mg. Br 2 = Magnesium Bromide • Ti. O 2 Titanium Oxide =

- Slides: 12