Ionic Bonding Key Knowledge common properties of ionic

Ionic Bonding Key Knowledge: -common properties of ionic compounds (brittleness, hardness, high melting point, difference in electrical conductivity in solid and liquid states) with reference to their formation, nature of ionic bonding and crystal structure including limitations of representations

4. 1 Properties and structures of ionic compounds • Ionic compounds are made from the chemical combination of metallic and non-metallic elements. • The materials are very common in the natural world because the Earth’s crust is largely made up of complex ionic compounds.

Common properties of ionic compounds (brittleness, hardness, high melting point, difference in electrical conductivity in solid and liquid states) with reference to their formation All ionic compounds have some properties in common. Generally ionic compounds: • Have high melting and boiling points • Are hard but brittle • Do not conduct electricity in the solid state • Are good conductors of electricity when in molten state or dissolved in water • Vary from very soluble to insoluble in water. THEY are not soluble in non-polar solvents such as oil.

But why do ionic compounds have these properties in common? Common properties of ionic compounds (brittleness, hardness, high melting point, difference in electrical conductivity in solid and liquid states) with reference to their formation Ionic compounds are created by positive and negative ions being attracted to each other by electrostatic forces of attraction. They form a lattice of positive and negative ions. So why does ions being held in a lattice give ionic compounds these properties. So how does this relate to common properties in reference to the formation?

Lets look at the example from the book (pg 85) In summary for ionic compounds generally: • The forces between particles are strong (which gives it the high melting and boiling points and mean that they are hard…. we will get back to the brittle) • There are no free moving particles present (metals have those delocalized electrons that move around the metal atoms) but ions are held in an 3 -D electrostatic lattice. • There are charged particles present (those ions…. cations and anions) but in solid state they are not free to move. When the substance is dissolved in water or in molten state the ions are then released…. The charged particles can now move around and therefore can conduct electricity.

Ionic Bonding Model When metallic and non metallic atoms react to form ionic compounds, the following steps occur. • Metal atoms lose their outer shell (valence) electrons to the non-metal atoms and so become positively charged ions (cations) • Non-metal atoms gain electrons from the metal atoms to fill their valence shell and so become negative ions (anions) • These anions and cations arrange themselves in a 3 -D lattice. • This lattice is held together strongly by electrostatic forces of attraction (opposites attract, + is attracted to -). The electrostatic forces of attraction holding the ions together is called ionic bonding. • Even though cations are close to other cations the attractive force between the cation and the surrounding anions outweighs the repulsive force.
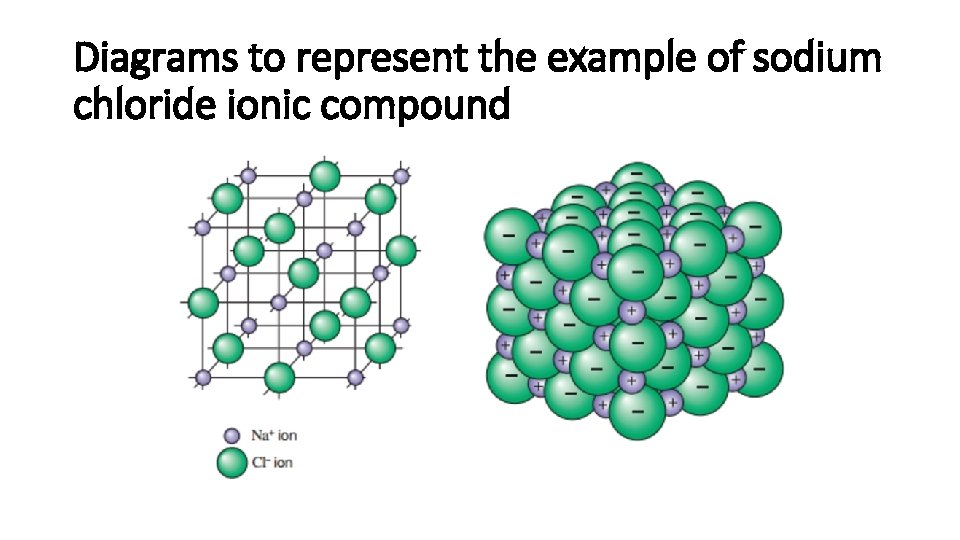
Diagrams to represent the example of sodium chloride ionic compound

4. 2 Using Ionic Bonding Model to explain properties Recap Common Ionic properties are: High melting point Hardness and Brittleness Electrical conductivity Solubility

High Melting Point • Looking at the example of sodium chloride You would need to heat Na. Cl to 801°C to give the ions enough energy to break free and move. This shows us that you need a large amount of energy is needed to overcome the electrostatic attraction between the oppositely charged ions and allow them to move. Therefore, the ionic bonds between the positive sodium ions and negative ions must be strong.

Hardness and Brittleness (see I told you I would get back to it) • The strong electrostatic forces of attraction between the ions in an ionic compound mean that the ions are held in a very rigid crystal lattice. To disrupt that lattice needs a lot of force. This means that ionic compounds are hard. • BUT, although they are hard if we were to hit an ionic compound it would shatter…. Which means they are brittle. • What is it about ionic compounds and the model that describes them that explains this?

Brittleness • Hitting an ionic compound causes the layers of ions to move relative to each other due to the force of the blow. During this movement, ions of like charges move relative to each other due to the force of that blow. Like charged ions are shifted so they are next to each other resulting in repulsion between the similarily charged ions…. and the crystals shatter.

Electrical conductivity • In solid form, ions in an ionic compounds are held in the crystal lattice and are not free to move, so solid ionic compounds do not conduct electricity.

Molten, or dissolved, ionic compounds and why they conduct electricity When ionic compounds melt, or are dissolved in water, they become free to move and therefore conduct electricity. When an electric current is applied to a molten or dissolved ionic compound, the positive ions move towards the negatively charged electrode and negative ions move towards the positively charged electrode.

Solubility Whether an ionic compound is soluble or insoluble depends on the relative strength of forces of attraction between the: • Positive and negative ions in the lattice • Water molecules and the ions
- Slides: 14