Ionic Bonding Ionic Bonding Ionic compounds are formed
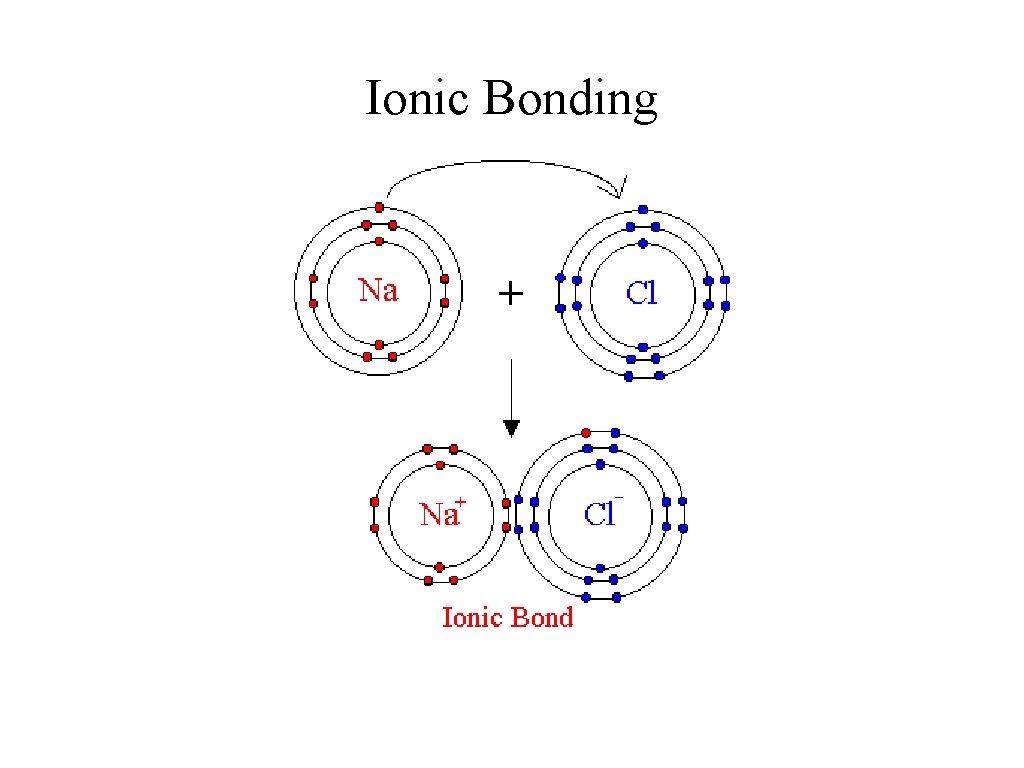
Ionic Bonding
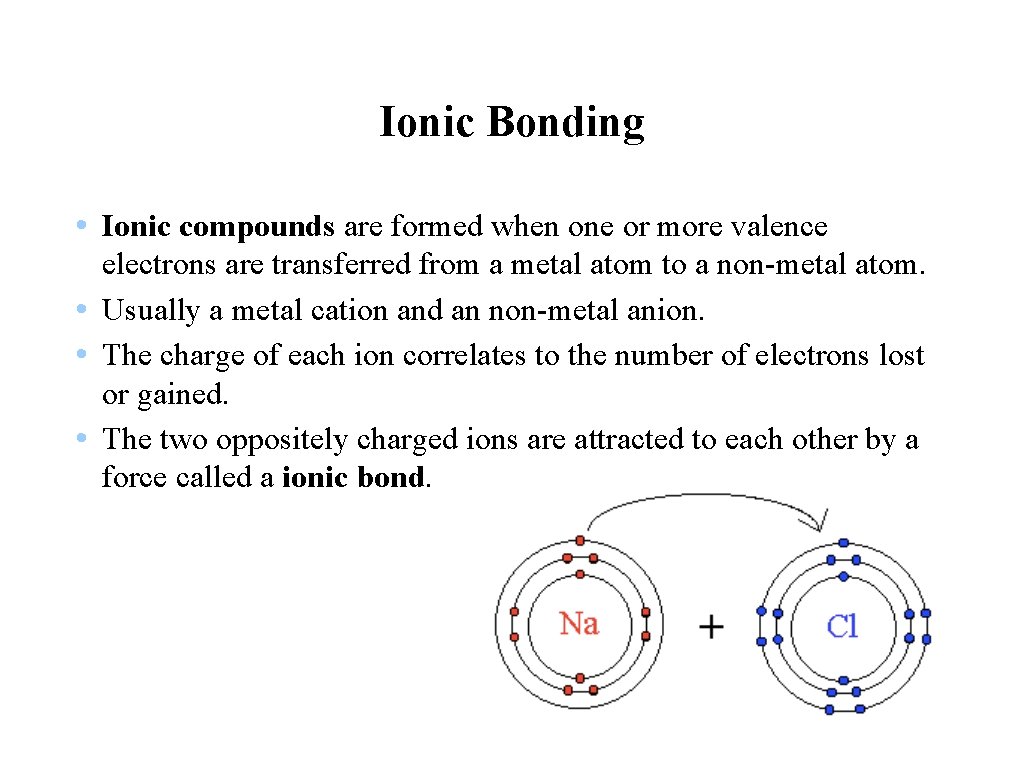
Ionic Bonding • Ionic compounds are formed when one or more valence electrons are transferred from a metal atom to a non-metal atom. • Usually a metal cation and an non-metal anion. • The charge of each ion correlates to the number of electrons lost or gained. • The two oppositely charged ions are attracted to each other by a force called a ionic bond.

Ionic Bonding • The smallest amount of substance that has the composition given by its chemical formula is the formula unit. • Eg. Na. Cl is a 1: 1 ratio, Mg. Cl 2 is a 1: 2 ratio.
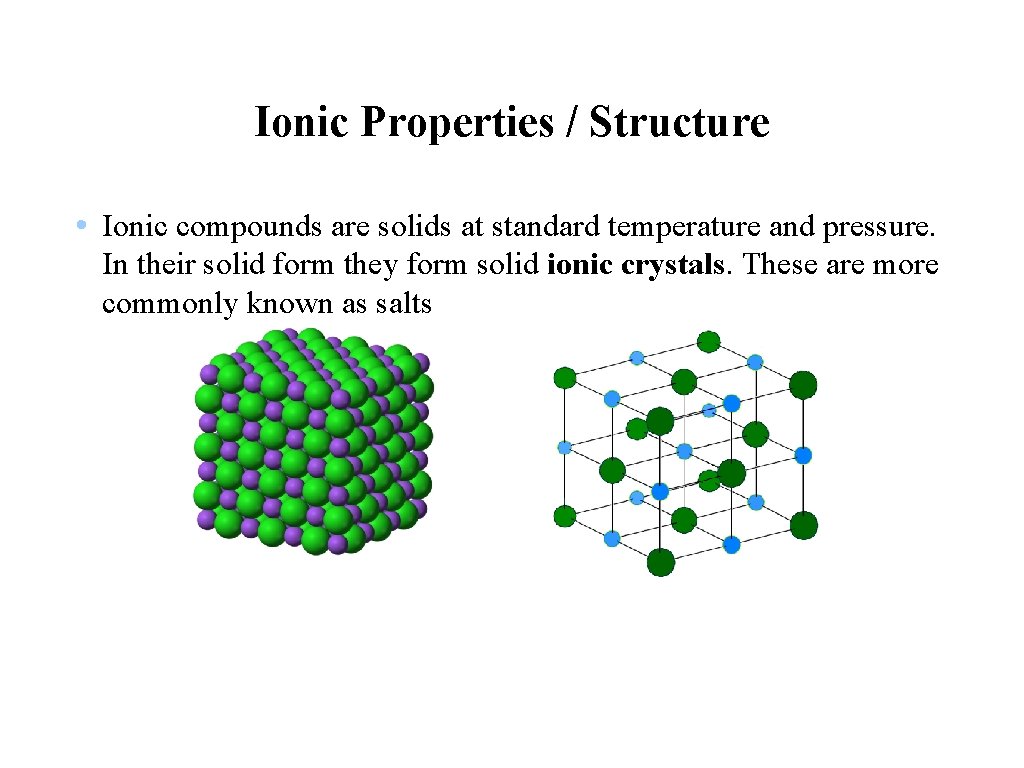
Ionic Properties / Structure • Ionic compounds are solids at standard temperature and pressure. In their solid form they form solid ionic crystals. These are more commonly known as salts

General properties • high MP, BP and hard. It takes a lot of energy to break the bonds between the ions. • Many are brittle • When ionic compounds are dissolved in water they dissociate into their ions. • Conduct electricity when dissolved in water • Na. Cl(s) Na+(aq) + Cl-(aq)
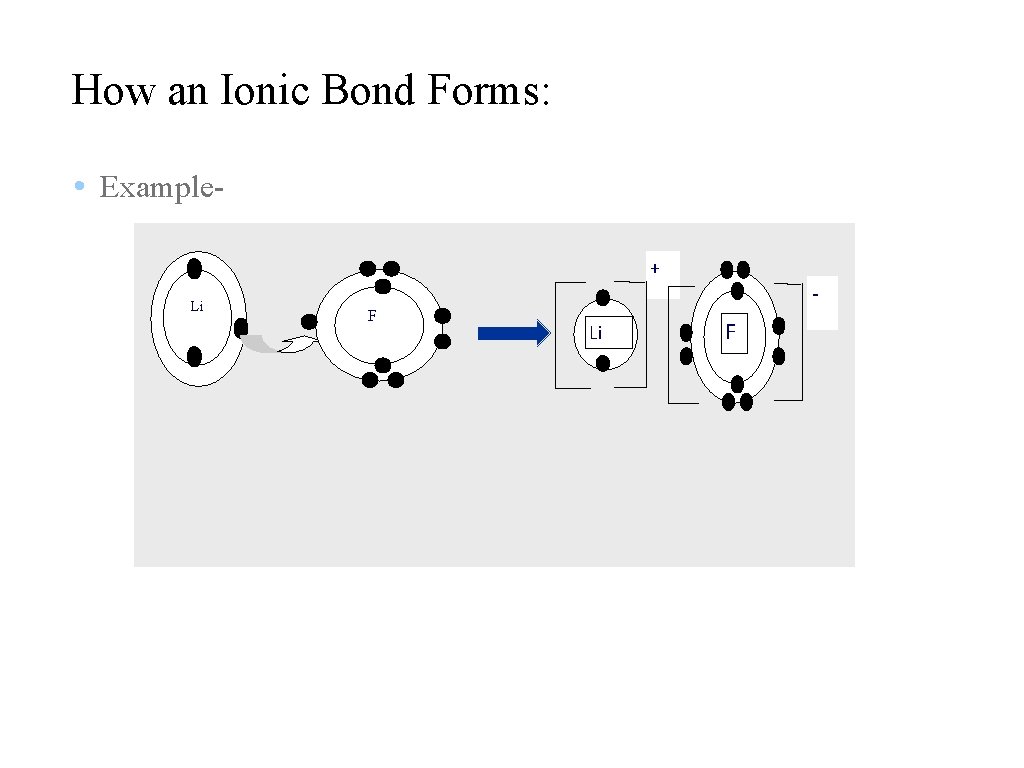
How an Ionic Bond Forms: • Example+ Li F
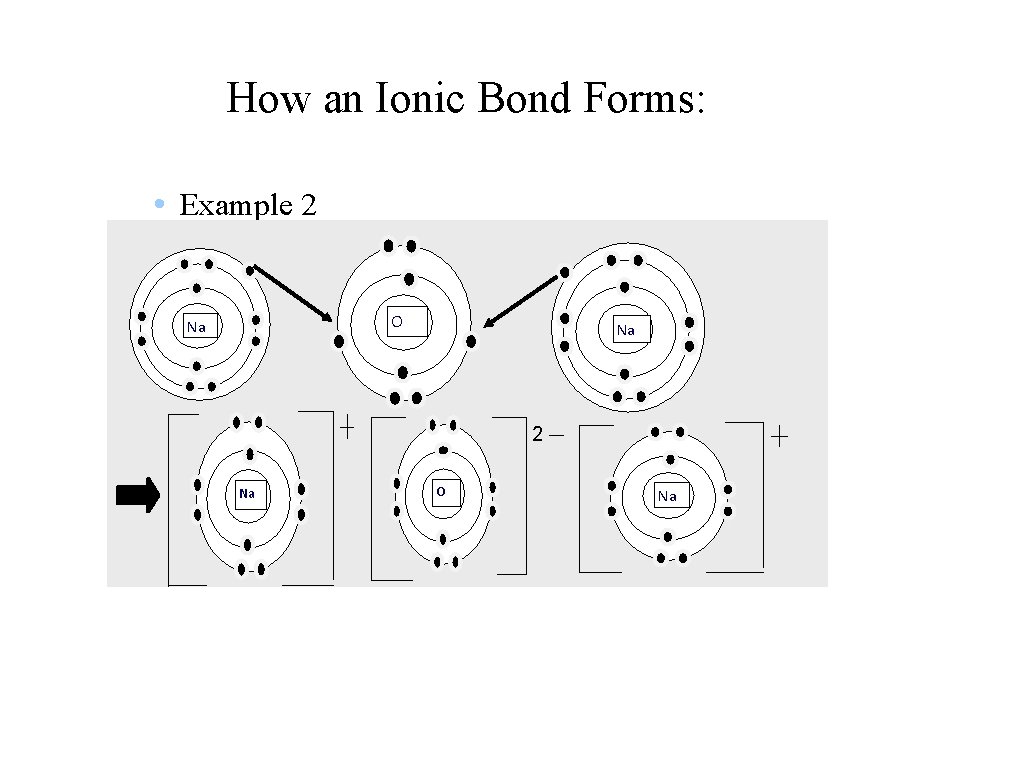
How an Ionic Bond Forms: • Example 2 O Na Na 2 Na O Na

The Cross over rule • This rule allows you to figure out how many atoms you will need of each element for bonding to occur without the need to draw Bohr diagrams

Cross Over Rule Step 1. Write the symbols, with the metal first (the element with the positive charge) Mg I Step 2. Write the Ionic charge above each symbol to indicate the stable ion that each element forms. 2+ Mg 1 I

Cross Over Rule • Step 3. Draw an arrow from the metals charge to the nonmetal and an arrow from the non-metal charge to the metal. (Cross over the arrows) 2+ Mg 1 I

Cross Over Rule Step 4. Fill in the number of atoms from each element will have by following the arrows. If need be reduce to lowest terms (in other words, if they are the same number, you don’t write those numbers down because you could divide the whole molecule by that number which would = 1) Mg. I 2 (if the number crossed is a 1, the 1 is not shown) Naming the compound Cation-gets the name of the element—> in this case Magnesium Anion-

Example 2 - Try yourself Ca O

Example 3 - Try yourself Zr O

Example 4 - Try yourself Al S

Example 5 - Try yourself Ni N
- Slides: 15