IONIC BONDS Ch 6 1 True False Statement

IONIC BONDS Ch 6. 1
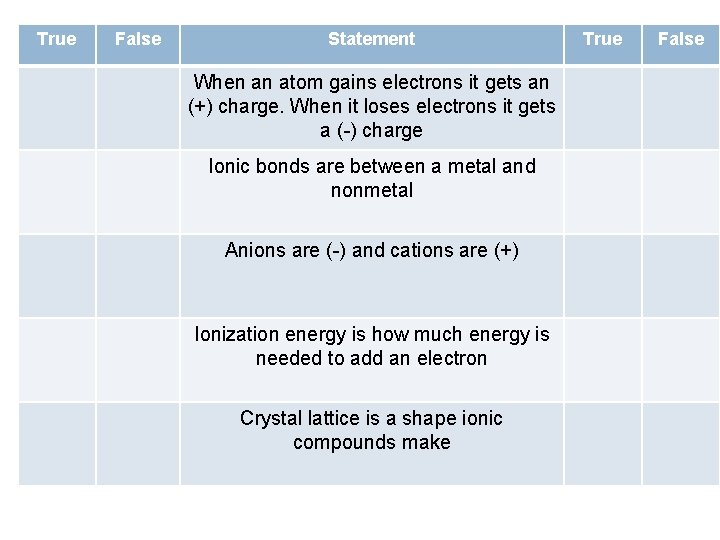
True False Statement When an atom gains electrons it gets an (+) charge. When it loses electrons it gets a (-) charge Ionic bonds are between a metal and nonmetal Anions are (-) and cations are (+) Ionization energy is how much energy is needed to add an electron Crystal lattice is a shape ionic compounds make True False

Electron Configuration Stable� Highest occupied energy level is filled with _______ � Atom not likely to react Electron Dot Diagram� Lewis Dot � Shows Valance Electrons
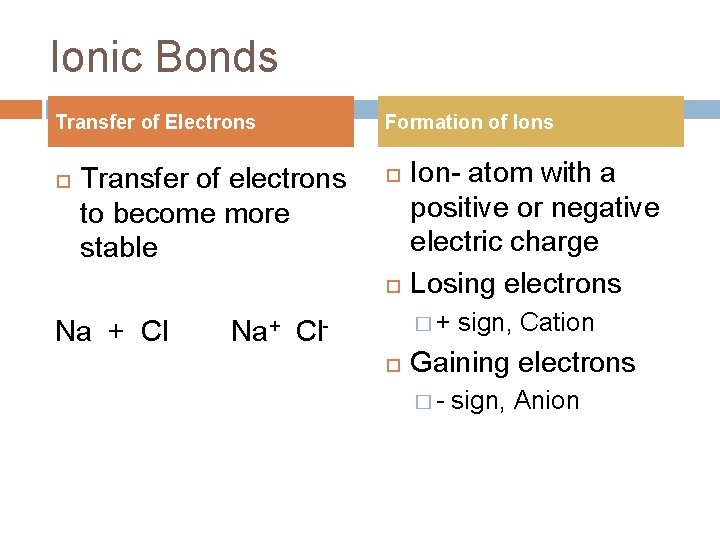
Ionic Bonds Transfer of Electrons Transfer of electrons to become more stable Formation of Ions Na + Cl Ion- atom with a positive or negative electric charge Losing electrons �+ Na+ Cl sign, Cation Gaining electrons �- sign, Anion

Ionic Bonds Continued Formation of Ionic Bonds Chemical Bondforce that holds atoms or ions together Ionic Bond- force that holds cations and anions together Ionization Energy - the amount of energy used to remove an electron Lower I-energy = easier to remove
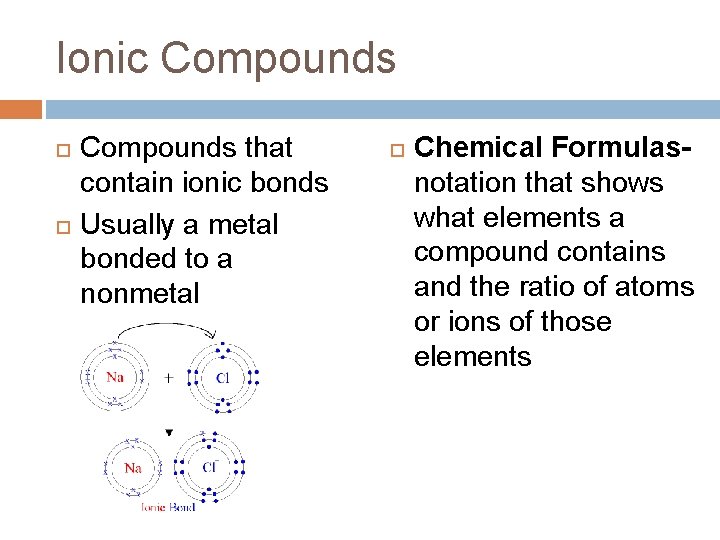
Ionic Compounds that contain ionic bonds Usually a metal bonded to a nonmetal Chemical Formulasnotation that shows what elements a compound contains and the ratio of atoms or ions of those elements
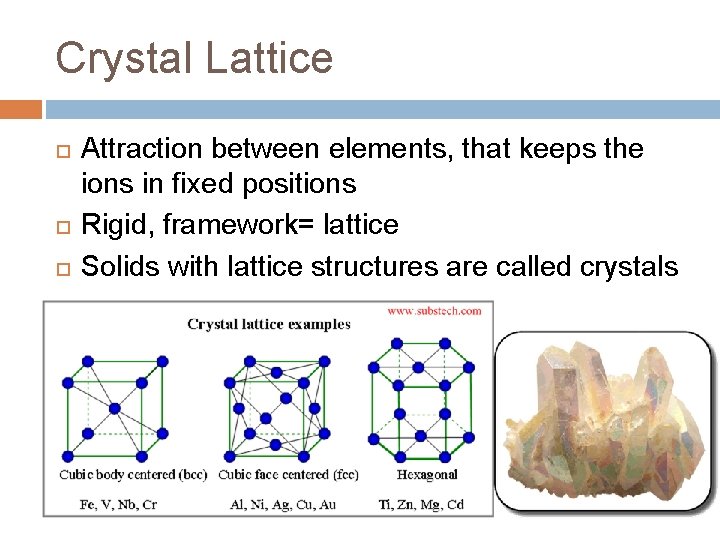
Crystal Lattice Attraction between elements, that keeps the ions in fixed positions Rigid, framework= lattice Solids with lattice structures are called crystals

Ionic Properties Strong attractions between ions within a crystal lattice When melted, great conductors of electricity Shatter when struck= _______

COVALENT BONDS Ch. 6. 2
True False Statement Covalent bonds are between 2 nonmetals, that share valence electrons Bonds can be single, up to quadruple Polar covalent bonds, are not shared equally Nonpolar bonds are stronger than polar bonds Polar bonds include, (-) and (+) signs to show polarity True False
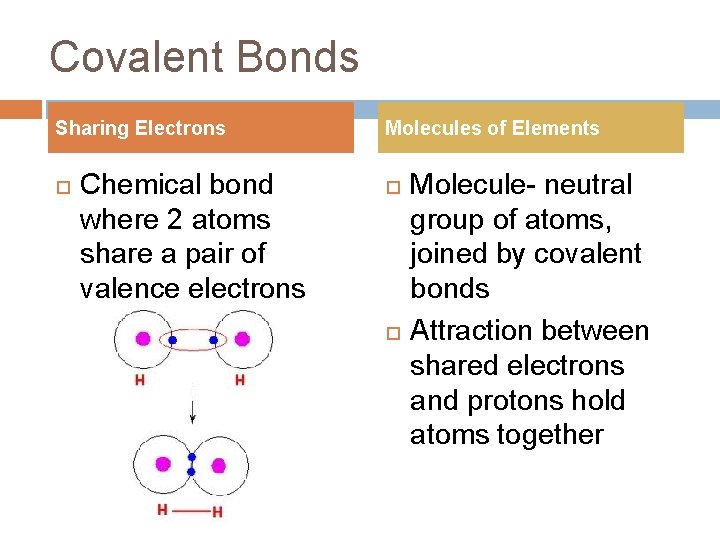
Covalent Bonds Sharing Electrons Chemical bond where 2 atoms share a pair of valence electrons Molecules of Elements Molecule- neutral group of atoms, joined by covalent bonds Attraction between shared electrons and protons hold atoms together
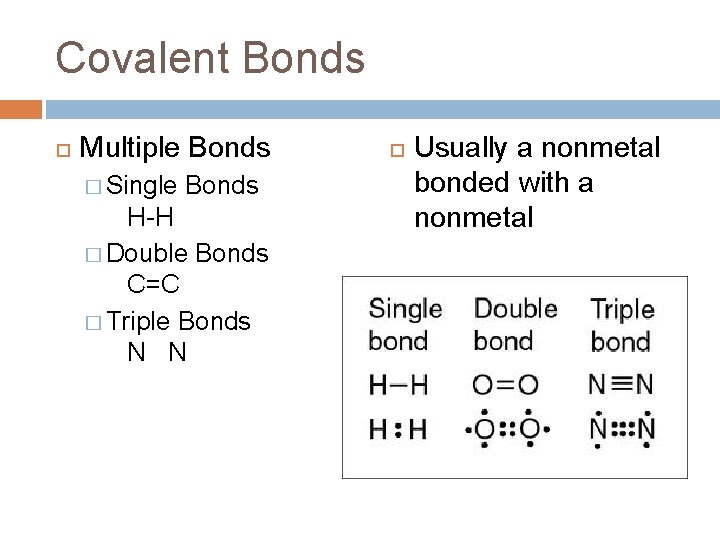
Covalent Bonds Multiple Bonds � Single Bonds H-H � Double Bonds C=C � Triple Bonds N N Usually a nonmetal bonded with a nonmetal
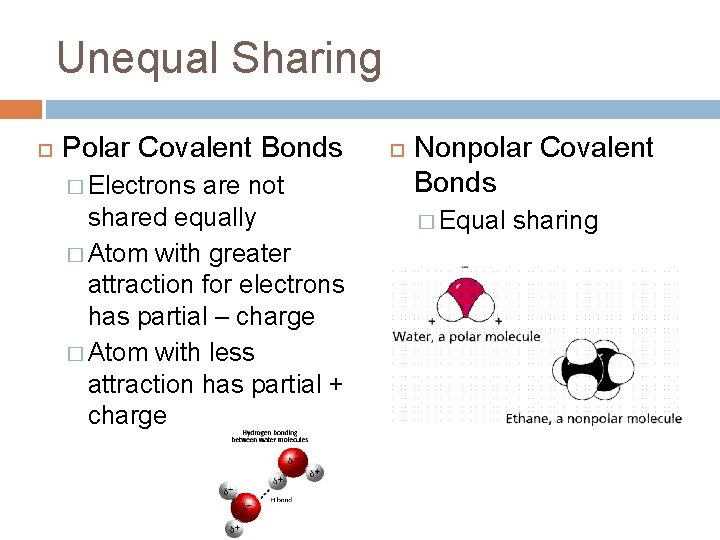
Unequal Sharing Polar Covalent Bonds � Electrons are not shared equally � Atom with greater attraction for electrons has partial – charge � Atom with less attraction has partial + charge Nonpolar Covalent Bonds � Equal sharing
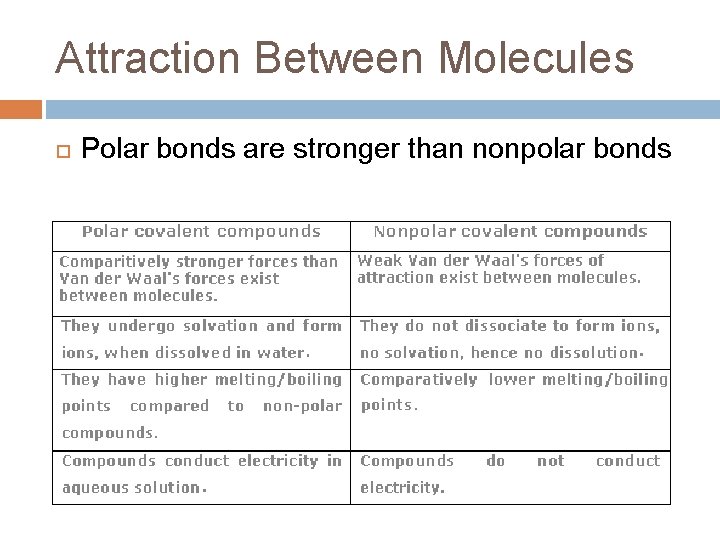
Attraction Between Molecules Polar bonds are stronger than nonpolar bonds

NAMING COMPOUNDS & WRITING FORMULAS Ch. 6. 3
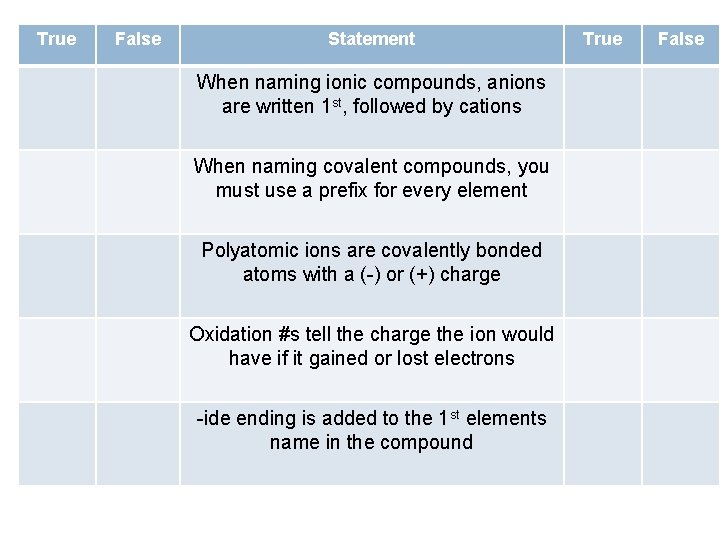
True False Statement When naming ionic compounds, anions are written 1 st, followed by cations When naming covalent compounds, you must use a prefix for every element Polyatomic ions are covalently bonded atoms with a (-) or (+) charge Oxidation #s tell the charge the ion would have if it gained or lost electrons -ide ending is added to the 1 st elements name in the compound True False

Naming Ionic Compounds Metal to nonmetal 1 st- Cation without change � Sodium Examples: � Li. F � Metal 2 nd- Anion � Nonmetal with suffix – ide � Chlorine = chloride 3 rd- Combine � Sodium chloride � KO � Cu. S � Be. Cl

Formulas for Ionic Compounds Calcium chloride 1 st- Write Symbols � Ca 3 rd- Find oxidation # � Ca+2 Cl-1 � Lose 2 electrons � Gain 1 electron Cl 2 nd- Locate � Ca Cl Cation anion 4 th- Criss cross, then write as subscript � Ca. Cl 2

Transition Metals Get oxidation # from anion � Must be neutral Backward crisscross Iron(II) oxide � Fe? O-2 � Fe+2 O-2 � Fe. O

Naming Examples Co. Br 2 � Cobalt(II) bromide Cr. Cl 3 � Chromium(III) chloride Mn. O 2 � Manganese(IV) Ag. O � Silver(II) oxide

Formula Examples Iron(II) oxide � Fe. O Iron(III) oxide � Fe 2 O 3 Gold(II) oxide � Au. O Platinum(III) oxide � Pt 2 O 3

Polyatomic Ions Covalently bonded group of atoms with either a positive or negative charge Most common list pg 173

Math Practice pg 174 1. 2. 3. 4.

Naming Molecular Compounds 2 Nonmetals 1 st- Count how many of each element � Use prefixes � Di-, pent 2 nd- Add element name and ending � Dinitrogen pentoxide

Naming Examples CO � Carbon CO 2 � Carbon monoxide dioxide N 2 O 4 � Dinitrogen tetraoxide N 2 O � Dinitrogen monoxide

Formula Examples Dicarbon tetraoxide � C 2 O 4 Tetraboron dichloride � B 4 Cl 2 Arsenic hexoxide � As. O 6 Silicon pentsulfide � Si. S 5
STRUCTURE OF METAL S Ch. 6. 4
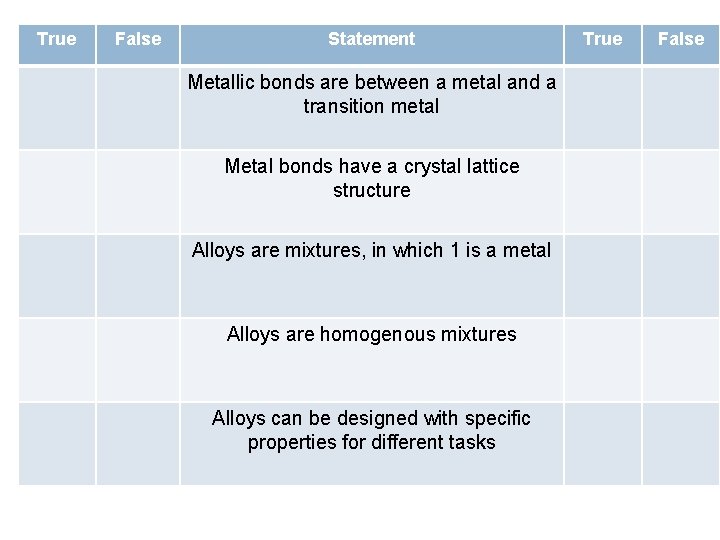
True False Statement Metallic bonds are between a metal and a transition metal Metal bonds have a crystal lattice structure Alloys are mixtures, in which 1 is a metal Alloys are homogenous mixtures Alloys can be designed with specific properties for different tasks True False

Metallic Bonds Attraction between a metal cation and the shared electrons surrounding it 2 metals, with no nonmetal to accept electrons Cations form a lattice=strong bonds between cations and valence electrons � Neutral charge
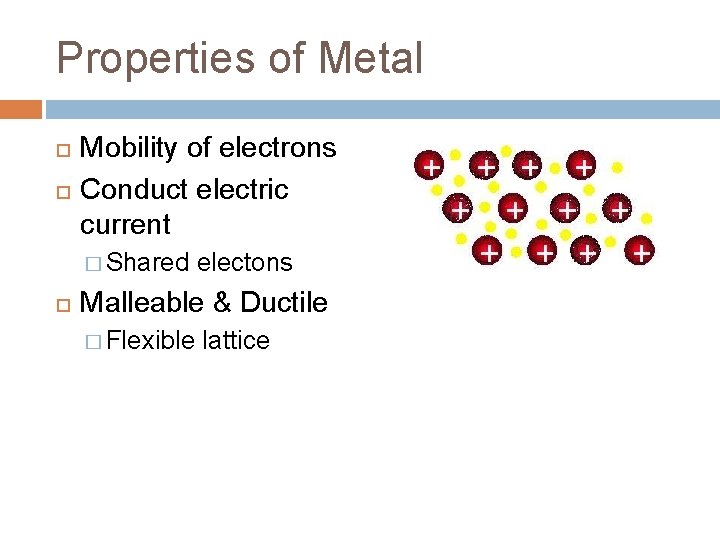
Properties of Metal Mobility of electrons Conduct electric current � Shared electons Malleable & Ductile � Flexible lattice

Alloys Mixture � 2 or more elements � 1 is a metal � heterogenous Characteristics of metals

Alloys Copper Bronze Tin Brass Harder, stronger than each metal alone Steel Iron Carbon Chromium Different mixtures for different needs
- Slides: 32