Ionic Compounds Review Naming and Writing Formulas Noble

Ionic Compounds Review Naming and Writing Formulas

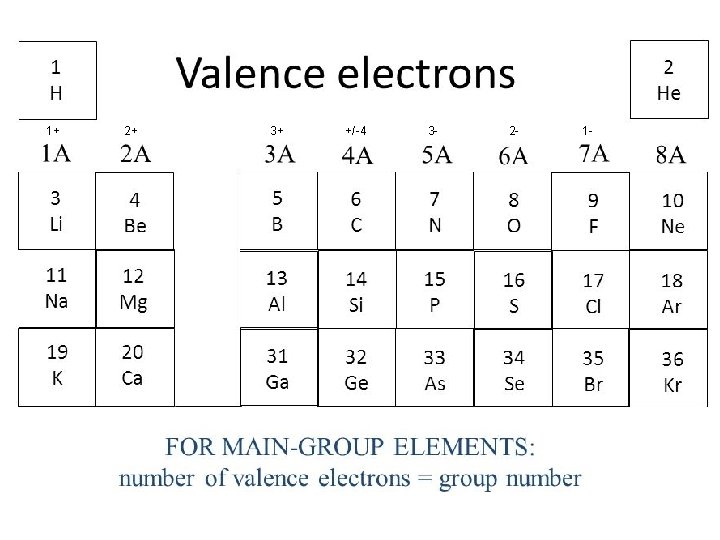
Noble Gases Have Stable Electron Configurations • Noble gases have a full octet (8 e-) • All elements want to look like noble gases Metals lose electrons to become cations Na+ Ca 2+ Al 3+ Non-metals gain electrons to become anions Cl. O 2 N 3 -

Naming Ions Atoms that lose electrons are called cations -Positively charged -No change in name (Na+ is a sodium ion) H Hydrogen H + Hydrogen ion

Naming Ions Atoms that gain electrons are called anions -Negatively charged -End of the name changed to -ide O oxygen N nitrogen O 2 - oxide N 3 - nitride Cl Cl Chlorine Chloride

Naming Binary Compounds Name the cation first and the anion last Na. Cl - sodium chloride Al. F 3 - aluminum fluoride Mg. O - magnesium oxide

Naming Ionic Compounds Is the cation a transition metal? Yes - use Roman Numerals with the cation’s name Fe. Cl 2 Iron(II) chloride Sn. I 4 Tin(IV) iodide Co. Cl 2 Cobalt(II) chloride

Naming Ionic Compounds Is there a polyatomic ion (group) in the formula? Use the polyatomic ion’s name just as it’s written! NH 4 Cl Ammonium chloride Na 2 SO 4 Sodium sulfate H 2 O 2 Hydrogen peroxide

Writing Chemical Formulas • Remember: Write the Cation first with its charge! • Write the Anion second with its charge! • Criss Cross to get subscripts Magnesium bromide Aluminum oxide 1 2 Al+3 O-2 2 3 Al 2 O 3

Writing Chemical Formulas Your Turn Potassium oxide Aluminum chloride Calcium phosphide
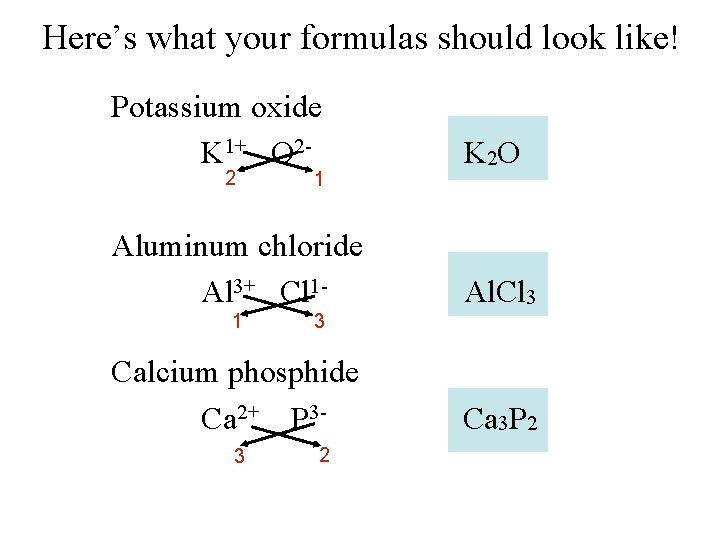
Here’s what your formulas should look like! Potassium oxide K 1+ O 2 - K 2 O Aluminum chloride Al 3+ Cl 1 - Al. Cl 3 2 1 1 3 Calcium phosphide Ca 2+ P 33 2 Ca 3 P 2

Molecules Naming and Writing Formulas
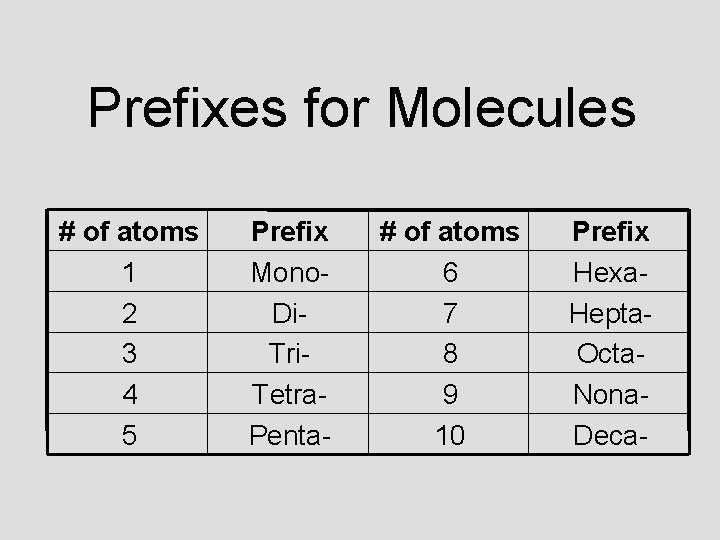
Prefixes for Molecules # of atoms 1 2 3 4 5 Prefix Mono. Di. Tri. Tetra. Penta- # of atoms 6 7 8 9 10 Prefix Hexa. Hepta. Octa. Nona. Deca-

Memorize Your Prefixes! Prefixes tell how many of each atom there is in a molecule. Molecules always have non-metals bonded to each other. A molecule’s name ALWAYS ends in -ide. Heads-up: A molecule’s name will NEVER start with the prefix mono!

Let’s Try Naming Molecules CCl 4 Carbon and chlorine are both non-metals. This is a molecule! The formula shows we have 1 carbon. The prefix for 1 is mono- but a molecule’s name NEVER starts with mono. So we will start with the element’s name, carbon. The formula shows we have 4 chlorine. The prefix for 4 is tetra. (tetrachlorine) Change the ending to -ide (tetrachloride) Our molecule’s name is: Carbon tetrachloride

Naming More Molecules NO 2 Nitrogen and oxygen are both non-metals. This is a molecule! The formula shows we have 1 nitrogen. The prefix for 1 is mono. Molecule’s names NEVER start with mono! So we will start with nitrogen! The formula shows we have 2 oxygen. The prefix for 2 is di- (dioxygen). Change the ending to -ide (dioxide). Our molecule’s name is Nitrogen dioxide

Name These Molecules! Se 2 Br 4 Te 3 I 9 N 2 O 4 P 3 O 5 As 4 F 6 CS 2 N 2 O 8 S 5 C 7 SO 2 CO 2

Writing Formulas We’ll reverse the process now! You definitely need to know your prefixes!

Let’s Try Some! Heptaphosphorus decaoxide Heptaphosphorus hepta- means 7, so we have 7 phosphorus decaoxide deca- means 10, so we have 10 oxygen How will you show 7 phosphorus and 10 oxygen (oxide) in a formula? P 7 O 10
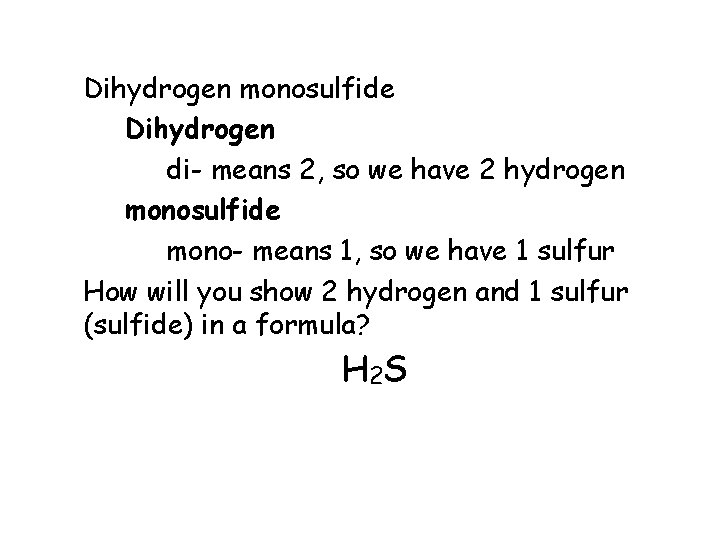
Dihydrogen monosulfide Dihydrogen di- means 2, so we have 2 hydrogen monosulfide mono- means 1, so we have 1 sulfur How will you show 2 hydrogen and 1 sulfur (sulfide) in a formula? H 2 S
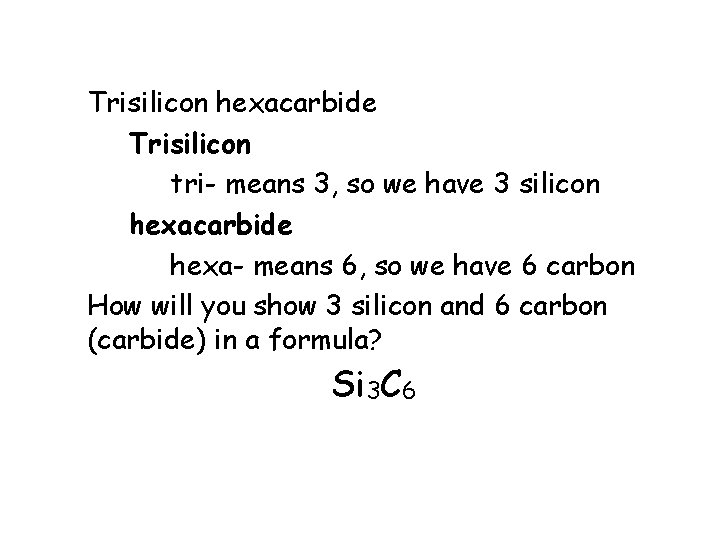
Trisilicon hexacarbide Trisilicon tri- means 3, so we have 3 silicon hexacarbide hexa- means 6, so we have 6 carbon How will you show 3 silicon and 6 carbon (carbide) in a formula? Si 3 C 6

Pentaoxygen octachloride Pentaoxygen penta- means 5, so we have 5 oxygen octachloride octa- means 8, so we have 8 chlorine How will you show 5 oxygen and 8 chlorine (chloride) in a formula? O 5 Cl 8

Write Formulas for the Following Molecules! Sulfur diiodide Carbon tetrabromide Boron trifluoride Dinitrogen tetraoxide Diphosphorus pentafluoride Tetraphosphorus heptaoxide
- Slides: 23