Ionic Compounds and Metals Bonding and Properties Chapter

Ionic Compounds and Metals Bonding and Properties Chapter 7 - pages 204 -229

Ions & Ionic Bonding • Section 7. 1: Ion Formation; pg 206 -209 • Learning Goals: – I understand how positive ions (cations) and negative ions (anions) are formed. – I understand why ions form and can relate ion formation to the valence electrons of an element. • Understanding the above is crucial to understanding chemistry- how elements combine to form compounds and chemical reactions.

Ion Formation Review • What is the octet rule? • Why do elements form ions? • Which electrons are involved when ions are formed? • What are positive ions called? • What are negative ions called?

Ion Formation • All elements are trying to become like a noble gas, in other words become more stable. • Elements achieve this stability by losing or gaining electrons to reach a full octet of valence electrons. • Some exceptions to the octet rule are: – Elements 1 -5: H, He, Li, Be and B. – Transition metals
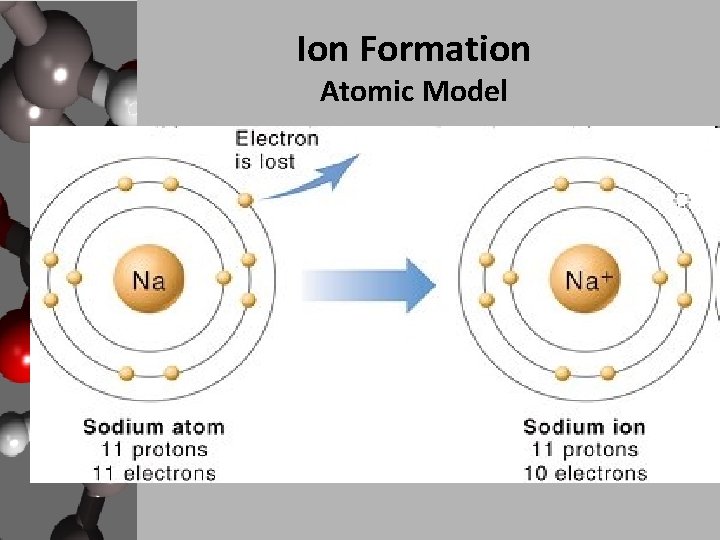
Ion Formation Atomic Model

Ion Formation • Lewis Dot Structures: – a simple shorthand way to represent valence electrons. – Used as an alternative to electron configurations and drawing atomic models. – Example: • Na → Na • Other ways to show ion formation: electron configurations & orbital diagrams.
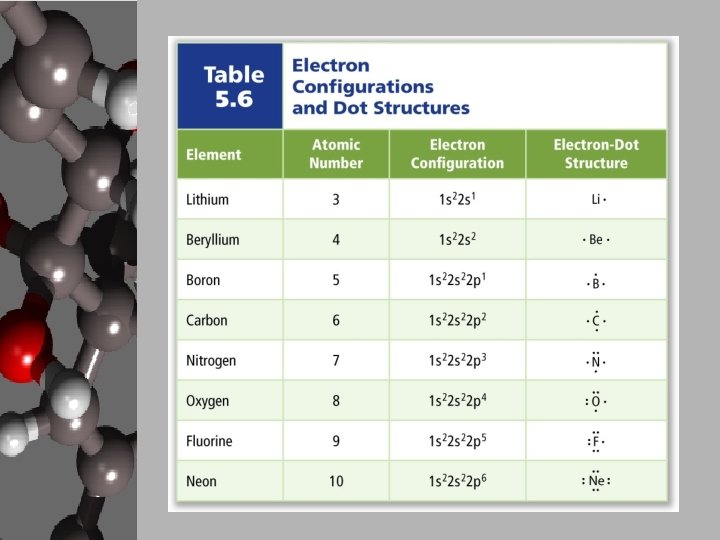
Lewis Dot Structures

Ion Formation • A cation is a positively charged ion. – Formed when a neutral element loses electrons. – All group #1 (alkaline metals), group #2 (alkaline earth metals) and group #13 (boron group) form cations. • An anion is a negatively charged ion. – Formed when a neutral element gains electrons. – All group #15 (nitrogen group), group #16 (oxygen group) and group #17 (halogen group) form anions. • Because it has 4 valence electrons, the carbon group can form both cations and anions, although we generally consider them to have a charge of zero.

Ionic Bonds and Ionic Compounds Section 7. 2, pages 210 -217

Ionic Bonding Learning goals: • Ionic bonds are formed when electrons are transferred from one atom to another. • Ionic bonds are held together by electrostatic attraction.

Ionic Bonding • Chemical Bond- is the force that holds 2 atoms together. • There are 3 general types of bonding – Ionic: ex. Na. Cl, Ca. Cl 2, Fe. S – Metallic: ex. copper, iron, brass – Covalent: ex. CH 4, C 6 H 12 O 6

Ionic Bonding: The Formation of Sodium Chloride q Sodium has 1 valence electron q Chlorine has 7 valence electrons q An electron transferred gives each an octet Na 1 s 22 p 63 s 1 Cl 1 s 2 s 2 p 3 s 3 p 2 2 6 2 5

Ionic Bonding: The Formation of Sodium Chloride This transfer forms ions, each with an octet: Na+ 1 s 22 p 6 Cl 1 s 2 s 2 p 3 s 3 p - 2 2 6

Ionic Bonding • Ionic bonds are held together by electrostatic attraction. This is the force of attraction by opposite charges. • The atom is held together by the electrostatic attraction of electrons (e-) and protons (p+). • Ionic compounds are held together by the electrostatic attraction of the cation (+) and anion (-).

Ionic Bonding • In general metals form cations and nonmetals form anions. • The oxidation state is the charge on a given ion. – Na+ – Ca+2 – Cl- oxidation state = +1 oxidation state = +2 oxidation state = -1 • Some metals can form ions with multiple oxidation states. – Fe+2, Fe+3 – Cu+, Cu+2

Naming Ionic Compounds • A cation retains the name from the element it was formed from. – Na+ – Ca+2 – Al+3 sodium cation Calcium cation aluminum cation • An anion is named by adding the suffix -IDE at the end of the name of the element it was formed from. – – Cl. S-2 O-2 N-3 chloride anion sulfide anion oxide anion nitride anion

Naming Ionic Compounds • Ionic compounds are named by combining the names of the cations and the anions. – Na. Cl – Na. F – Ca. Cl 2 – Mg. O – Fe. S sodium chloride sodium fluoride calcium chloride magnesium oxide iron sulfide

Ionic Compounds Properties • The packing of these positively charged cations and negatively charged anions must balance the forces of attraction and repulsion between the atoms.

Ionic Compounds Properties • The repeating pattern of particle packing in an ionic compound is called an ionic crystal.

Ionic Compounds Properties • A crystal lattice is the three-dimensional geometric arrangement of particles, and is responsible for the structure of many minerals. Quartz, Si. O 2 Pyrite, Fe. S 2

Ionic Compounds Properties • In a solid, ions are locked into position and electrons cannot flow freely—solid ions are poor conductors of electricity. • Liquid ions or ions in aqueous solution have electrons that are free to move, so they conduct electricity easily. • An ion in aqueous solution that conducts electricity is an electrolyte.

Physical Properties of Ionic Compounds • The properties of ionic compounds can be explained by the strong attractive forces between the cations and anions-electrostatic attraction. • Ionic compounds have very high melting and boiling points compared to most other solid compounds. • Ionic bonds are very strong compared to metallic and covalent bonds.
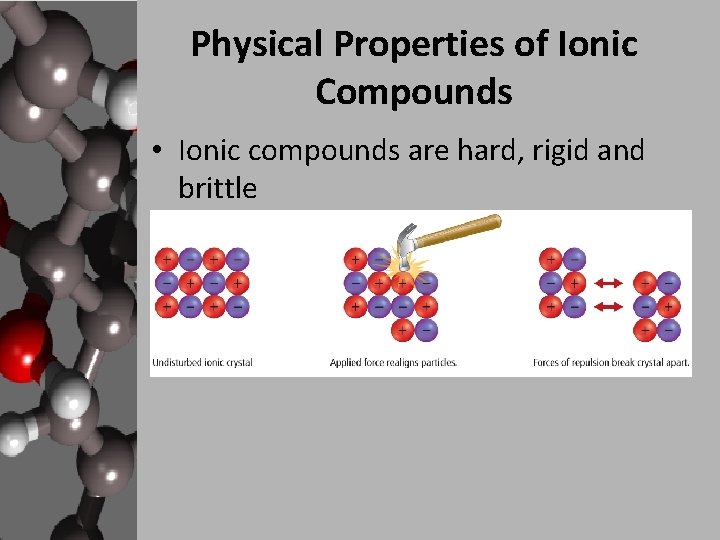
Physical Properties of Ionic Compounds • Ionic compounds are hard, rigid and brittle

Ionic Bonding • Positive and negative ions exist in a ratio determined by the number of electrons transferred from the metal atom to the non-metal atom. • Example: – Na. Cl – Ca. Cl 2

Names and Formulas for Ionic Compounds • • • Sodium + Chlorine Sodium + Oxygen Potassium + Iodine Calcium + Oxygen Calcium + Bromine

Ionic Compounds • https: //www. youtube. com/watch? v=c -n. Mmwf. TV-w • https: //www. youtube. com/watch? v= muk. CRQ 0 Gzf. Q&feature=related

Ionic Bonding
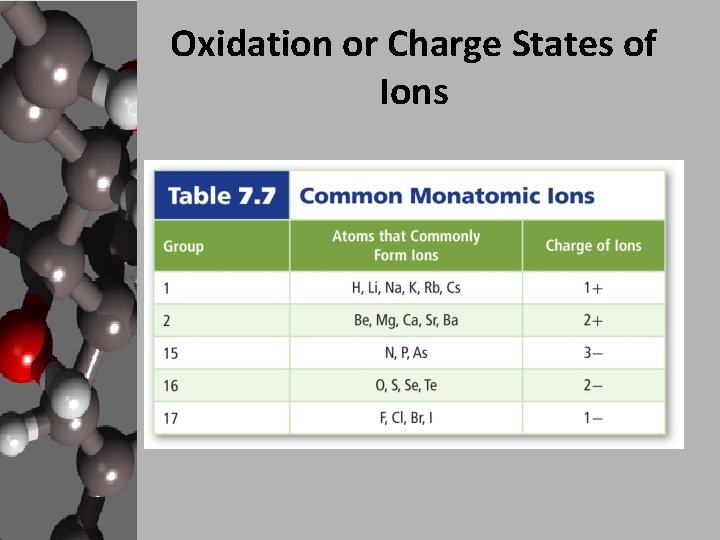
Oxidation or Charge States of Ions

Assessment 1. Explain how an ionic compound made up of charged particles can be electrically neutral. 2. Name 3 properties of ionic compounds. 3. Why are ionic compounds so strong? 4. How would you write the following ionic compounds: a. Potassium and fluoride b. Magnesium and chloride c. Sodium and oxygen

Review on Ionic Bonding • Which elements/groups form cations? Why? • Which elements/groups form anions? Why?

Metals & Metallic Bonding • Section 7. 4 - pages 225 -228. Learning Goals: • I can describe a metallic bond and I understand how a metallic bond differs from an ionic bond. • I understand the physical properties of metals and can relate these properties to their valence electrons. • I can define and describe what alloys are.
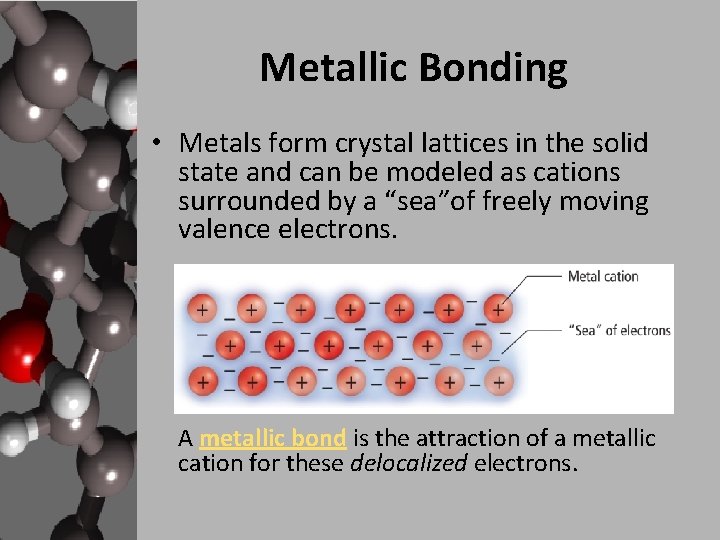
Metallic Bonding • Metals form crystal lattices in the solid state and can be modeled as cations surrounded by a “sea”of freely moving valence electrons. A metallic bond is the attraction of a metallic cation for these delocalized electrons.

Metallic Bonding • The metal ions are tightly packed in the crystal and their “d” electron orbitals overlap. • The electron sea model proposes that all metal atoms in a metallic solid contribute their valence electrons to form a "sea" of electrons. • The electrons are free to move around are referred to as delocalized electrons.

Review from last week 1. What are the 3 types of bonds? 2. Why do ions form? 3. What interaction holds ionic bonds together? 4. Last week’s BIG IDEA: oppositely charged ______ attract each other, forming electrically ___________. 5. What ion does Ca form? What is its oxidation state?
- Slides: 34