Video 6 1 Review Gas es Liquids and































































- Slides: 71

Video 6. 1 Review Gas es, Liquids, and Solids With Phase Change Dia grams

Objectives 0 By the end of this video you should be able to… 0 Identify and explain the three phases of matter. 0 Label and read phase change diagrams.

Kinetic Molecular Theory of Gases KMT describes perfect gases: 0 Gases move in constant, random, straight-line paths. 0 Gases are separated by large distances, much larger than their particle size. Therefore, gases volume is negligible. And gases are easily compressed. 0 Gases do not have attractive or repulsive forces between molecules. 0 Collisions between molecules can transfer energy but the total energy of the system is constant. This is called an elastic system.

Kinetic Molecular Theory of Gases In summary, Perfect gases: 0 Have no mass 0 Have no volume 0 Have no intermolecular forces

Kinetic Molecular Theory of Gases 0 But we don’t have perfect gases. How do real gases deviate from ideal gases? 0 They have a volume, mass and small IMF under high pressure and low temperature. 0 So, a real gas must be hot and under low pressure to behave like an ideal gas.

Pressure 0 Gases exert a pressure on surrounding substances because they are constantly moving and colliding with other surfaces. 0 Only in a vacuum, where there are no molecules, there is no pressure. 0 Gas pressure can be measured in atmospheres or kilopascals, according to reference table A.

Liquids 0 No definite shape 0 Definite volume 0 Constant motion 0 No arrangement 0 Molecules are closer together than a gas

Solids 0 Definite shape 0 Definite volume 0 Constant vibration 0 Molecules are packed tightly in a geometric (crystalline) pattern

Phase Changes Identify the phase change and if it’s endothermic or exothermic: Liquid to gas endothermic 0 Evaporation Gas to liquid exothermic 0 Condensation 0 Melting Solid to liquid endothermic 0 Freezing Liquid to solid exothermic 0 Sublimation Solid to gas endothermic 0 Deposition gas to solid exothermic

Thermochemistry 0 The study of energy changes that occur in chemical reactions. 0 Kinetic energy refers to energy of motion. (Temperature) 0 Potential Energy refers to stored energy.

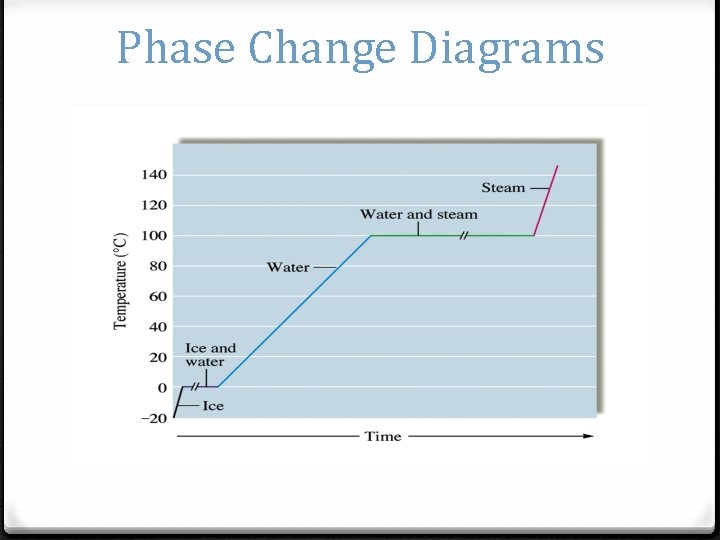
Phase Change Diagrams

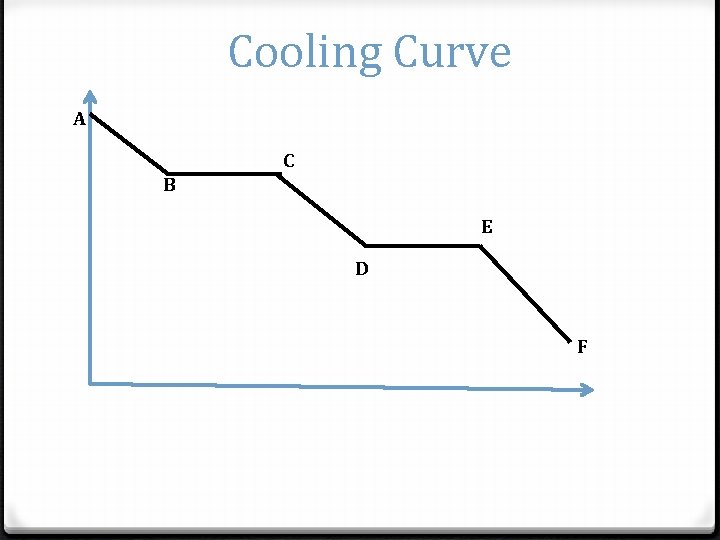
Cooling Curve A C B E D F

Objectives 0 Now you must be able to… 0 Identify and explain the three phases of matter. 0 Label and read phase change diagrams.

Video 6. 2 Q=mcΔT

Objectives 0 By the end of this video you should be able to… 0 Describe system and surroundings in terms of heat flow. 0 Describe and calculate the specific heat of a substance. 0 Calculate the heat of a reaction suing q=mcΔT

System and Surroundings 0 The system includes the molecules we want to study (here, the hydrogen and oxygen molecules). 0 The surroundings are everything else (here, the cylinder and piston).

Systems 0 An open system allows for energy and mass to change, like an open cup of coffee or beaker. 0 A closed system allows for changes in energy only, like a closed coffee cup or beaker. 0 An isolated system doesn’t allow any change in temperature or heat to occur, like a perfect calorimeter.

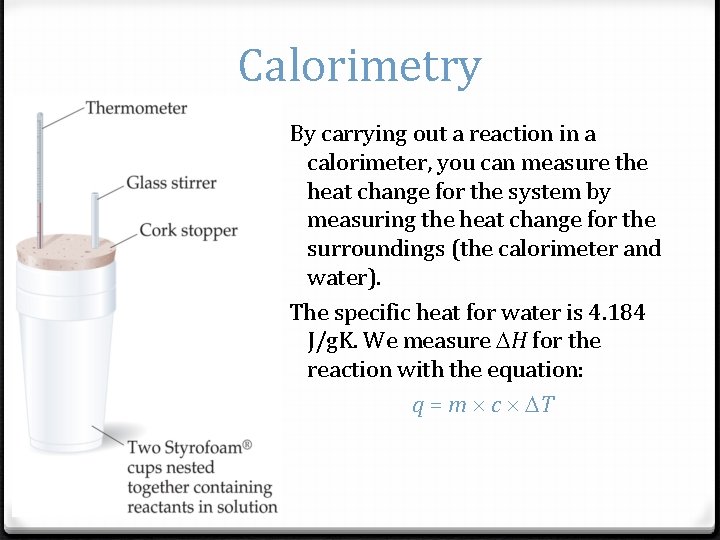
Calorimetry Since we cannot measure the exact heat contained inside of the reactants or products, we measure H through calorimetry, the measurement of heat flow.

Calorimetry By carrying out a reaction in a calorimeter, you can measure the heat change for the system by measuring the heat change for the surroundings (the calorimeter and water). The specific heat for water is 4. 184 J/g. K. We measure H for the reaction with the equation: q = m c T

Calculating Heat of reactions 0 q is the symbol for heat. 0 If q is positive, the heat is endo. 0 If q is negative, the heat is exo. 0 q is measure in Joules, (J) or kilojoules (k. J). 0 The heat of a reaction is based on: 0 the mass of the substance 0 the temperature change it undergoes 0 specific heat.

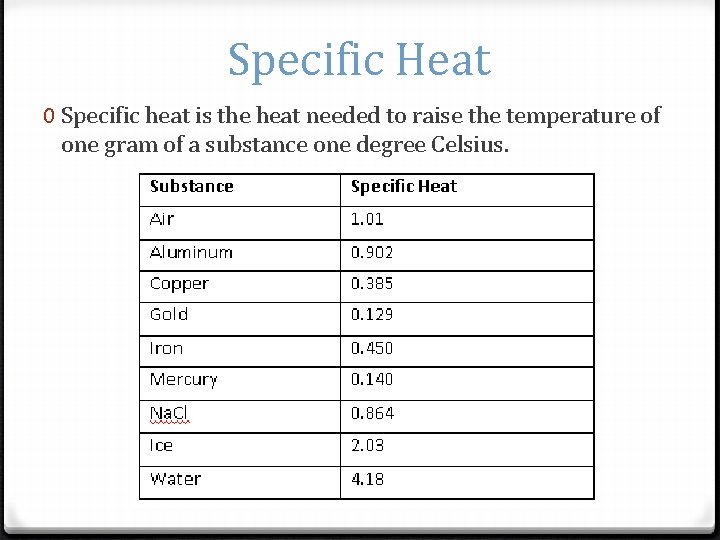
Specific Heat 0 Specific heat is the heat needed to raise the temperature of one gram of a substance one degree Celsius.

q = mcΔT The temperature of 95. 4 g of copper increases from 25 to 48 C and absorbed 849 J. Calculate copper’s specific heat. 849= (95. 4) (x) (48 -25) q = 0. 387 J/g. C

q = mcΔT How much heat is needed to raise the temperature of 100 g of water 50 C? q = (100) (4. 18) (50) q = 20900 J or 20. 90 KJ

q = mcΔT Find the mass of a substance that requires 450. 0 J to raise the temperature 5. 00 C. Its specific heat is equal to 8. 00 g/JC. (450. 0) = (x)(8. 00)(5. 00) x = 11. 3 g

q = mcΔT Find the specific heat of a substance that requires 350. 0 J to raise the temp of 50. 0 grams 11. 0 C. (350. 0)=(50. 0)x(11. 0) x = 0. 636 J/g. C

q = mcΔT 3. If 600 J are needed to heat 50 g of water to 100 C, what is the initial temperature? 600 = (50) (4. 18) (x) X = 2. 87 97. 13 C

Objectives 0 Now you must be able to… 0 Describe system and surroundings in terms of heat flow. 0 Describe and calculate the specific heat of a substance. 0 Calculate the heat of a reaction suing q=mcΔT

Video 6. 3 Table I

Objectives 0 By the end of this video you should be able to… 0 Determine if a reaction is endothermic or exothermic using Table I. 0 Fid the heat of a reaction using Table I.

Table I 0 Exothermic reactions release heat and have negative values. The products are more stable than the reactants were. 0 Example: When Carbon and Oxygen react they release 393. 5 k. J of heat per mole reacted. 0 Endothermic reactions absorb heat and have positive values. The products are less stable than the reactants were. 0 Example: When Nitrogen and Oxygen react they absorb 182. 6 k. J of heat per mole.

Table I Examples 0 When C 2 H 4 is formed, is heat released or absorbed? 0 Table I: 2 C + 2 H 2 C 2 H 4 ΔH=52. 4 k. J 0 When 2 moles of CH 4 burn in oxygen, how much heat is released? 0 Table I: CH 4 + 2 O 2 CO 2 + H 2 O ΔH=-890. 4 k. J 0 2 CH 4 2(-890. 4) = -1780. 8 KJ

Table I Examples 0 Reactions that release the most energy are the most stable. Which reaction becomes the most stable? 0 4 Al + 3 O 2 2 Al 2 O 3 ΔH=-3351 k. J 0 Where did these values come from?

Objectives 0 By the end of this video you should be able to… 0 Determine if a reaction is endothermic or exothermic using Table I. 0 Fid the heat of a reaction using Table I.

Video 6. 4 q=m. H q=m H f v

Objectives 0 By the end of this video you should be able to… 0 Calculate the heat during a phase change.

When can you use q=mcΔT? 0 Only on the solid, liquid and gas only lines. (Where the temperature changes) 0 So, what equations do we use if the temperature is not changing?

Table T 0 Heat of vaporization: heat needed to change a substance from gas to liquid or liquid to gas. q=m. Hv 0 Heat of fusion: heat needed to change a substance from solid to liquid or liquid to solid. q=m. Hf 0 If the IMF is strong, the heats of vaporization and fusion is high.

q=m. Hv 1. Calculate the number of joules needed to vaporize 423 g of H 2 O. q = (423) (2260) 955, 980 J or 955. 98 KJ

q=m. Hf 0 How much heat is needed to melt ice at 0 C if the sample weighs 255 g? q = (255) (334) 85, 170 J or 85. 17 KJ

Heat of a Phase Change A 50. 0 g compound requires 5000. 0 J to change from solid to liquid. What is its heat of fusion? q = m. Hf 5000. 0= 50. 0(x) x = 100. J/g

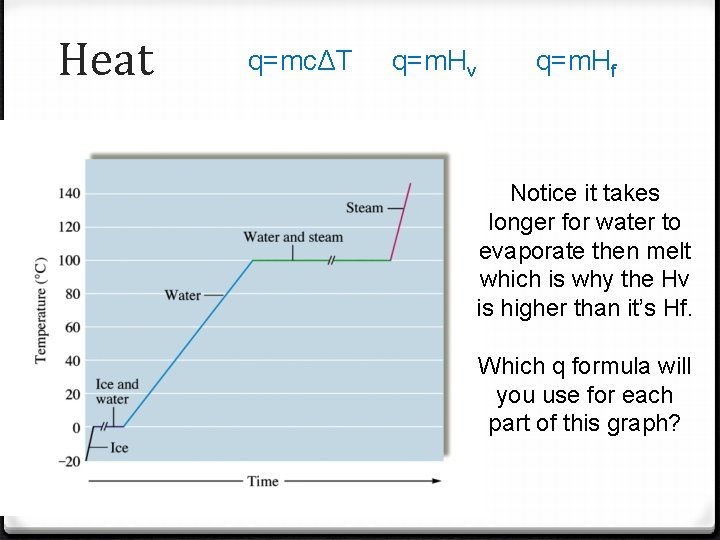
Heat q=mcΔT q=m. Hv q=m. Hf Notice it takes longer for water to evaporate then melt which is why the Hv is higher than it’s Hf. Which q formula will you use for each part of this graph?

Objectives 0 Now you must be able to… 0 Calculate the heat during a phase change.

Video 6. 5 Heat of Form ation and H ess’ Law

Objectives 0 By the end of this video you should be able to… 0 Calculate heats of reactions using heats of formation values. 0 Calculate heats of reactions using Hess’ Law.

Enthalpies of Formation A standard enthalpy of formation, Hf°, is defined as the enthalpy change for the reaction in which one mole of a compound is made from its elements in their elemental forms at 1 atm and 25 C. Elements are zero if the element is at it’s standard state. Hrxn° is the standard heat of a reaction at 1 atm and 25 C. This is obtained by Hf°(P-R)

Enthalpy of Formation Example Given the values below, calculate the ΔHrxn° of: 2 Al + Fe 2 O 3 2 Fe + Al 2 O 3 Fe 2 O 3 ΔHf° = -822. 1 k. J/mol Al 2 O 3 ΔHf° = -1676 k. J/mol [2(0) + -1676] - [2(0) + -822. 1] = -854 k. J/mol Notice all elements are zero and units are in k. J/mol This tells us only that the reaction is exothermic and may be favorable.

Enthalpy of Formation Example Given the values below, calculate the ΔHrxn° of: C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) C 3 H 8 ΔHf° = -103. 85 k. J/mol CO 2 ΔHf° = -393. 5 k. J/mol H 2 O ΔHf° = -285. 8 k. J/mol [3(-393. 5) + 4(-285. 8)] - [-103. 85 + 5(0)] = -2220. 0 k. J/mol

Hess’s Law 0 H° is well known for many reactions, and it is inconvenient to measure H° for every reaction in which we are interested. 0 However, we can estimate H° using other H° values that are published. 0 Hess’s law states that “If a reaction is carried out in a series of steps, H° for the overall reaction will be equal to the sum of the enthalpy changes for the individual steps. ”

Example Find the heat of reaction: Pb. Cl 2 + Cl 2 Pb. Cl 4 given: Pb + 2 Cl 2 Pb. Cl 4 ΔH = -329. 2 k. J/mol Pb. Cl 2 Pb + Cl 2 Δ H = 359. 4 k. J/mol ΔH = 30. 2 k. J/mol

Harder Example Given: H 2 O(s) H 2 O(l) CH 4(g)+2 O 2(g) CO 2(g)+2 H 2 O(l) Calculate the heat of reaction of: 2 CH 4(g)+4 O 2(g) 2 CO 2(g)+ 4 H 2 O(s) 2 CH 4(g)+ 4 O 2(g) 2 CO 2(g)+4 H 2 O(l) H=2(-890. 4 KJ) Multiply equation 2 by 2: Reverse quadruple equation 1: Combine new equations : H°=6. 01 KJ H°=-890. 4 KJ 4 H 2 O(l) 4 H 2 O(s) H=-4(6. 01 KJ) H = 2(-890. 4)+-4(6. 01) = -1804. 84 k. J

Objectives 0 Now you must be able to… 0 Calculate heats of reactions using heats of formation values. 0 Calculate heats of reactions using Hess’ Law.

Video 6. 6 Entropy

Objectives 0 By the end of this video you should be able to… 0 Define spontaneous reactions and entropy. 0 Determine which species have higher entropy values.

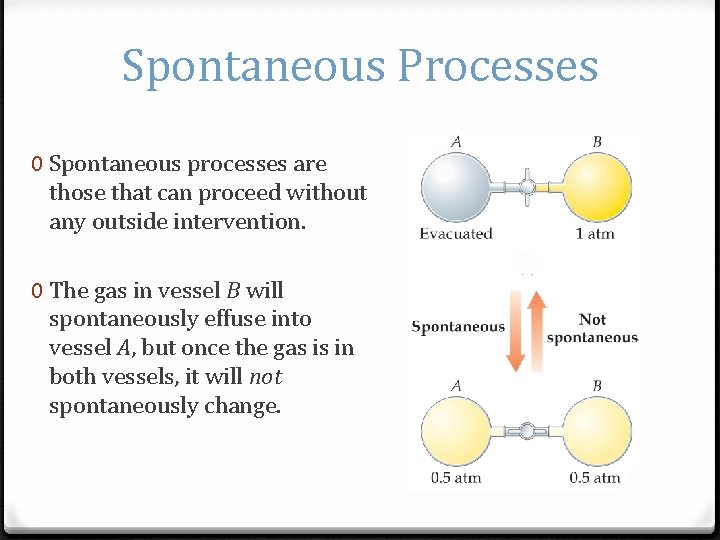
Spontaneous Processes 0 Spontaneous processes are those that can proceed without any outside intervention. 0 The gas in vessel B will spontaneously effuse into vessel A, but once the gas is in both vessels, it will not spontaneously change.

Spontaneous Processes 0 Processes that are spontaneous at one temperature may be nonspontaneous at other temperatures. 0 Above 0 C it is spontaneous for ice to melt. 0 Below 0 C the reverse process is spontaneous.

Spontaneous Processes 0 Spontaneity depends on two important issues: 0 Entropy can be thought of as a measure of the randomness of a system. 0 Enthalpy is the heat change of a reaction.

Second Law of Thermodynamics 0 The second law of thermodynamics states that the entropy of the universe increases for spontaneous processes.

Entropy Just like the standard enthalpies of formation, the standard Entropies are calculated: S r = S S (products) S S (reactants) f f This number can tell you if a reaction is random/chaotic or not at a given temperature. if S°rxn is negative it is spontaneous. if S°rxn is positive it is not spontaneous. But we will just compare samples….

Entropy is dependent on: 0 Temperature (phase changes) S(g) > S(l) > S(s) 0 The number of independently moving molecules Ssolute < Ssolution 0 The change in phase usually has a larger affect than the number of molecules. 0 Polarity provides order to the particles because they align themselves like poles of magnets. Entropy decreases when polar molecules form.

Third Law of Thermodynamics The entropy of a pure crystalline substance at absolute zero is 0.

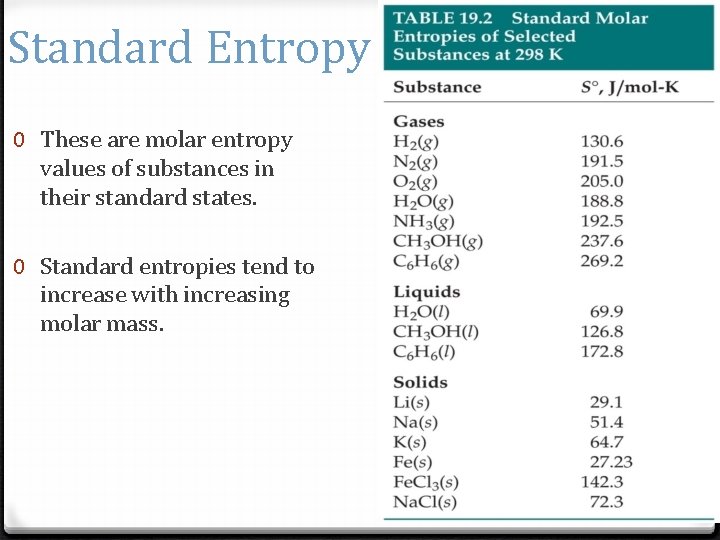
Standard Entropy 0 These are molar entropy values of substances in their standard states. 0 Standard entropies tend to increase with increasing molar mass.

Standard Entropies Larger and more complex molecules have greater entropies.

Examples 0 Which has a greater entropy? 0 Na. Cl(s) or Na. Cl(aq) 0 CO 2(s) or CO 2(g) 0 KI(aq) at 45 C or 100 C 0 Is the entropy positive (increasing) or negative? 0 2 H 2(g) + O 2(g) 2 H 2 O(l) 0 NH 4 Cl(s) NH 3(g) + HCl(g)

Objectives 0 Now you must be able to… 0 Define spontaneous reactions and entropy. 0 Determine which species have higher entropy values.

Video 6. 7 Gibbs Free E nergy

Objectives 0 By the end of this video you should be able to… 0 Use Gibbs Free Energy to determine if a reaction is spontaneous.

Standard Free Energy Changes Just like the standard enthalpies and entropies of formation, the standard Gibbs free-energy: G r = S G (products) S G (reactants) f f This number can tell you if a reaction is spontaneous or not at a given temperature. if G°rxn is negative it is spontaneous. if G°rxn is positive it is not spontaneous. But we will just compare samples….

Gibbs changes with Temperature G= H T S

Examples Will the following reactions be spontaneous? 0 A reaction that is exothermic and produces many moles of gas. 0 A reaction that is endothermic and produces solids. 0 A reaction that is cold to the touch and condenses. 0 A reaction that is hot to the touch and forms smoke.

Examples that are Temperature Dependent The following will be spontaneous only at high or low temperatures: low entropy and is ____ exo thermic. 0 H 2 O(g) H 2 O(l) + Heat has a _____ low temperatures. It will be spontaneous at ______ high entropy and is ____ endothermic. It 0 Heat + Cu(s) Cu(l) has a _____ high temperatures. will be spontaneous at ______ 0 Heat + C 6 H 12 O 6(s) 6 H 2 O(g) + 6 C(s) has a _____ high entropy and is ____ thermic. It will be spontaneous at ______ endo high temperatures.

Objectives 0 Now you must be able to… 0 Use Gibbs Free Energy to determine if a reaction is spontaneous.