DSQ 1 Whats the difference between a physical

DSQ 1. What’s the difference between a physical and chemical change? 2. What’s the difference between a mixture and a solution? 3. What is an atom? 4. Are all pure substances elements?

DSQ 1. Mixtures are divided into two classes, what are they and how are they different? 2. Pure substances are divided into two parts, what are they and how are they different?

Demo Coke and Milk

Matter Notes Matter – anything that has mass and takes of space.
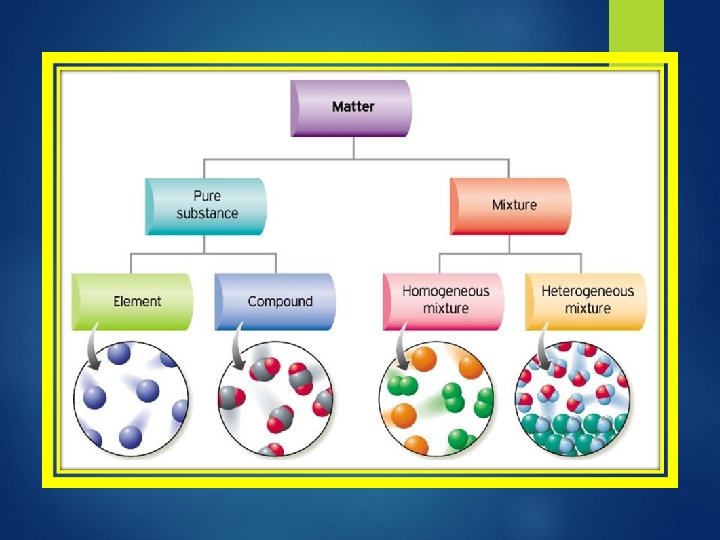
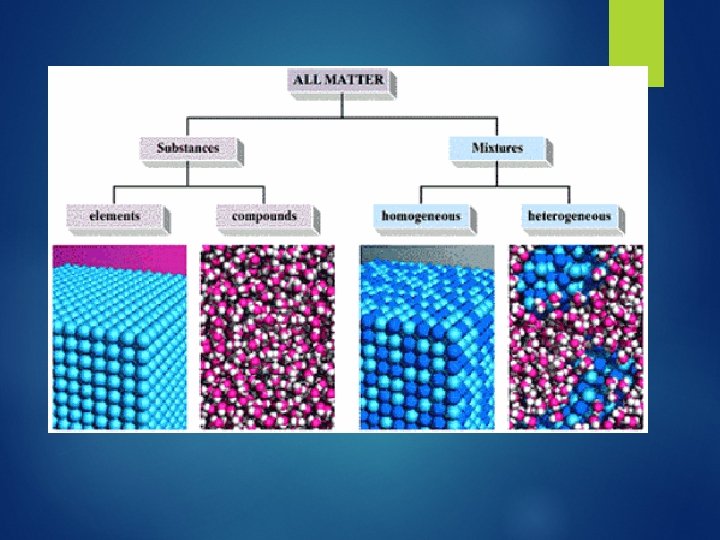

Two types of matter: Mixtures Pure substances

Mixtures: Consists of two or more substances that are mixed together but not chemically combined Solution – liquid mixture Ex: pre-baked cookies Soda; sea water 2 types of mixtures Homogeneous – uniform composition Heterogeneous – not uniform composition


Pure Substances Is made of only one kind of matter and has definite properties. Ex: sugar, salt, gold, iron Two Types: 1. Elements – are pure substances that cannot be broken down anymore. Look at periodic table 2. Compounds – is a pure substance formed from chemically combining two or more different elements H 2 O, CO 2, C 6 H 12 O 6 Atoms – smallest particle of a element

Pure Substances
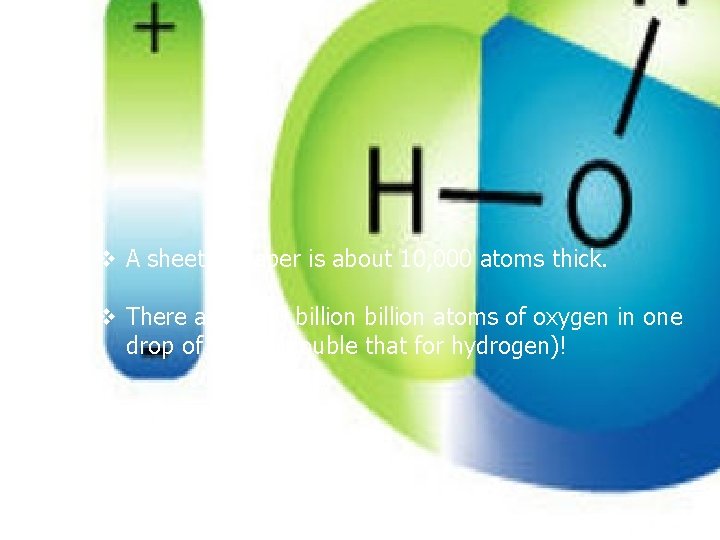
v A sheet of paper is about 10, 000 atoms thick. v There are 2000 billion atoms of oxygen in one drop of water (double that for hydrogen)!

Atoms are held together by a chemical bond. A molecule is a group of atoms that are joined together and act as a single unit. For example, H 2 O is made up of 2 hydrogen atoms and 1 oxygen atom and is VERY different from hydrogen alone or oxygen alone. There are 3 atoms in one molecule of water.

4 states of matter: Solids Liquids Gasses
4 th State of Matter is Plasma. Just be aware of it…

Solids: have a definite volume and a definite shape. will keep that volume and shape in any position and in any container. Particles in a solid are packed very tightly together and stay in fixed positions. These particles do move back and forth slightly—like balls connected by tight springs.

Crystalline solids-particles form regular, repeating patterns. e. g. salt, sugar, sand. Amorphous solids—no pattern to particles. e. g. Butter, glass, plastic, rubber.


Liquids: have no shape of their own, but do have a definite volume. takes shape of container. Particles in a liquid are packed tightly together but they move around each other freely.

Viscosity: - the resistance of a liquid to flowing. More viscous = more “thick”.

Gases: have no definite shape or volume. can change volume easily. Particles in a gas fly at high speeds in all directions, filling all the space available to them. They can be spread out to “fill” any space or they can compact together in a smaller space.
Draw this

Why Do Different Phases Exist? Behavior of Molecules …but they are always in motion, constantly bumping into one another

So How Do We Classify Them? How fast the molecules move!!! Solids Liquids Molecules move slow and vibrate in place Move faster than solids and overcome some attraction Gases Plasmas Move fast Move and incredibly overcome fast but all of break apart attraction

5 Major Changes of State • Freezing • Melting • Boiling • Evaporation • Sublimation

Freezing: The change of state from liquid to solid As liquids cool their particles begin to slow, bringing them closer together forming crystals

Melting Melting: The change of state from solid to liquid As a substance absorbs heat energy the objects particles begin to heat up and move faster and farther away from one another

Boiling Boiling: process that occurs when vaporization takes place inside a liquid as well as on the surface Dependant on how strongly the particles in an object are held together

Evaporation Evaporation: occurs when a liquid acquires enough energy to become a gas only on the surface of the liquid
Condensation Condensation: Occurs when a gas loses enough thermal energy to become a liquid Particles in a gas lose energy and begin to move slower, coming closer together

Sublimation Sublimation: occurs when the surface particles of a solid gain enough energy to become a gas

Demo Dry Ice? ? ?

What happens when things melt, boil, etc. ? During a phase change, Thermal Energy is either absorbed or released Solid to Liquid to Gas to Liquid to Solid

What happens to substances when they are When objects are heated. . . they heated or cooled? • When objects are cooled. . . they ***Water is the only exception. . . it actually expands as it cools

Change of State Graph What changes occur as you slowly heat a beaker of ice from – 10 to 110 degrees Celsius?

Phet demonstration https: //phet. colorado. edu/sims/html/states-ofmatter/latest/states-of-matter_en. html

Demos Bell Jar Demo (water) Water bottle Demo (maybe)

Characteristics Properties Of matter A properties - is a characteristic that remains the same. Examples: water freezes at 32 degrees boils at 212 Jenna has brown hair.

2 Types: Physical Changes – alter the form of a substance Boiling Point Melting point Chemical Changes – one or more substances combine or break apart to form new substance (the chemical formula changes) Wood being burnt. Burning in general

Demos: physical or chemical Rip paper Soda Pop Can Gummy bear and potassium chlorate Balloon in bell jar Shaving crème in bell jar Marshmallow in bell jar Salt or sugar in water Mix sugar and salt Starch in water temperature decrease? ? Iodine in starch Coke and milk Na. OH and phenophalline Tums and acetic acid


Are you seeing pysical change or chemical reaction with these changes?

- Slides: 44