25 November 2015 Todays Title Miscible or Immiscible

25 November 2015 Today’s Title: Miscible or Immiscible? Aims: • Define the terms immiscible and miscible (f) • Understand the difference between miscible and immiscible substances (f) • Describe how immiscible and miscible liquids can be separated (f) RWCM: write notes to describe how liquids can be separated To do: What do these words mean? • Miscible • Immiscible • Fractional distillation

How do you separate mixtures of liquids? Write down how you would separate out oil and vinegar. You have 3 minutes.

Immiscible • Immiscible means that the liquids do not dissolve in each other – oil and water are an example. • Stick in your diagram of the separating funnel. Describe in your own words how oil and water can be separated. Oil and water can be separated using a funnel

What have we learnt so far? 1. What does immiscible mean? a) Does dissolve b) Doesn't dissolve c) Will catch fire 2. Which would be an example of an immiscible mixture? a) Oil and water b) Liquid air Use whiteboards c) Food dye to show your answers

What do you think a miscible liquid is? Write a definition Use whiteboards to show your answers

Miscible • Miscible liquids are liquids that dissolve in each other to make a solution. • Miscible liquids are often separated using fractional distillation. • This is possible as miscible liquids have different boiling points.
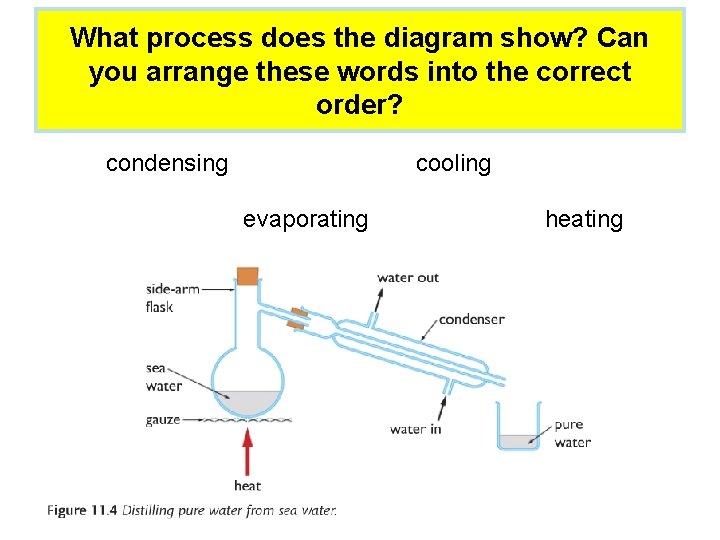
What process does the diagram show? Can you arrange these words into the correct order? condensing cooling evaporating heating
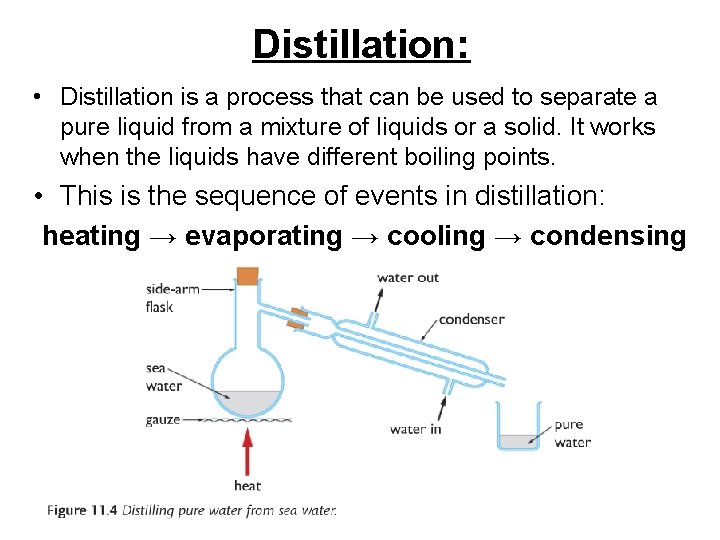
Distillation: • Distillation is a process that can be used to separate a pure liquid from a mixture of liquids or a solid. It works when the liquids have different boiling points. • This is the sequence of events in distillation: heating → evaporating → cooling → condensing

Fractional distillation • Remember: Distillation is a process that can be used to separate a pure liquid from a mixture of liquids. • Fractional distillation takes advantage of the fact that different liquids will have different boiling points • heating → evaporating → cooling → condensing
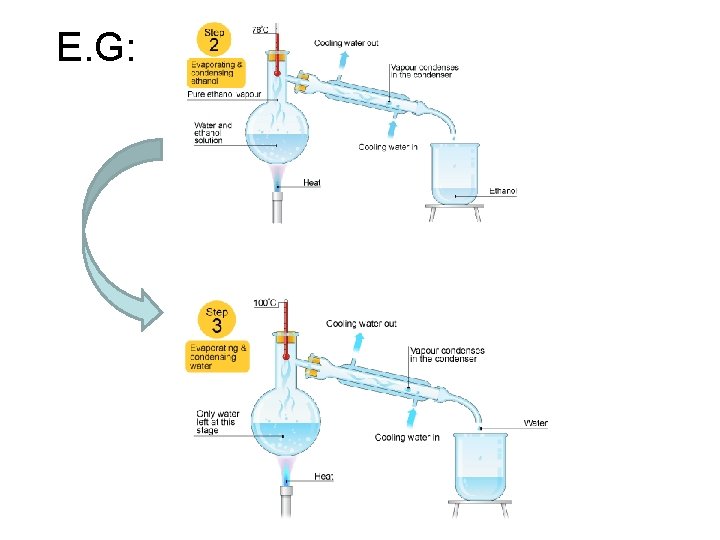
E. G:

Freezing and boiling points of air States of matter: • Gas • Liquid • Solid hot cold Freezing and boiling points of gases in air Water Freezes at 0 o. C Carbon dioxide Freezes at -79 o. C Oxygen Boils at -183 o. C Nitrogen Boils at -196 o. C
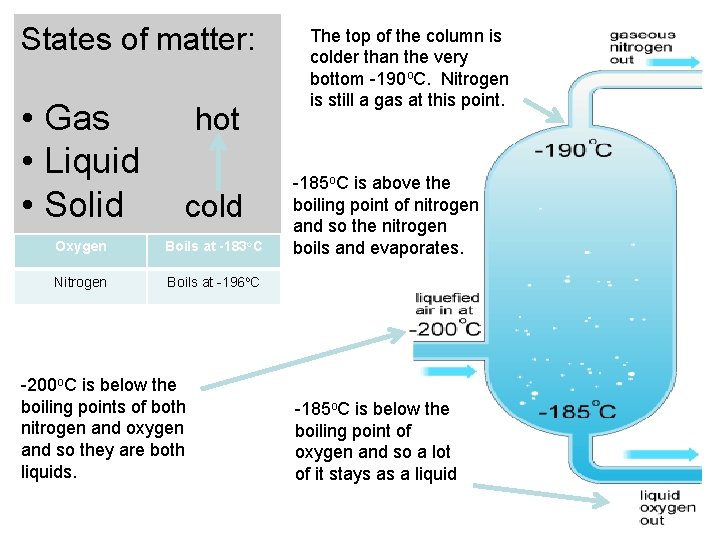
States of matter: • Gas • Liquid • Solid cold Oxygen Boils at -183 o. C Nitrogen Boils at -196 o. C hot -200 o. C is below the boiling points of both nitrogen and oxygen and so they are both liquids. The top of the column is colder than the very bottom -190 o. C. Nitrogen is still a gas at this point. -185 o. C is above the boiling point of nitrogen and so the nitrogen boils and evaporates. -185 o. C is below the boiling point of oxygen and so a lot of it stays as a liquid

Use the help list below to label the diagram on fractional distillation of air • AIR IN • GASEOUS N 2 COLLECTED • -200 o. C • FRACTIONATING COLUMN • H 2 O AND CO 2 FREEZE • -185 o. C ABOVE BOILING POIONT N 2 SO BOILS AND EVAPORATES • O 2 CONDENSES • LIQUID O 2 PIPED OUT Swap sheets with someone else in the classroom to peer ass when you are finished • -185 o. C below BOILING POINT O 2 SO MOST STAYS LIQUID • -190 o. C ALL O 2 LIQUID SO FALLS TO BOTTOM OF COLUMN

Plenary 1. Which of the following liquefies at the lowest temperature? a) b) c) Nitrogen Oxygen Carbon dioxide 2. What is the name of the process that separates air into the different gases? a) b) c) Cracking Fractional distillation Chromatography 3. What piece of equipment can be used to separate immiscible liquids? a) b) c) Evaporating basin Filter funnel Separating funnel 4. What property allows mixtures to be separated by fractional distillation? a) Density b) Boiling point c) Type of bonding
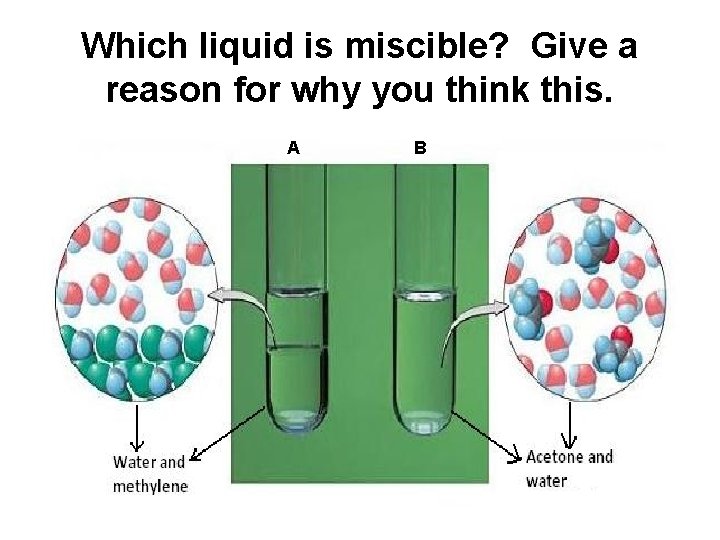
Which liquid is miscible? Give a reason for why you think this. A B

Aims: Extension work Explain how we can separate oil from vinegar and oxygen from air. Make sure that you include the words fractional distillation, immiscible, miscible and separating funnel in your answer (6 marks). Define the terms immiscible and miscible (f) Describe how immiscible and miscible liquids can be separated (f) Understand the difference between miscible and immiscible substances (f)
Definitions Vector Scalar

What have we learnt so far? Speed Velocity Temperature Acceleration Distance Displacement Area Force Volume Air resistance Weight Vector Aims: Sort vectors from scalars(f) Explain the difference between vectors and scalars(h) Scalar

What have we learnt? 1. How would you tell the difference between a miscible and a immiscible liquid? (f) 2. How could you separate out miscible liquids? Can you explain how this works? (a) Aims: Sort vectors from scalars(f) Make an electricity recap poster (f/h) Explain the difference between vectors and scalars(h)
- Slides: 20