Thermochemistry Forms of Energy Energy Conversions Heat Temperature
















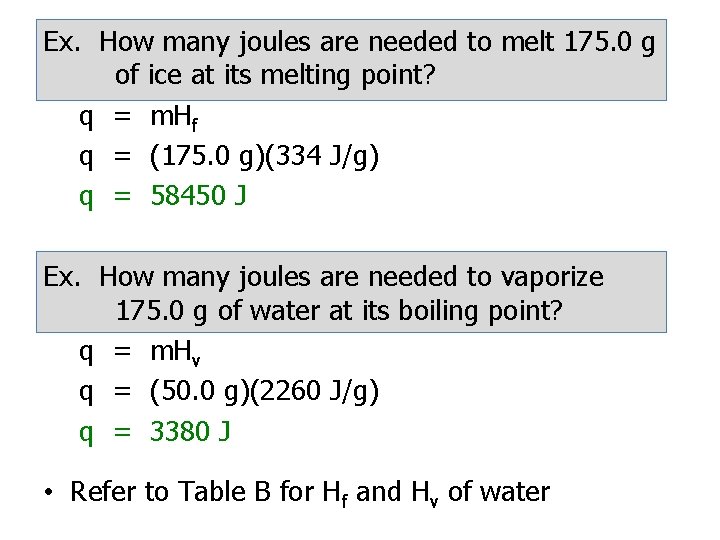




































- Slides: 65

Thermochemistry Forms of Energy & Energy Conversions Heat & Temperature Scales

Energy • Energy is the capacity to do work • Measured in Joules (J) or calories (cal)

There are 2 Classes of Energy: 1. Potential Energy (stored energy) 2. Kinetic Energy (energy of motion)

Energy Comes in Many Forms Sound Energy Electromagnetic Energy Electrical Energy Chemical Energy Nuclear Energy Heat Energy Mechanical Energy Magnetic Energy

Law of Conservation of Energy • States that “Energy can be neither created nor destroyed. ” • Energy can be changed from one form to another • The total amount of energy remains the same during energy conversions. • In chemistry, energy can be measured in terms of the amount of heat it can be changed into. chemical mechanical radiant chemical mechanical electrical thermal

Heat vs. Temperature RELATED CONCEPTS BUT…NOT THE SAME THING!!! • Heat – Total energy of molecular motion in a substance – Depends on: 1. Speed of particles 2. Number of particles (the size or mass) 3. Type of particles – Flow of heat is measured in Joules or calories • (1 calorie = 4. 18 joules)

Measurement of Heat 1 calorie = the amount of heat energy needed to increase the temperature of 1 gram of a substance by 1 °C • This is called the specific heat capacity – Tells us how much energy is required to heat or cool an object to a specific temperature – Depends on the type of substance Specific heat capacity (J/g-°C) water 4. 18 ethyl alcohol 2. 46 ice 2. 10 aluminum 0. 92 glass 0. 84 iron 0. 45 copper 0. 38 silver 0. 24 lead 0. 13

• Temperature – a measure of the average energy of molecular motion in a substance – NOT a form of energy – Does not depend on the size or type of object – Measured with a thermometer – Ex. • The temperature of a small cup of water might be the same as the temperature of a large bucket of water, but the bucket of water has more heat because it has more water and thus more total thermal energy.

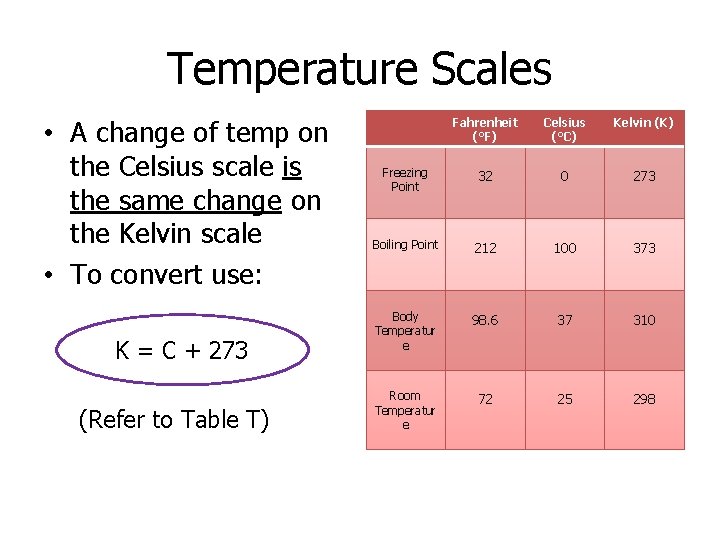
Temperature Scales • A change of temp on the Celsius scale is the same change on the Kelvin scale • To convert use: K = C + 273 (Refer to Table T) Fahrenheit (°F) Celsius (°C) Kelvin (K) Freezing Point 32 0 273 Boiling Point 212 100 373 Body Temperatur e 98. 6 37 310 Room Temperatur e 72 25 298

Heat Changes Temperature Removing heat DECREASES temperature Adding heat INCREASES temperature • Higher temperatures mean that the molecules are moving, vibrating and rotating with more energy.

Heat Transfer • Heat DOES NOT flow between objects with the same temperature • If the temperature of one object is higher than that of another object, there will be a transfer of heat energy from the hotter to the colder object until both objects reach the same temperature.

Heat • Particles of matter are in continuous motion • The faster the particles move, the greater the kinetic energy of the particles • The average kinetic energy of the particles increases as the temperature increases • At “absolute zero” the kinetic energy of the particles would be 0

Endothermic Reactions • When heat enters a body, (endothermic) it may be used to increase the kinetic energy of the particles. • Thus, the temperature will increase. In an endothermic process, the system gains heat as the surroundings cool down.

Exothermic Reactions • When a body cools, it decreases in kinetic energy (exothermic). • This energy may be given off as heat In an exothermic process, the system loses heat as the surroundings heat up.

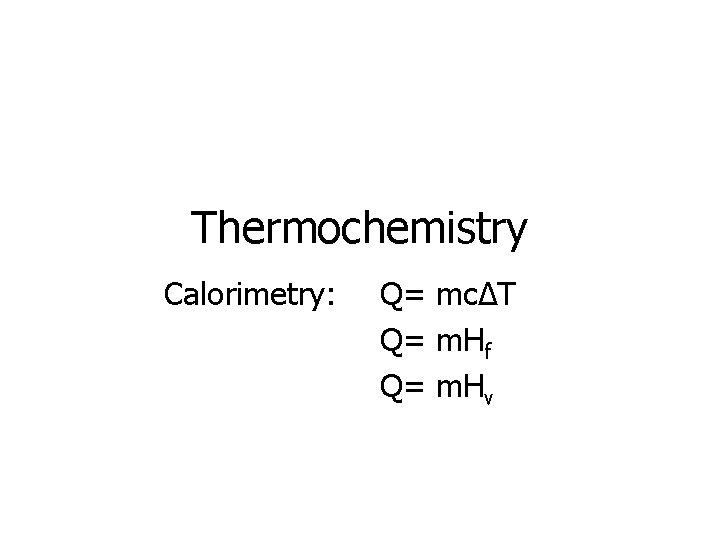
Thermochemistry Calorimetry: Q= mcΔT Q= m. Hf Q= m. Hv

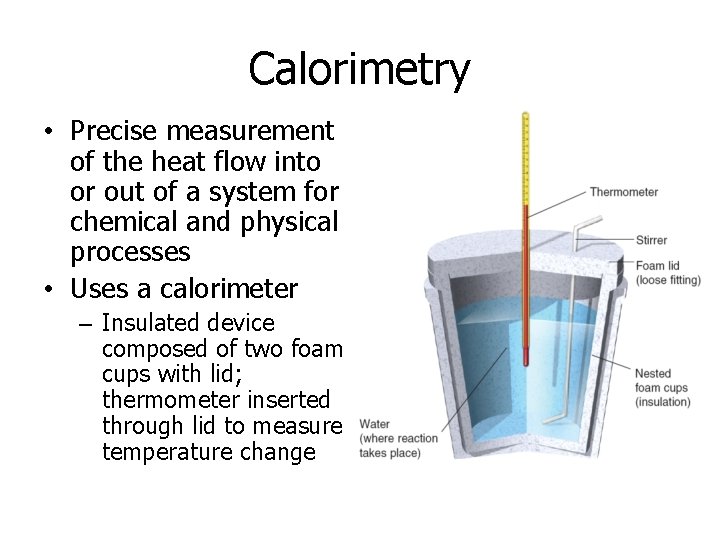
Calorimetry • Precise measurement of the heat flow into or out of a system for chemical and physical processes • Uses a calorimeter – Insulated device composed of two foam cups with lid; thermometer inserted through lid to measure temperature change

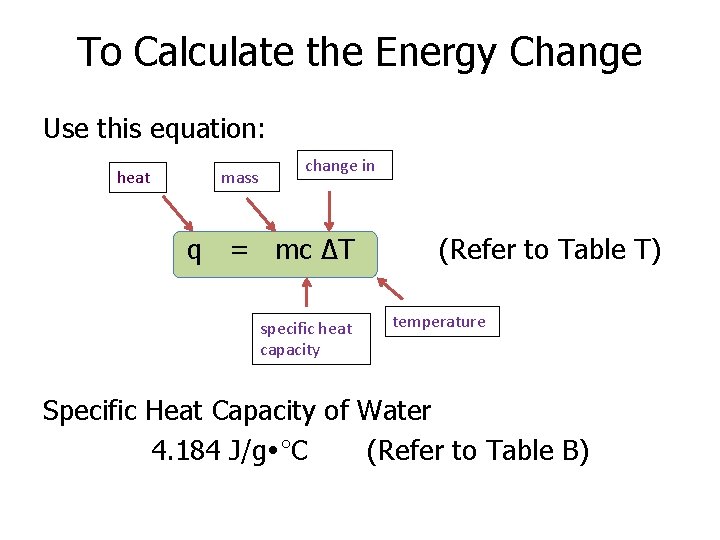
To Calculate the Energy Change Use this equation: heat mass change in q = mc ΔT specific heat capacity (Refer to Table T) temperature Specific Heat Capacity of Water 4. 184 J/g °C (Refer to Table B)

Example: Solving for q Calculate the heat, in Joules, to warm 110 g of water from 25. 0 °C to 26. 2 °C. q = mcΔT q = (110 g)(4. 184 J/g °C)(26. 2 °C – 25. 0 °C) q = 550 J

Try This: Solving for Tf What would be the final temperature if 3. 31 x 103 J were added to 18. 5 grams of water at 22. 0°C?

Try This: Solving for m A sample of lead, specific heat 0. 138 J/g °C, released 1. 20 x 103 J when it cooled from 93. 0°C to 29. 5°C. What was the mass of this sample of lead?

Try This: Solving for c Calculate the specific heat of platinum if 1092 J of heat were released when 125 grams of platinum cooled 65. 2 °C.

Phase Changes • In many instances, a phase change may occur when a substance is heating or cooling. • To calculate the amount of heat needed to melt a substance use: Heat of Fusion: (Hf) q = m. Hf The heat needed to melt 1 g of a substance at its melting point • To calculate the amount of heat needed to vaporize a substance use: Heat of Vaporization: (Hv) q = m. Hv The heat needed to vaporize 1 g of a substance at its boiling point • Refer to Table T
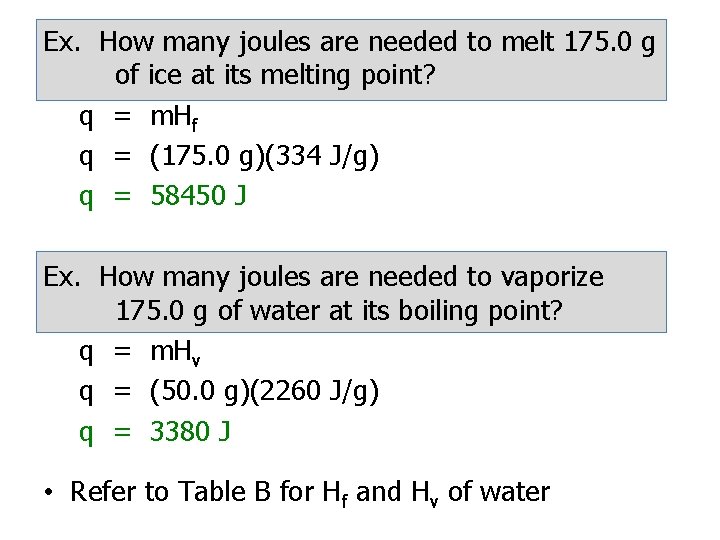
Ex. How many joules are needed to melt 175. 0 g of ice at its melting point? q = m. Hf q = (175. 0 g)(334 J/g) q = 58450 J Ex. How many joules are needed to vaporize 175. 0 g of water at its boiling point? q = m. Hv q = (50. 0 g)(2260 J/g) q = 3380 J • Refer to Table B for Hf and Hv of water

Try This: Think, Think!!! Ex. The temperature of 50. 8 g sample of water increases from 46. 6°C to 105. 5°C. How much heat is absorbed?

Thermochemistry qlost = qgained

qlost = qgained • Heat flows from warmer objects to cooler objects. • The amount of heat lost by one substance is equal to the amount of heat gained by another substance. • Heat will no longer flow once the temperature of both substances is the same. • We are able to calculate the final temperature of the mixture using q = mcΔT.

Calculating the Tf when two substances are mixed together. qlost = qgained We know that q = mcΔT so… - mc(Tf – Ti) = mc(Tf – Ti) (lost) (gained)

Ex. A 50. 5 g sample of water at 68. 5°C is mixed with 22. 8 g of water at 96. 4°C. Calculate the final temperature of the mixture assuming no heat is lost to the containers and surroundings. -mc(Tf – Ti) = mc(Tf – Ti) -(22. 8 g)(4. 184 J/g·°C)(Tf - 96. 4°C) = (50. 5 g)(4. 184 J/g·°C)(Tf - 68. 5°C) -(22. 8)(Tf - 96. 4) = (50. 5)(Tf - 68. 5) - 22. 8 Tf + 2197. 92 = 50. 5 Tf - 3459. 25 73. 3 Tf = 5657. 17 73. 3 Tf = 77. 2°C

Ex. A 230 g iron cube at 100. 0°C is placed in a plastic foam cup containing 575 g of water at 43. 5°C. Calculate the final temperature of the mixture assuming no heat is lost to the containers and surroundings. The specific heat of iron is 0. 46 J/g·°C. -mc(Tf – Ti) = mc(Tf – Ti) -(230 g)(0. 46 J/g·°C)(Tf - 100. 0°C) = (575 g)(4. 184 J/g·°C)(Tf - 43. 5°C) -(105. 8)(Tf - 100. 0) = (2405. 8)(Tf - 43. 5) -105. 8 Tf + 10580 = 2405. 8 Tf - 104652. 3 2511. 6 Tf = 115232. 3 2511. 6 Tf = 46°C

Thermochemistry Phases of Matter Vapor Pressure Phase Diagrams Heating/Cooling Curves

Phases of Matter 1. Solid – – – Definite shape, definite volume; not easily compressed Molecules are close together Strong force of attraction between particles Particles move slightly; vibrate Particles arranged in an orderly, repeating, 3 -dimensional pattern called a crystal lattice

Phases of Matter 2. Liquid – – – Indefinite shape; definite volume; not easily compressed Particles slide about each other Particles move within a confined space

Phases of Matter 3. Gas – – Indefinite shape; indefinite volume; easily compressed Particles moving very quickly Particles take up all space provided Minimal forces of attraction between particles

Changes of Phase 1. Exothermic Phase Changes – Releasing heat during a chemical reaction – Object/substance cools – Condensation: – Freezing: – Deposition: gas liquid solid gas solid

Changes of Phase 2. Endothermic Phase Changes – Absorbing heat during a chemical reaction – Object/substance warms – Melting/Fusion: – Vaporization: (boiling/evaporation) – Sublimation: (CO 2& I 2) solid liquid gas solid gas

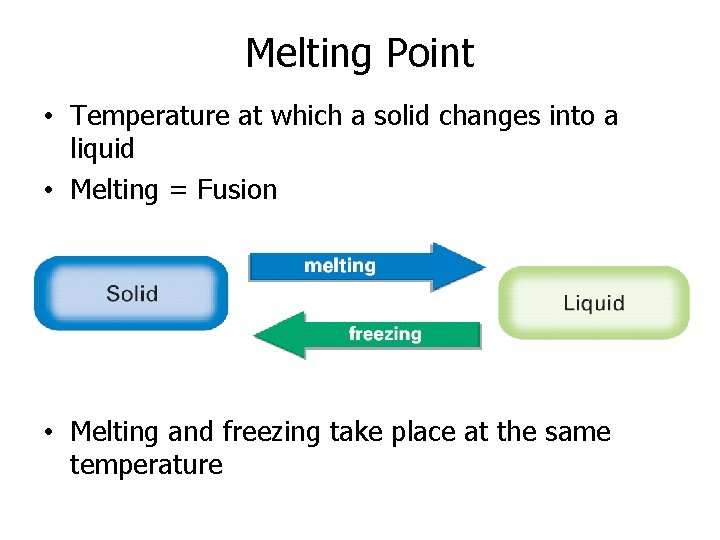
Melting Point • Temperature at which a solid changes into a liquid • Melting = Fusion • Melting and freezing take place at the same temperature

Boiling Point • Temperature at which liquid changes into a vapor (vaporization) • Vaporization and condensation take place at the same temperature

Boiling • When the vapor pressure equals the atmospheric pressure, we can see bubbles of gas forming below the surface of the liquid. • The formation of the bubble is opposed by atmospheric pressure. • All liquids don’t boil at the same temperature

• We can boil water at any temperature by changing the air pressure. 1. At a lower external pressure, the boiling point decreases. Ex. Mile High Stadium Lower air pressure, water boils at lower temperature – slower cooking (need to cook food longer) 2. At a higher external pressure, the boiling point increases. Ex. Pressure Cooker Greater pressure, water boils at higher temperature - faster cooking (need to cook food less time)

Vaporization vs. Evaporation • Evaporation – When a conversion from liquid to vapor occurs at the surface of a liquid that is not boiling – Only particles at the surface of the sample have enough kinetic energy to change to a vapor • Vaporization – If the substance is boiling, all particles in the sample with enough kinetic energy would change to a vapor

Open Vs. Closed System • In an open container, molecules that evaporate can escape from the container. • In a closed container, the molecules cannot escape. They collect as a vapor above the liquid. Some molecules condense back into a liquid.

Vapor – Liquid Equilibrium • In a system at constant vapor pressure, a dynamic equilibrium exists between the vapor and the liquid. • The system is in equilibrium because the rate of evaporation of liquid equals the rate of condensation of vapor.

Vapor Pressure • A measure of the force exerted by a vapor above a liquid when it’s in equilibrium with its liquid • Amount of vapor pressure produced by a liquid depends on: 1. Temperature of the liquid (Higher the temp the higher the vapor pressure) 2. Nature of the substance (Weak intermolecular forces of attraction high vapor pressure

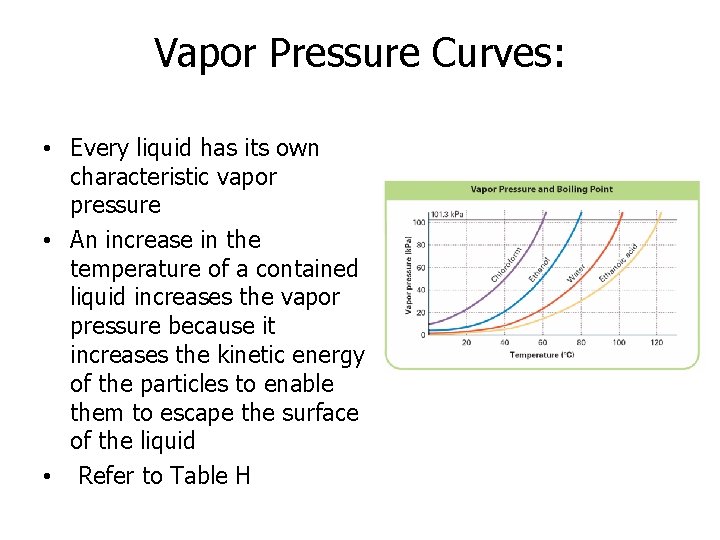
Vapor Pressure Curves: • Every liquid has its own characteristic vapor pressure • An increase in the temperature of a contained liquid increases the vapor pressure because it increases the kinetic energy of the particles to enable them to escape the surface of the liquid • Refer to Table H

Vapor Pressure and Temperature Change

Normal Boiling Point • Because a liquid can have various boiling points depending on pressure, the normal boiling point is defined as the boiling point of a liquid at a pressure of 101. 3 k. Pa. • It means at 100°C, the vapor pressure of water equals 101. 3 k. Pa. (101. 3 k. Pa = 1 atm = 760 mm Hg = 760 torres) • Higher normal boiling point = stronger IMF

Sublimation • The change of a substance from a solid to a vapor without passing through the liquid state • Occurs in solids with vapor pressures that exceed atmospheric pressure at or near room temperature.

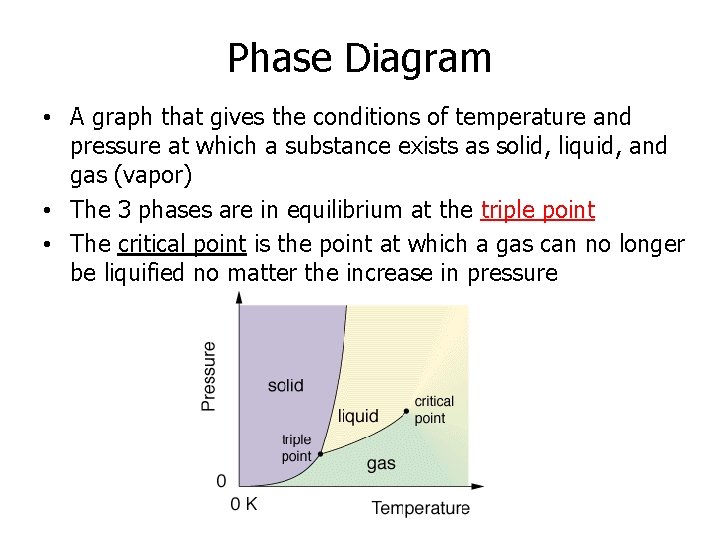
Phase Diagram • A graph that gives the conditions of temperature and pressure at which a substance exists as solid, liquid, and gas (vapor) • The 3 phases are in equilibrium at the triple point • The critical point is the point at which a gas can no longer be liquified no matter the increase in pressure

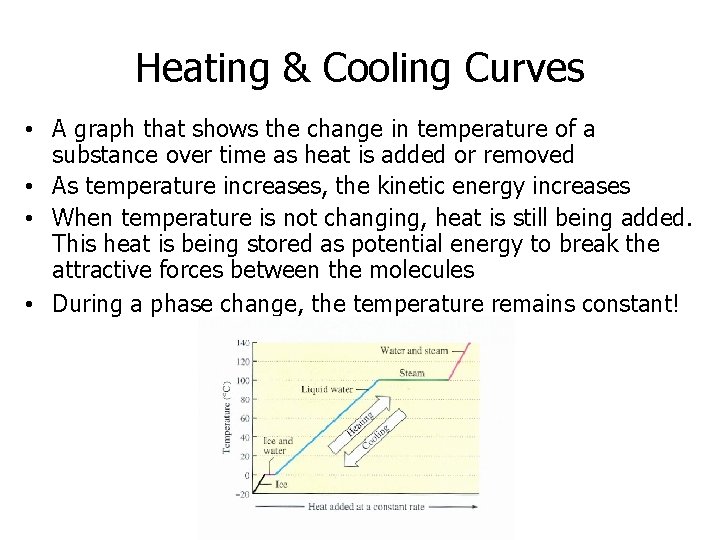
Heating & Cooling Curves • A graph that shows the change in temperature of a substance over time as heat is added or removed • As temperature increases, the kinetic energy increases • When temperature is not changing, heat is still being added. This heat is being stored as potential energy to break the attractive forces between the molecules • During a phase change, the temperature remains constant!

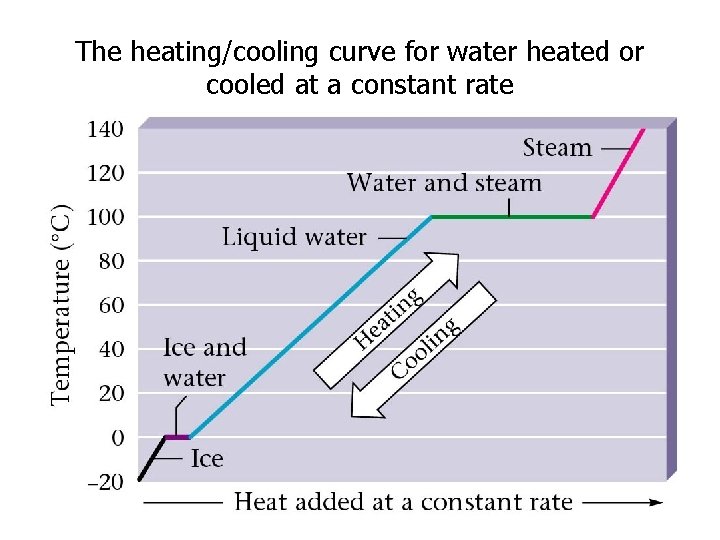
The heating/cooling curve for water heated or cooled at a constant rate

Thermochemistry Calculating ΔH Using Table I

Chemical Reactions • Any reaction, chemical or physical, involves either the absorption or release of energy. • The energy is: – measured in the form of heat • expressed in k. J (or J) – Called enthalpy or heat of reaction (ΔH)

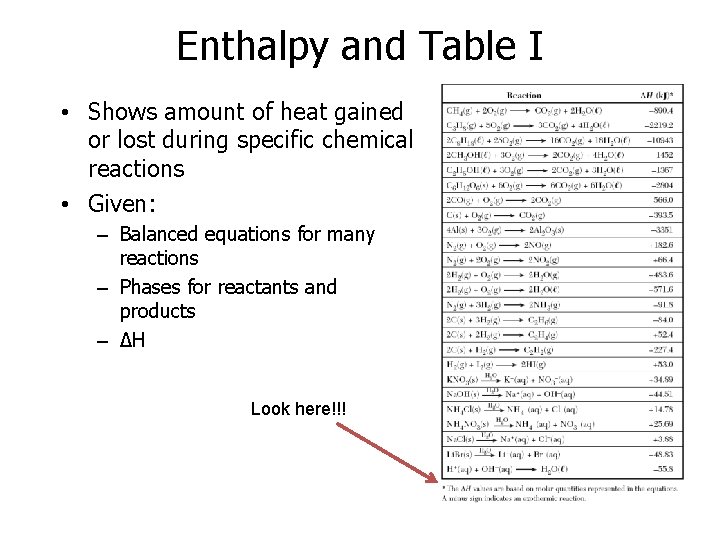
Enthalpy and Table I • Shows amount of heat gained or lost during specific chemical reactions • Given: – Balanced equations for many reactions – Phases for reactants and products – ΔH Look here!!!

Exothermic Reactions • Indicated by a negative ΔH • When included in the chemical equation, heat is included on the product side • Ex. The first exothermic reaction on the table would be written as: CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) + 890. 4 k. J • PE of products is lower than that of reactants

Endothermic Reactions • Indicated by a positive ΔH • When included in the chemical equation, heat is included on the reactant side • Ex. The first endothermic reaction on the table would be written as: N 2(g) + O 2(g) + 182. 6 k. J 2 NO(g) • PE of products is greater than that of reactants

Thermochemistry Calculating ΔHo(rxn) Using ΔHf

Calculating ΔHo Using ΔHf • Standard enthalpy of formation (ΔHf) – change of enthalpy that accompanies the formation of 1 mole of a substance from its constituent elements – For a(n)element in its basic state, ΔHf is 0 • This includes all of the diatomics – Refer to Standard Enthalpies of Formation Table for values (NOT on Chemistry Reference Tables) • Standard enthalpy of reaction (ΔHorxn) – difference between the standard enthalpies of formation of the products and the reactants – To calculate, use the following equation: ΔHo(rxn) = ΣHof(products) - ΣHof(reactants)

Ex. Using the standard enthalpies of formation, calculate the ΔHorxn for the following reaction: CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) ΔHo(rxn) = ΣHof(products) - ΣHof(reactants) ΔHo(rxn) = [(-393. 5 k. J + 2(-285. 8 k. J)] - [-74. 8 k. J + 2(0 k. J)] = (-965. 1 k. J) – (-74. 8 k. J) = -890. 3 k. J

Try These! 1) Na. OH(s) + HCl(g) Na. Cl(s) + H 2 O(g) 2) 2 NO(g) + O 2(g) 2 NO 2(g) 3) 2 H 2 S(g) + 3 O 2(g) 2 H 2 O(l) + 2 SO 2(g)

Thermochemistry Hess’s Law

Hess’s Law States that when going from a particular set of reactants to a particular set of products, the change in enthalpy (ΔH) is the same whether the reaction takes place in one step or in a series of steps.

Example: The overall reaction can be written as: N 2(g) + 2 O 2(g) 2 NO 2(g) ΔH= 68 k. J OR The reaction can be carried out in two steps where the sum of the ΔH’s for each reaction is equal to the ΔH for the overall reaction. N 2(g) + O 2(g) 2 NO(g) ΔH= 180 k. J 2 NO(g) + O 2(g) 2 NO 2(g) ΔH= -112 k. J --------------------------N 2(g) + 2 O 2(g) 2 NO 2(g) ΔH= 68 k. J

Important to Remember The chemical equations given to you to solve the problem MAY NOT be exactly how you need them. So… 1. If a reaction is reversed, the sign of ΔH is also reversed. 2. If the coefficients in a balanced reaction are multiplied/divided by an integer, the value of ΔH is multiplied/divided by the same integer.

Try This! Calculate ΔH for the following reaction using the information provided. S(s) + O 2(g) SO 2(g) Given: S(s) + 3/2 O 2(g) SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3(g) ΔH= -395. 2 k. J ΔH= -198. 2 k. J

Try This! Calculate ΔH for the following reaction using the information provided. Fe. O(s) + CO(g) Fe(s) + CO 2(g) Given: Fe 2 O 3(s) + 3 CO(g) 2 Fe(s) + 3 CO 2(g) ΔH= -23 k. J 3 Fe 2 O 3(g) + CO(g) 2 Fe 3 O 4(s) + CO 2(g) ΔH= -39 k. J Fe 3 O 4(s) + CO(g) 3 Fe. O(s) + CO 2(g) ΔH= +18 k. J