Energy Forms and Energy Conversion Energy The capacity














- Slides: 27

Energy Forms and Energy Conversion

Energy • The capacity of vigorous activity • The ability to act • The capacity of a body or a system to do work (and heat).

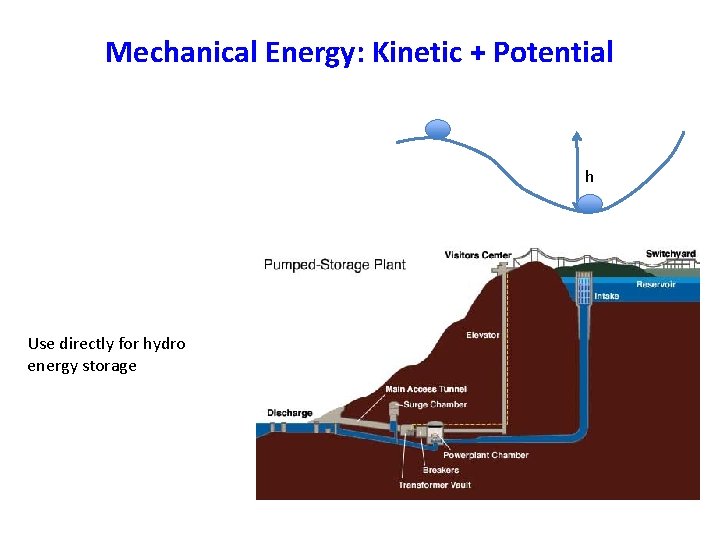
Mechanical Energy: Kinetic + Potential h Use directly for hydro energy storage

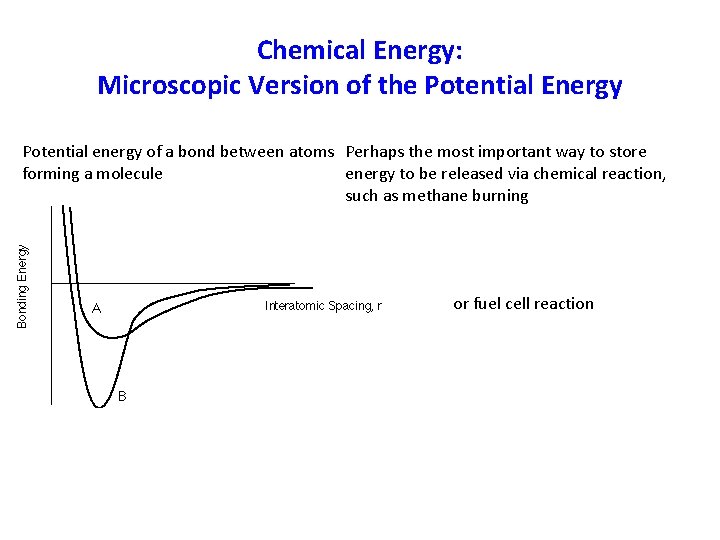
Chemical Energy: Microscopic Version of the Potential Energy Potential energy of a bond between atoms Perhaps the most important way to store forming a molecule energy to be released via chemical reaction, such as methane burning or fuel cell reaction

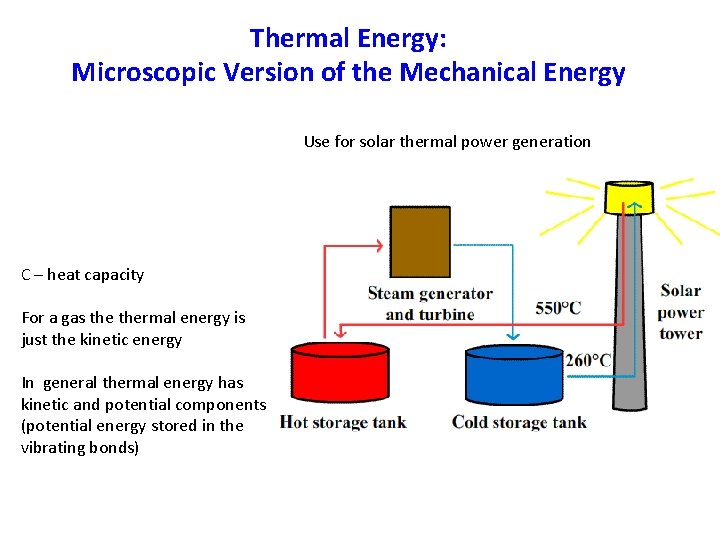
Thermal Energy: Microscopic Version of the Mechanical Energy Use for solar thermal power generation C – heat capacity For a gas thermal energy is just the kinetic energy In general thermal energy has kinetic and potential components (potential energy stored in the vibrating bonds)

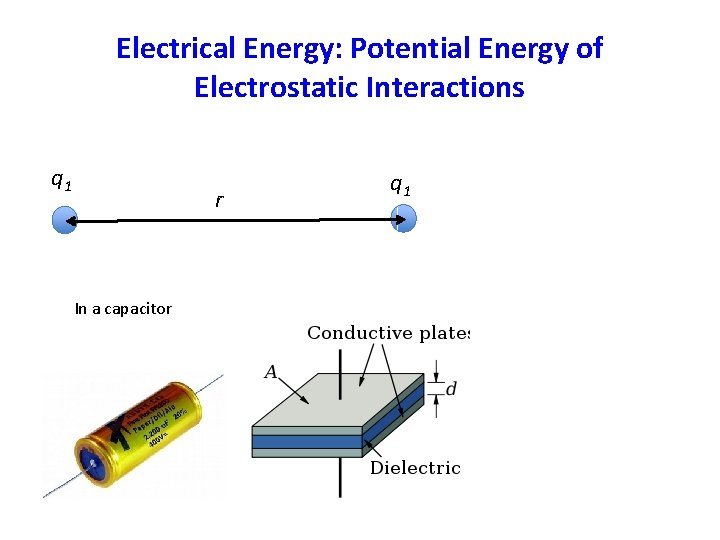
Electrical Energy: Potential Energy of Electrostatic Interactions q 1 r In a capacitor q 1

Electromagnetic Energy in oscillating electromagnetic waves These waves can be considered as massless particles called photons traveling with the speed of light Photon energy hv (h –Planck constant, v frequency) Power generator turns chemical into mechanical into electromagnetic, into electric current

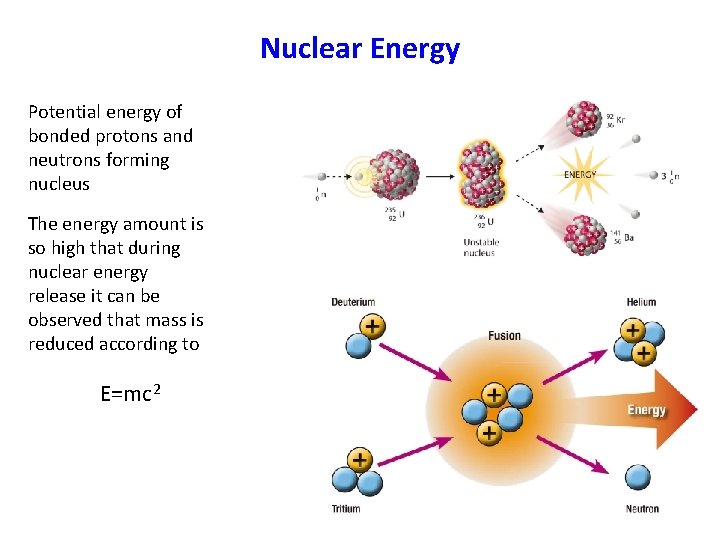
Nuclear Energy Potential energy of bonded protons and neutrons forming nucleus The energy amount is so high that during nuclear energy release it can be observed that mass is reduced according to E=mc 2

Energy Conservation Energy can not be created or destroyed – it can be only converted Energy of the system can be changed by the flow of energy form or to the outside in the form of work, W, and heat, Q ΔE = W + Q

Work = force times displacement W = Fd The unit of work is the same as the unit of energy The unit of force thus the unit of energy This unit is called Joule

Heat, as work is also a process – it is essentially work done by molecules thus it can change the energy of the system

Energy and Power is the energy change rate W = ΔE/Δt The unit of power is Watt (W) The World uses about 15 Terra Watts of power, with about 2. 5 TW of electricity

Energy Units - I Joule = Watt x second Kilowatt-hour = 3. 6 Megajoules– on your electric bill Therm = 105 Megajoules = 100, 000 British thermal units (BTU) – about 100 cubic feet natural gas burning equivalent BTU – energy needed to heat one pound of water by one °F

Energy Units - II Calorie - heating 1 g of water by 1 °C = 4. 2 Joules Electronovolt – energy gained by moving one electron across potential difference of one Volt = 1. 6 x 10 -19 Joules (one Volt = 1 J/Coulomb) Energy = Volt x Coulomb = Volt x Ampere x time Power = Volt x Ampere

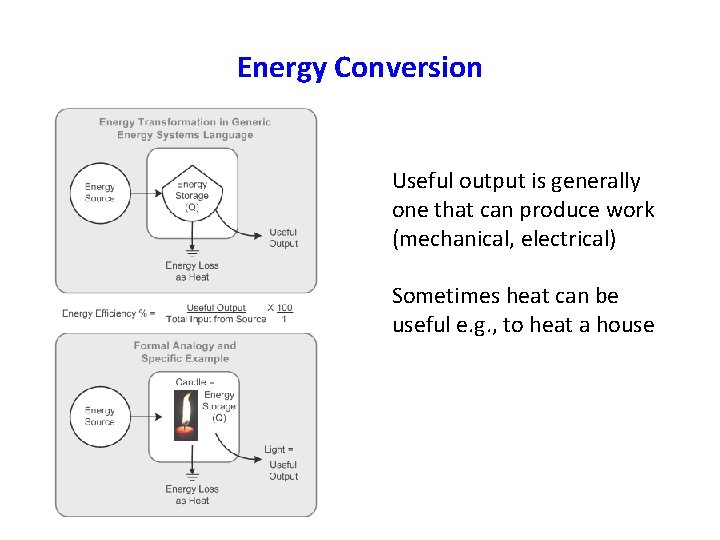
Energy Conversion Useful output is generally one that can produce work (mechanical, electrical) Sometimes heat can be useful e. g. , to heat a house

Chemical Energy Conversion Most important is combustion – potential chemical energy is transformed to kinetic energy of the gas (high temp and pressure). For example, Yields about 240 k. J/mole energy leading to a high pressure gas. This energy shows as the kinetic energy This energy can be used directly as mechanical energy (car) or converted to other useful energy, such as electrical energy

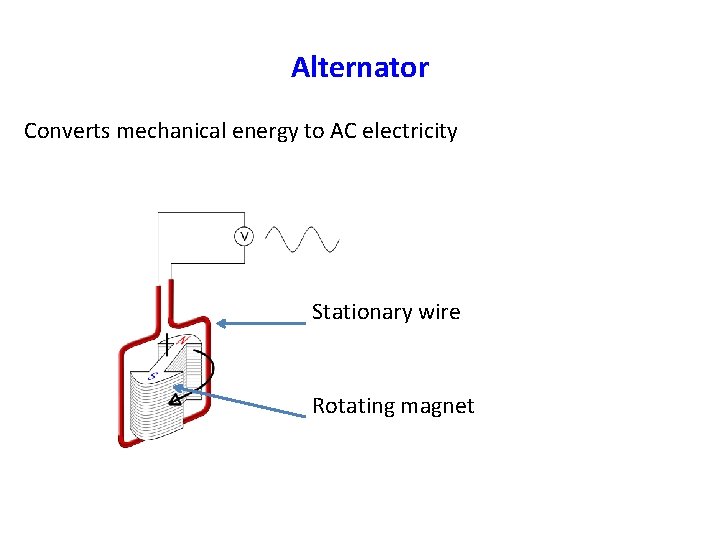
Alternator Converts mechanical energy to AC electricity Stationary wire Rotating magnet

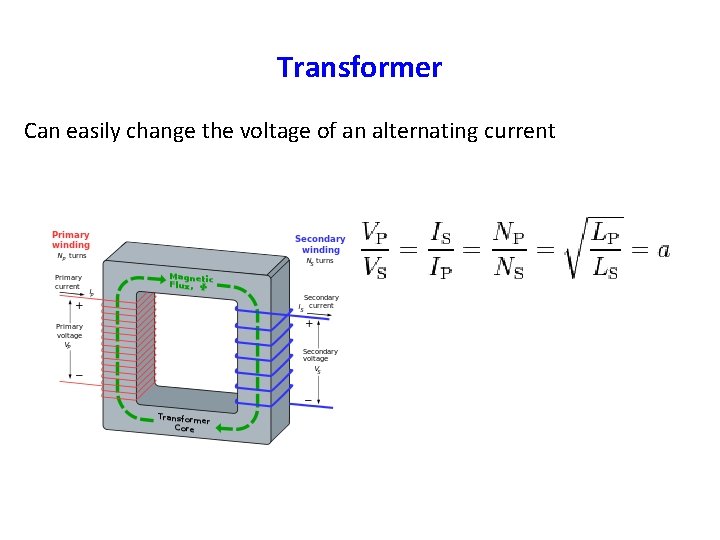
Transformer Can easily change the voltage of an alternating current

Why AC won over DC AC transformers can easily increase and decrease voltage The input power is P=VI (voltage times current) The voltage on a transmission wire is V = IR The power loss on the transmission wire is Ploss = I 2 R, and since I=P/V, Ploss =R P 2/V 2 Higher voltage means much lower transmission loss This was a rare time that Thomas Edison lost big time to Tesla and Westinghouse (War of Currents).

AC vs. DC in the future Distributed power generation, e. g. , by solar, allows to avoid transmission power losses due to short lines. Transformation losses are eliminated Many devices work on DC only So Edison might be right in the future

Solar to Electricity Photon excites negatively charged electrons and separates them from positively charged holes – this generates electrostatic energy and associated voltage (DC) Limits of efficiency is mostly due to two factors Not all photons lead to excitation Electrons and holes recombine and just generate heat

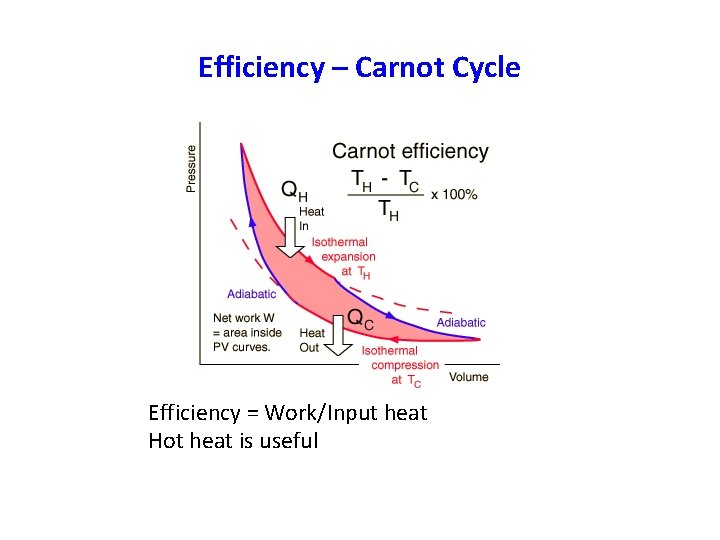
Efficiency – Carnot Cycle Efficiency = Work/Input heat Hot heat is useful

Efficiency and Thermal Energy Storage Carnot conversion efficiency (Thot-Tcold)/Thot*100% Increases with increasing Thot But stored heat radiates at the rate ~ (Thot)4 thus the loss increases with increasing Thot

Efficiency in General Efficiency = Useful Energy/Energy Input Typical fossil fuel power plant up to 40% Hydro – about 90% Solar 6 -40%, 15% typical Combustion engine ~ 30% Electric engine 70 -90% Photosynthesis few percent

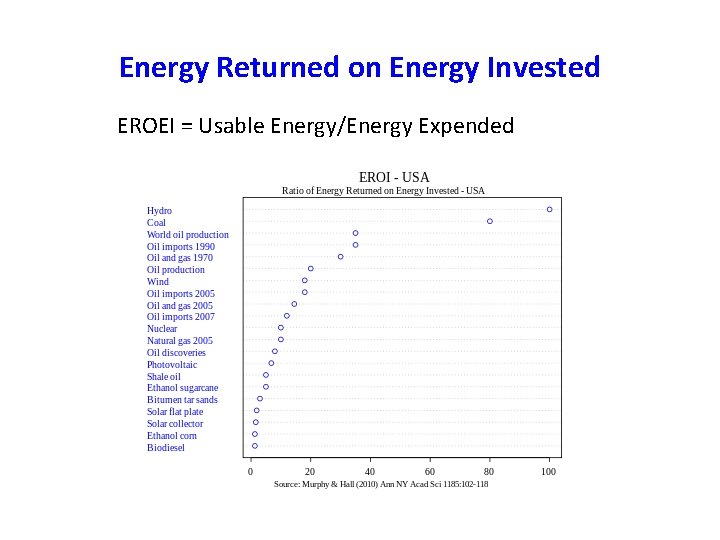
Energy Returned on Energy Invested EROEI = Usable Energy/Energy Expended

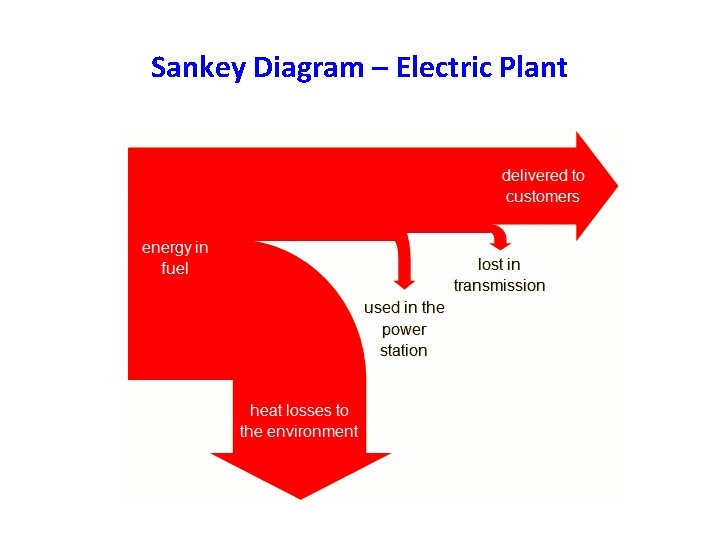
Sankey Diagram – Electric Plant

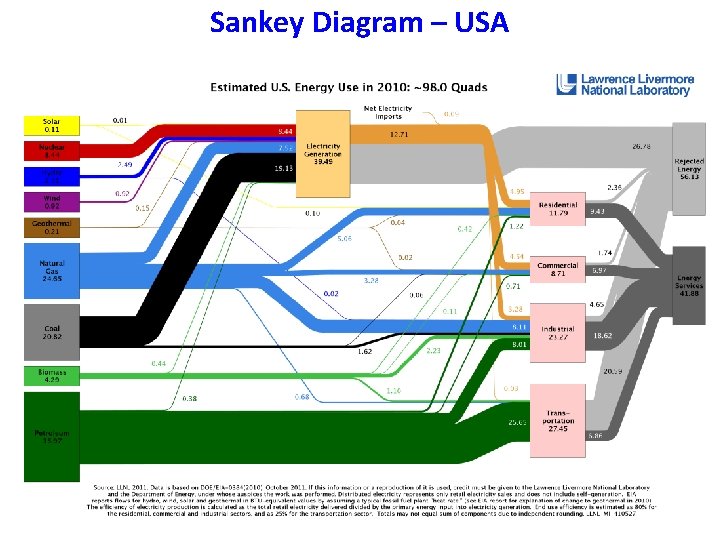
Sankey Diagram – USA