The Chemical Level of Organization Unit I Organization























- Slides: 39

The Chemical Level of Organization Unit I. Organization of the Human Body

Essential Concepts • Matter is composed of atoms held together by chemical bonds • During a chemical reaction, bonds are formed, rearranged, or broken • Water is the most important and abundant inorganic compound in the body • Carbohydrates, lipids, proteins, nucleic acids, and ATP are the most important organic compounds in the body

Chemical Elements HOW MATTER IS ORGANIZED

Matter • anything that occupies space and has mass Mass • the amount of matter in an object; constant Weight • The force of gravity on an object; variable

Chemical Elements • Matter exists in three states: – Solids – Liquids – Gases • All matter is composed of chemical elements • Elements - the building blocks of matter

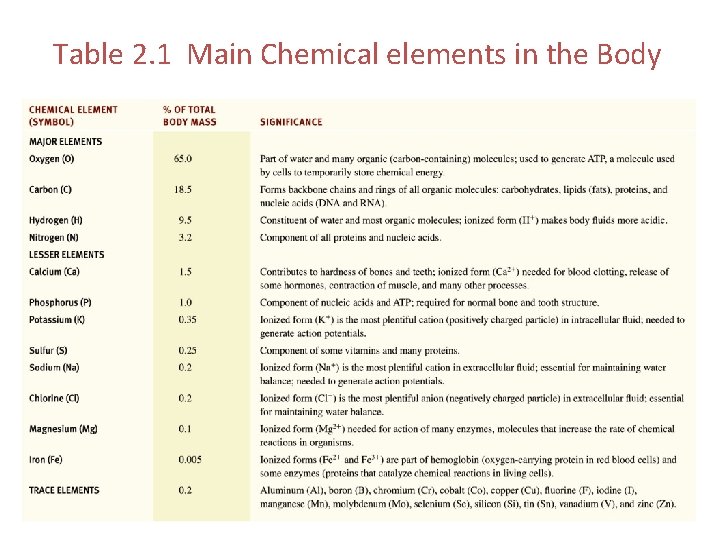
Major Elements Carbon Hydrogen Oxygen Nitrogen

Minor Elements Calcium Phosphorus Potassium Sulfur Sodium Chlorine Magnesium Iodine Iron

Trace Elements • Examples include copper and zinc – Copper works with iron to form RBCs. Keeps vessels, nerves, bones, immune system healthy. – Zinc is necessary for immunity. Helps with mitosis, interphase and healing. Needed for smell and taste.

Table 2. 1 Main Chemical elements in the Body

Atomic Structure HOW MATTER IS ORGANIZED

Atoms • Atom – the smallest unit of matter • An element contains the same kind of atoms • Example: a pure sample of the element carbon contains only carbon atoms

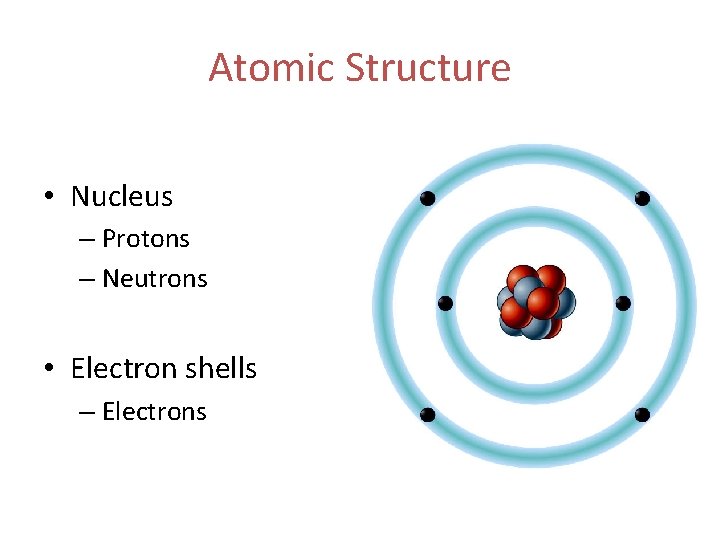
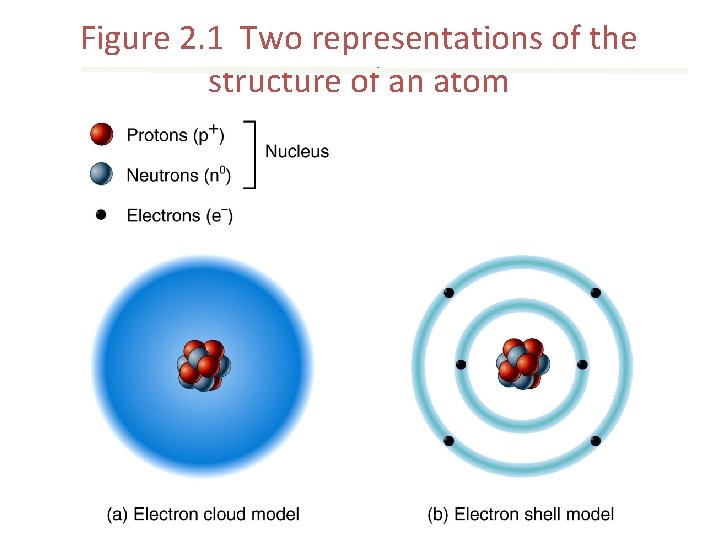
Atomic Structure • Nucleus – Protons – Neutrons • Electron shells – Electrons

Figure 2. 1 Two representations of the structure of an atom

Atomic Structure Protons • Positively charged particles Neutrons • Neutrally charged particles Electrons • Negatively charged particles

Atomic Charge • The number of protons in an atom is equal to the number of electrons • Therefore, atoms have no charge • Example: Oxygen – 8 protons are balanced by 8 electrons

Atomic Number and Mass Number HOW MATTER IS ORGANIZED

Atomic Number and Atomic Mass Atomic number • the number of protons in an atom’s nucleus Mass number • the sum of the number of protons and neutrons in an atom’s nucleus

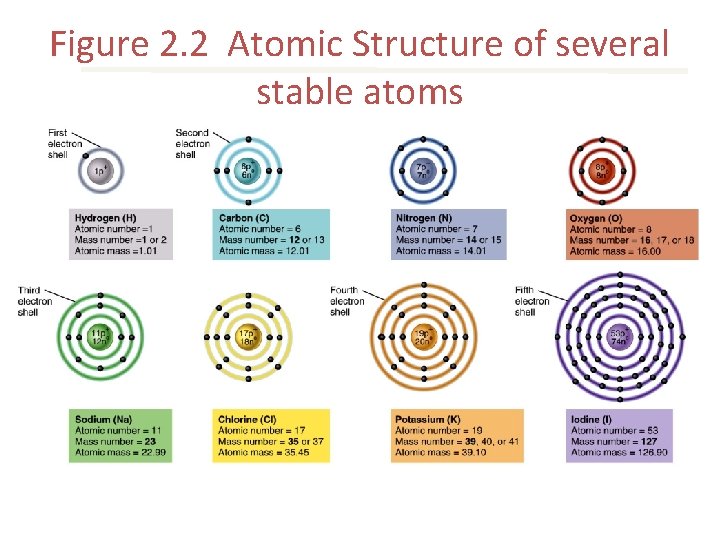
Figure 2. 2 Atomic Structure of several stable atoms

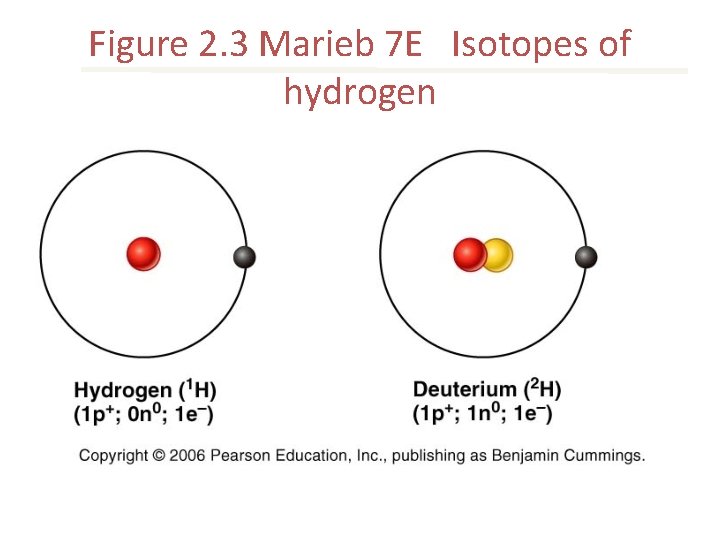
Isotopes • atoms of an element that have different numbers of neutrons and therefore different mass numbers Radioactive isotopes • unstable isotopes that emit radiation as they decay

Figure 2. 3 Marieb 7 E Isotopes of hydrogen

Ions, Molecules, and Compounds HOW MATTER IS ORGANIZED

Ions • Ion - forms when an atom loses or gains electrons

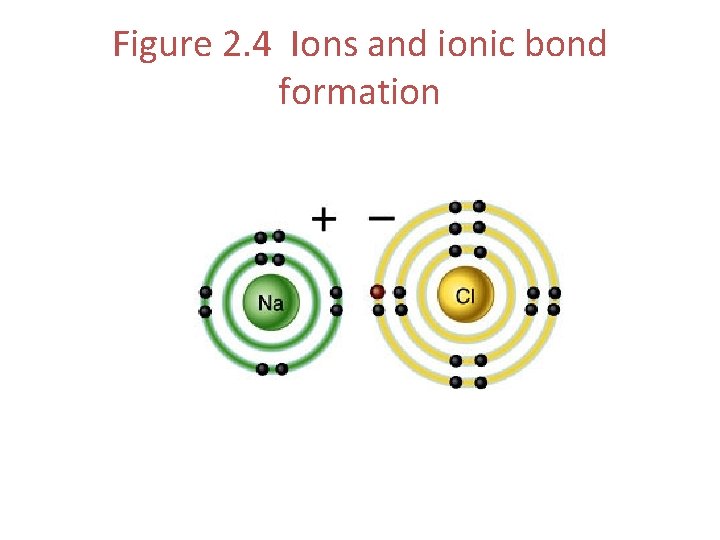
Ions Cation • An atom that has lost electrons • Positively charged Anion • An atom that has gained electrons • Negatively charged

Figure 2. 4 Ions and ionic bond formation

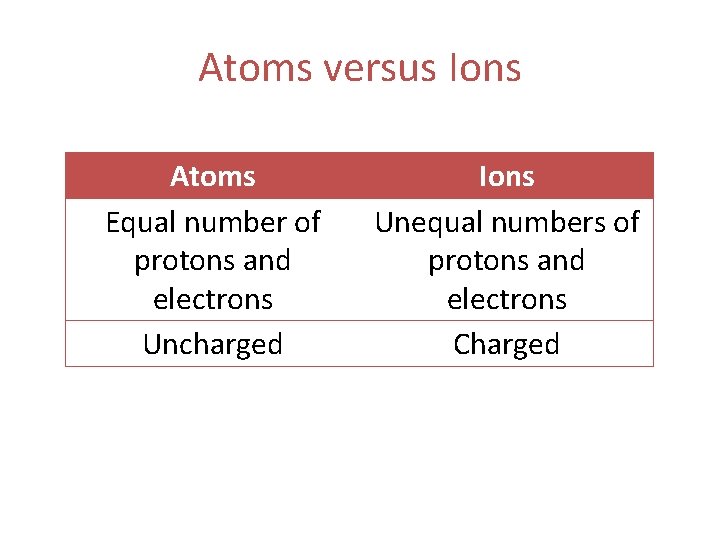
Atoms versus Ions Atoms Equal number of protons and electrons Uncharged Ions Unequal numbers of protons and electrons Charged

Molecules and Compounds Molecule • Forms when two or more atoms share electrons Compound • A type of molecule composed of two or more different atoms

Molecules and Compounds • H 2 O and O 2 are molecules Why is H 2 O a compound? Why is O 2 not a compound?

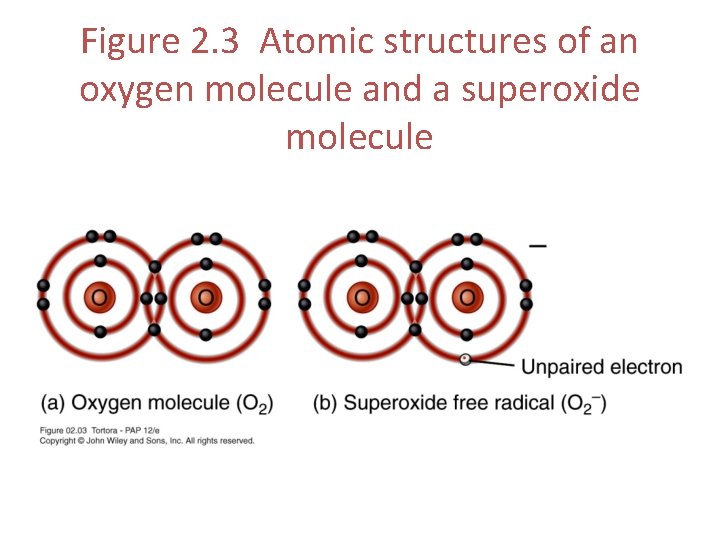
Free Radicals • Free radical – an ion or electrically charged molecule with an unpaired electron in its outermost shell • Are extremely unstable and highly reactive • Become stable by donating or accepting electrons, which may destroy nearby molecules • Like robbers deficient in energy • Snatch energy from stable molecules to satisfy themselves • Antioxidants help inactivate free radicals

Figure 2. 3 Atomic structures of an oxygen molecule and a superoxide molecule

CHEMICAL BONDS

Chemical Bonds • The forces that hold together a molecule’s atoms • Chemical bonds occur between reacting atoms’ electrons

The Role of Electrons • Electrons are found in shells • Each shell has space for a specific number of electrons – First shell has room for two electrons – Second shell has room for eight electrons • Only the outermost valence shell is important in bonding

The Octet Rule • Two atoms will bond with each other if doing so leaves both with eight valence electrons • Can “get to eight” by giving up, accepting, or sharing electrons • Hydrogen has to “get to two”

Ionic Bonds CHEMICAL BONDS

Ionic Bonds • Ionic Bond – the force of attraction that holds a cation and anion together • Formed when one atom donates an electron and another atom accepts it

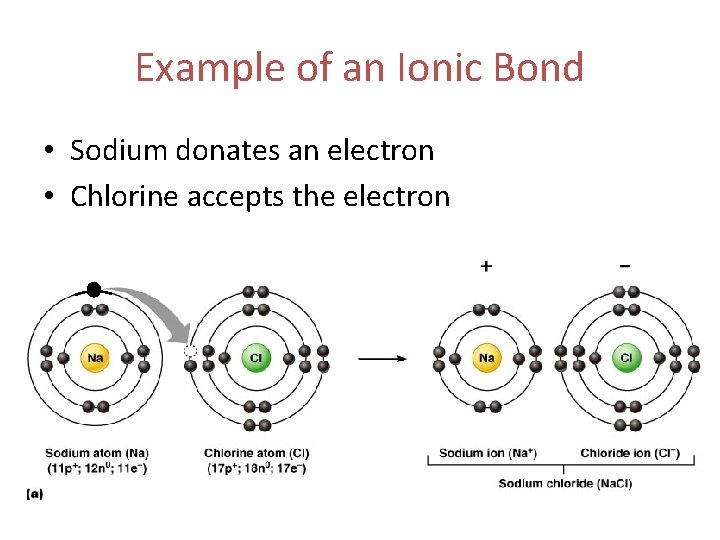
Example of an Ionic Bond • Sodium donates an electron • Chlorine accepts the electron

Ionic Compounds • Most ionic compounds exist as crystals • Ionic compounds dissolve in water to form electrolytes – Positive and negative ions

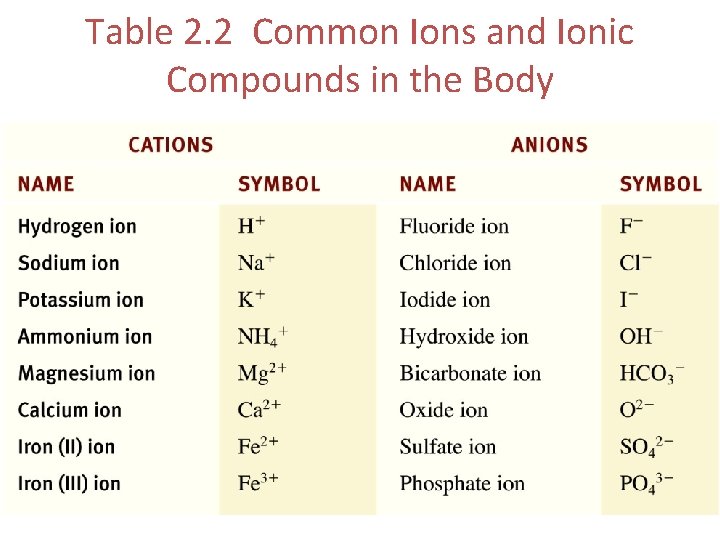
Table 2. 2 Common Ions and Ionic Compounds in the Body

Covalent Bonds CHEMICAL BONDS