Chapter 2 The Chemical Level of Organization Matter











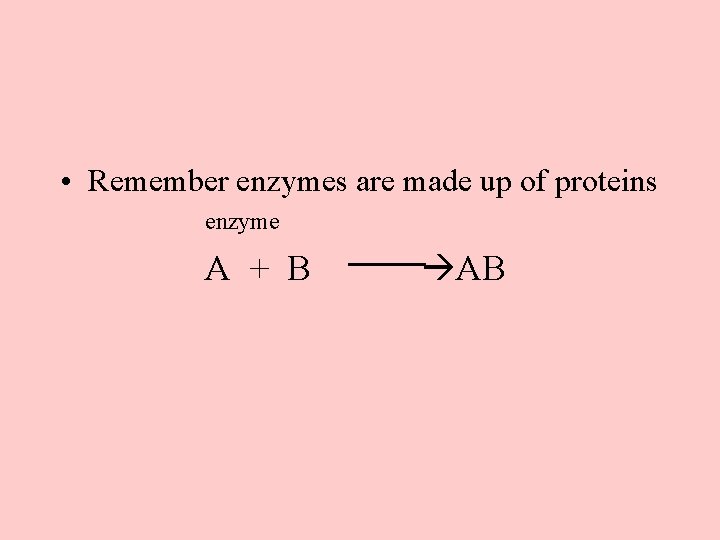






















- Slides: 37

Chapter 2 The Chemical Level of Organization • Matter – Anything that has mass and takes up space • Atom- The smallest particle into which a substance can be broken by ordinary chemical means. • Element – Groups of the same type of atom • Compound – Two or more elements chemically combined ex. H 2 O

MOLECULE- GROUPS OF ATOMS BONDED TOGETHER & ACTING AS A GROUP ORGANELLES- BASIC STRUCTURES WITHIN CELLS – has a specific function CELL- BASIC UNIT OF LIVING ORGANISMS

• TISSUE- TISSUES ARE GROUPS OF CELLS WITH A COMMON FUNCTION. • ORGAN- OFTEN LARGE AND COMPOSED OF SEVERAL DIFFERENT TISSUES • ORGAN SYSTEM- A GROUP OF ORGANS CARRYING OUT A MAJOR BODY PROCESS • ORGANISM- (A GROUP OF ORGAN SYSTEMS IN AN INDIVIDUAL) – any living thing

TYPES OF BONDS (Bonds store energy) • COVALENT BONDS- WHEN ATOMS SHARE ELECTONS (the strongest bond) – e. x. Peptide bonds – between C and N in proteins • IONIC BONDS- ELECTRONS ARE TRANSFERRED FROM ONE ATOM TO ANOTHER AND IONS ARE FORMED • HYDROGEN BONDS- A BOND BETWEEN HYDROGEN AND 2 SMALL ELECTRONEGATIVE ATOMS eg. F, N, O (the weakest bond)

Reactions • Chemical Reactions – the process of breaking down chemical bonds and/or forming new ones (a chemical equations shows this) – To do this an activation energy is needed ( the energy that is needed to get the reaction going) • Exothermic – there is a net release of energy (it feels warm) • Endothermic – there is a net absorption of energy (feels cool)

1. Synthesis – small molecules are combined into larger molecules – always involves the formation of new chemical bonds • Ex. A + B AB Dehydration synthesis (condensation) – the formation of complex molecules by the removal of H 2 O Ex A-B-C-H + HO-D-E A-B-C-D-E + H 20 This is the opposite of hydrolysis

• Anabolism – the synthesis of complex organic compounds from simpler compounds (within the body) * requires energy (anabolic steroids)

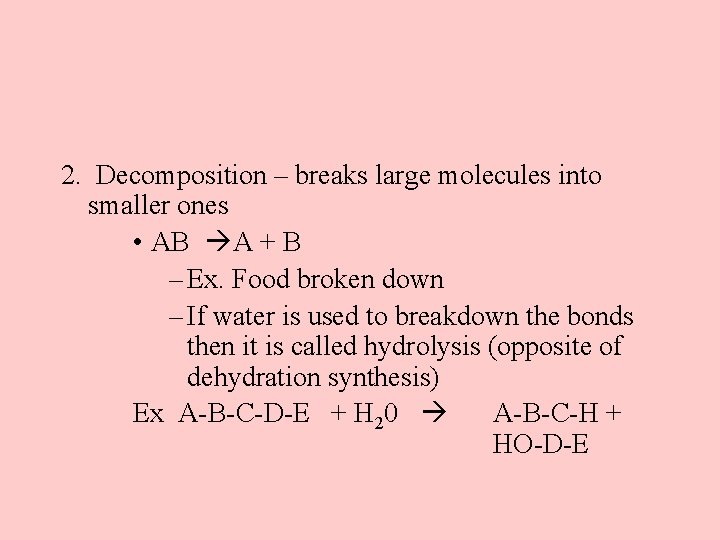
2. Decomposition – breaks large molecules into smaller ones • AB A + B – Ex. Food broken down – If water is used to breakdown the bonds then it is called hydrolysis (opposite of dehydration synthesis) Ex A-B-C-D-E + H 20 A-B-C-H + HO-D-E

• Catabolism – the breakdown of complex organic molecules into simpler components, it releases E that can do work • Ex. Growth, movement, reproduction

3. Exchange (replacement) – Parts of the reacting molecules are moved around AB + CD AD + CB (decomp) (synthesis) May absorb or release energy usually in the form of heat

4. Reversible – A+B ⇌ AB Equalibrium – the rates at which the two equations proceed in balance

Enzymes and Chemical Reactions Activation Energy – the amount of E necessary to start a reaction. Enzymes – protein in nature that catalyzes a specific biochemical reaction. (catalyst – accelerates chemical reactions – they enter into a reaction but do not become part of it). They do this by lowering the activation E …… this makes it “safe” for the cell
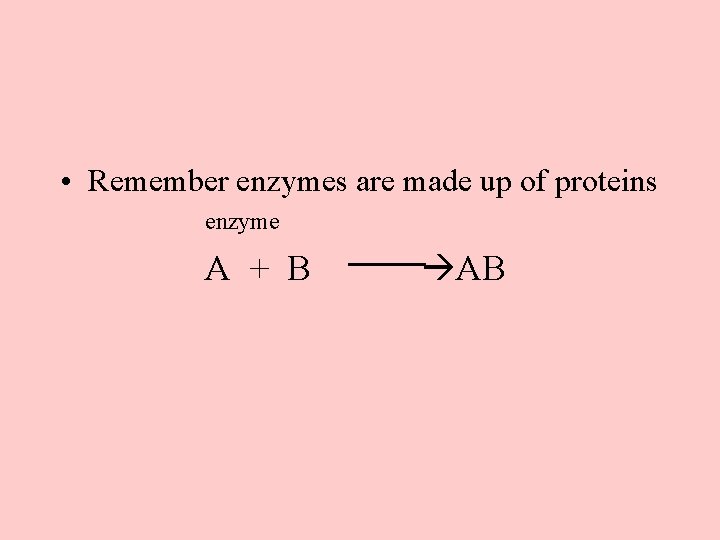
• Remember enzymes are made up of proteins enzyme A + B AB

Inorganic Compounds • Generally do not contain C – Include H 2 O, Na. Cl, CO 2, NO, and CO – H 2 O – the universal solvent • 67% of body is water • Dehydration synthesis and Hydrolysis • Lubrication (reduces friction) • High heat capacity (Carries heat away) • Requires a lot of E to change temp. – So thermoregulation

– Hydrophilic – reacts with water (hydro – water philos – loving) – Hydrophobic – doesn’t react w/water (phobic – fear) Electrolytes – soluble inorganic compounds whose ions conduct electrical current in solution ex Na. Cl Na+ Cl-

Mixtures • Mixtures – two or more substances not chemically combined – therefore they retain their own properties – Solutions – evenly combined • Solvent – does the dissolving ex. Water • Solute – the substance being dissolved ex. Sugar – Suspensions –The particles will spread out and eventually settle – large particles

• Example sand in water (whole blood) -Colloid – a mixture that has medium particles – held in solution by their association with water ex. Liquid jello…. . Mayonnaise

p. H • p. H IS A WAY TO MEASURE THE ACIDITY OR ALKALINITY (BASICITY) OF A SUBSTANCE • ACIDS HAVE A HIGH CONCENTRATION OF (H+) H 3 O + • BASES HAVE A HIGH CONCENTRATION OF OH-

• Acids and bases are measured on a scale called the p. H scale (power of Hydrogen). This scale measures how many hydronium ions (H 3 O+) are present in a solution (now called just H+). The more hydronium (hydrogen) ions the more acidic (lower) the p. H, also the fewer the hydroxide ions (OH-). The more hydroxide ions the higher the p. H (more alkaline – meaning a base) and the fewer the hydronium ions

• THE p. H SCALE RANGES FROM 1 -14 • ON THE p. H SCALE, 7 IS NEUTRAL • A p. H BELOW 7 IS ACIDIC • A p. H ABOVE 7 IS (BASIC) Alkaline • (see drawings on board)

• THE FARTHER A p. H IS FROM 7, THE STRONGER THE SUBSTANCE IS • BUFFERS ARE SUBSTANCES THAT HELP STABILIZE p. H IN THE BODY • BICARBONATE IS THE MOST Important buffer (how CO 2 mostly appears in the blood – keeps the blood from becoming to acidic or alkaline)

ORGANIC MOLELCULES • ALL ORGANIC MOLECULES CONTAIN CARBON AND ARE FOUND IN LIVING ORGANISMS • THERE ARE 4 MAIN GROUPS – CARBOHYDRATES – PROTEINS – LIPIDS – NUCLEIC ACIDS

CARBOHYDRATES • ARE SUGARS, STARCHES AND RELATED COMPOUNDS • THEIR MOST IMPORTANT FUNCTION IS TO SERVE AS A FUEL SOURCE FOR CELLS • THEY ARE ALSO COMPONENTS OF CELL MEMBRANES AND NUCLEIC ACIDS

• CARBOHYDRATES ARE CHAINS OF CARBON ATOMS THAT ARE BONDED TO HYDROXYL (OH) GROUPS AND HYDROGEN ATOMS (H) • CARBOHYDRATES ARE MADE OF C, H & O • C, H & O ARE IN A 1: 2: 1 RATIO • THERE ARE 3 MAIN GROUPS OF CARBOHYDRATES

3 GROUPS OF CARBOHYDRATES • MONOSACCHARIDES- SIMPLE SUGARS eg. GLUCOSE, FRUCTOSE, GALACTOSE • DISACCHARIDES- 2 SIMPLE SUGARS eg, LACTOSE, SUCROSE, MALTOSE • POLYSACCHARIDES- MANY SIMPLE SUGARS eg. STARCH, CELLULOSE, GLYCOGEN • SUGARS END IN - OSE

MONOSACCHARIDES • GLUCOSE, FRUCTOSE & GALACTOSE ARE ISOMERS. • ALL HAVE THE CHEMICAL FORMULA C 6 H 12 O 6 • ISOMERS- ARE MOLECULES WITH THE SAME CHEMICAL FORMULA, BUT A DIFFERENT ARRANGEMENT OF ATOMS.

MONOSACCHARIDES: GLUCOSE • GLUCOSE- BLOOD SUGAR – cells use this for energy, which is metabolized during cellular respiration. Our major source is from fruits/plants. Plants produce during photosynthesis. • NORMAL LEVEL IS 80 -120 mg/100 ml. • BELOW THIS LEVEL CAN RESULT IN HYPOGLYCEMIA & ABOVE THIS LEVEL CAN RESULT IN DIABETES MELLITUS MEANS “SWEET URINE”

FRUCTOSE FRUIT SUGAR THE SWEETEST OF ALL THE SUGARS

GALACTOSE • NOT FOUND FREE IN NATURE, ONLY IN MILK • WE CANNOT USE GALACTOSE BUT MUST CHANGE IT TO GLUCOSE

ENZYME A (BIRTH)** GALACTOSE GLUCOSE ENZYME B (ONE YEAR) **ENZYME A IS MISSING IN CHILDREN WITH GALACTOSEMIA.

THIS DISORDER IS CALLED GALACTOSEMIA WHICH MEANS GALACTOSE IN THE BLOOD.

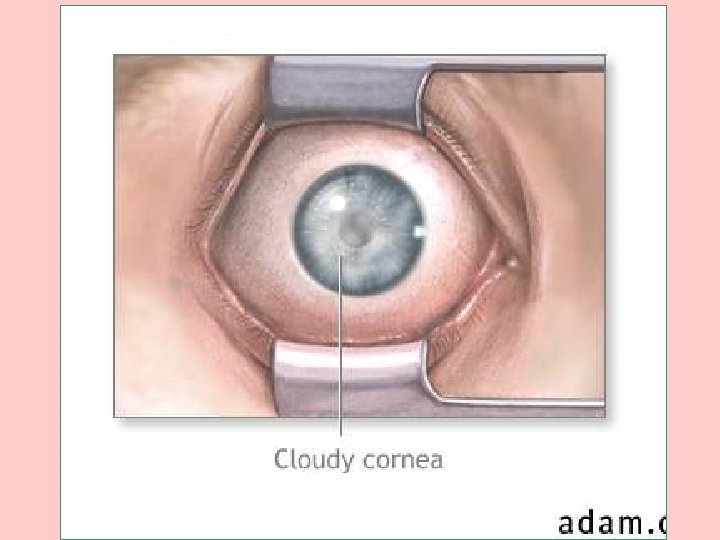
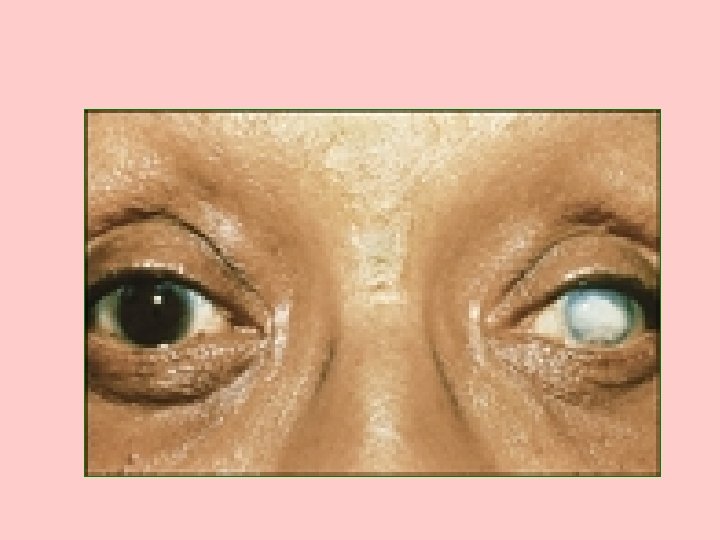
Sx OF GALACTOSEMIA • CLOUDING OF THE CORNEA AT ABOUT 4 WEEKS • ENLARGED LIVER AND SPLEEN AT ABOUT 4 MONTHS • CATARACTS AT ABOUT 6 MONTHS • MENTAL RETARDATION AT 12 MONTHS • ALL ARE REVERSIBLE, EXCEPT MENTAL RETARDATION



HOW WOULD YOU TREAT GALACTOSEMIA? FEED THE BABY A GALACTOSE FREE FORMULA, NO COW’S OR MOTHER’S MILK. BOTH CONTAIN THIS SUGAR.

DISACCHARIDES – when two monosaccharides bond in a dehydration synthesis reaction H+ and OH- are removed and form water LACTOSE: MILK SUGAR GLUCOSE + GALACTOSE SUCROSE: TABLE SUGAR GLUCOSE + FRUCTOSE MALTOSE: MALT SUGAR BREWING INDUSTRY GLUCOSE + GLUCOSE

POLYSACCHARIDES – three or more monosaccharides STARCH: STORAGE FORM OF GLUCOSE IN PLANTS GLYCOGEN: STORAGE FORM OF GLUCOSE IN ANIMALS CELLULOSE: CELL WALLS OF PLANTS FOR STRENGTH ALL 3 ARE LONG CHAINS OF GLUCOSE