Euromet 2006 Influence of impurities on the melting















- Slides: 52

Euromet 2006 Influence of impurities on the melting temperature of Aluminum Pr. B. Legendre & Dr S. Fries EA 401

Impurities • • B, C, Cr, Cu, F, Fe, Mg, Mn, N, Ni, O, Pt S, Sc, Si, Ti, V, Zn. Al – X Optimized Al – Y Non optimized (or bad optimized), but the phase diagram is known • Al – F no data

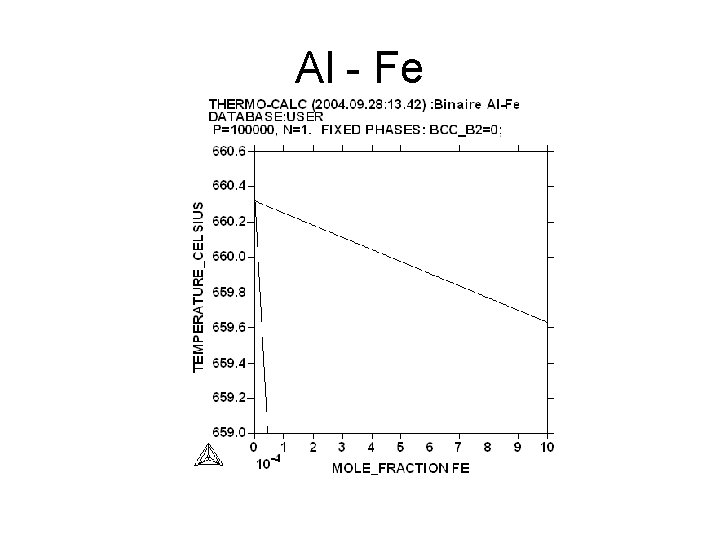
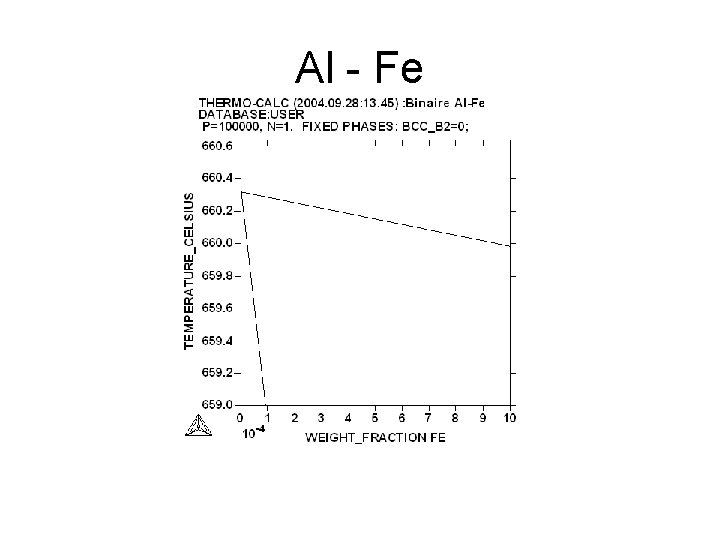
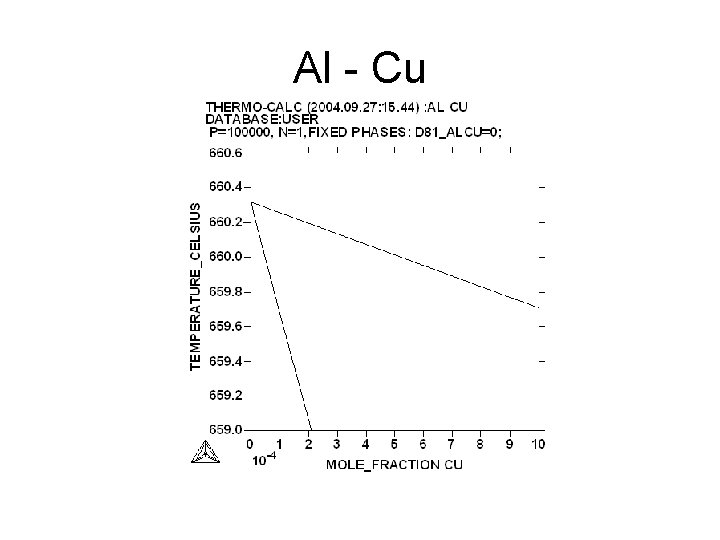
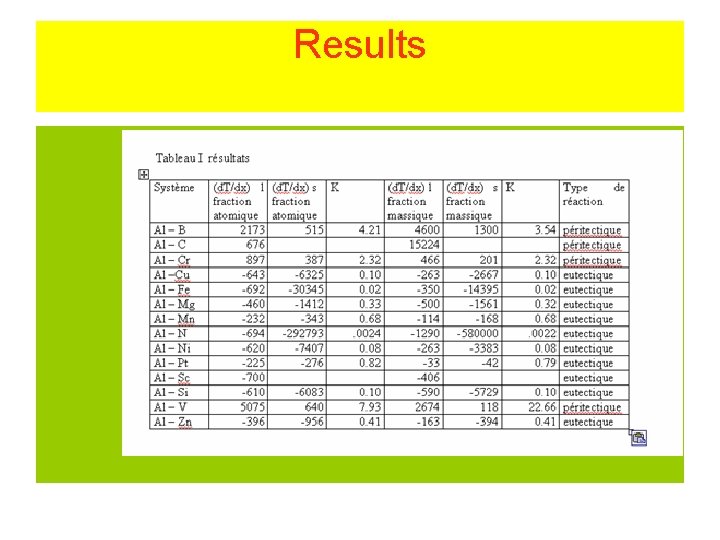
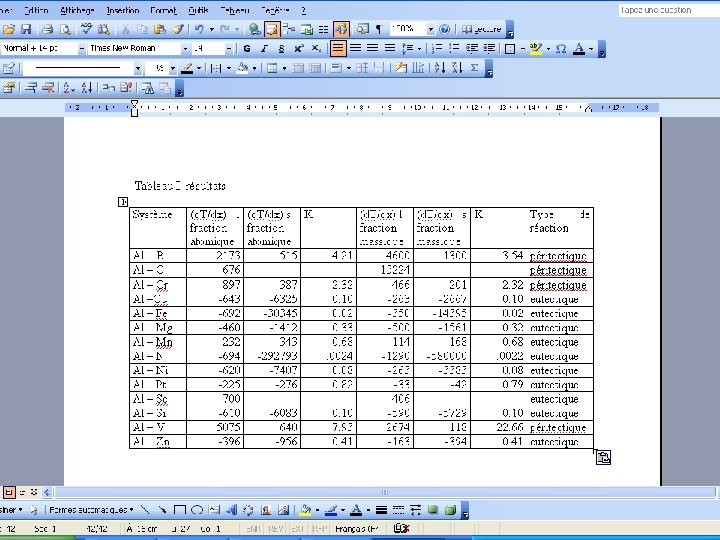
Aim • Measurement of the slopes of the liquidus and of the solidus : = (d. T/dx) • l : for the liquidus s : for the solidus • The slope may be expressed in atomic fraction or in weight fraction. • Determination of K : • K = l/ s • For Al Tfus= 660. 323°C

Phase diagram • For this subject we are interested only by a very small region in temperature and composition of binaries Al-X, but it is absolutely necessary to have a perfect knowledge of the full diagram. And to know the Gibbs function versus of T and x for each phase at a fixed pressure.

Phase diagram : Calphad Method • A phase diagram is the graphical expression of phase equilibria, for each phase in a binary system G = f(xi, T, P) • The limits of a two phase field are given by the common tangent of the G =f(xi)PT curves. • For a eutectic: • The three curves G=f(xi)p, T, have the same tangent. •

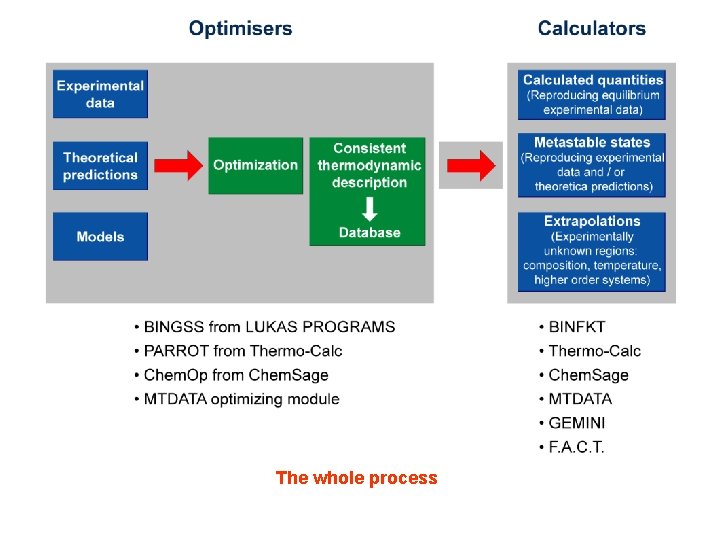
The whole process

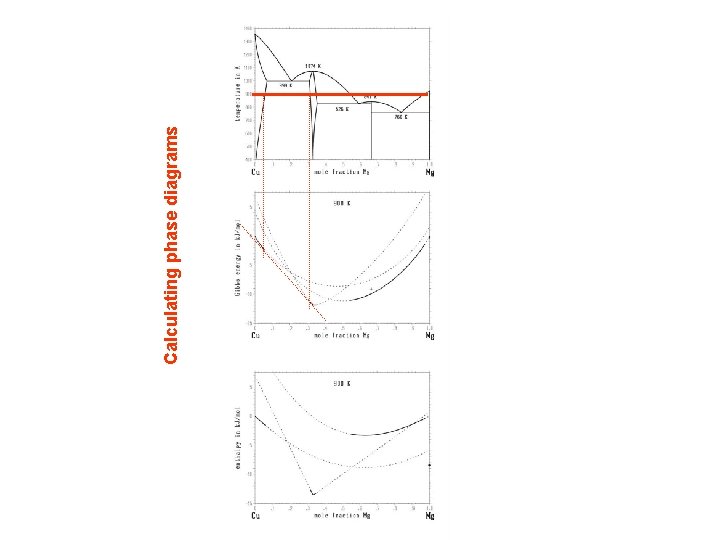
Calculating phase diagrams

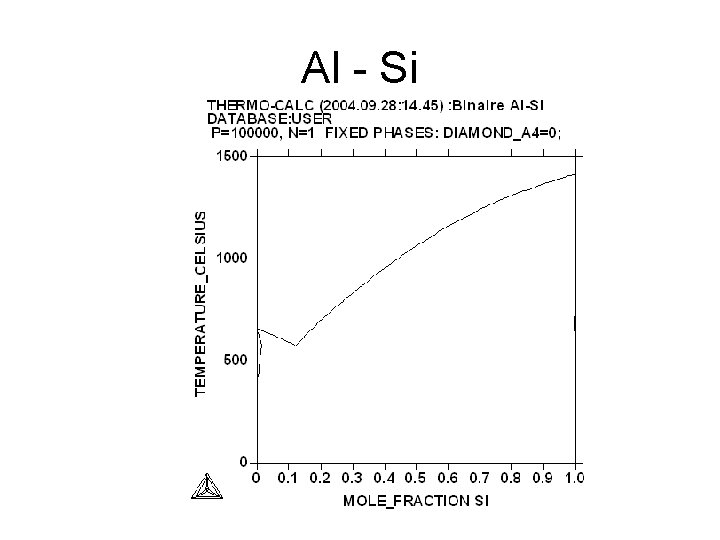
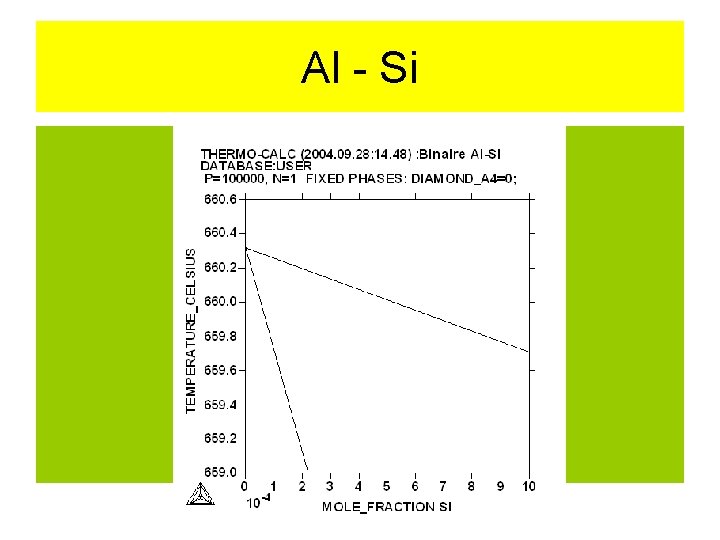
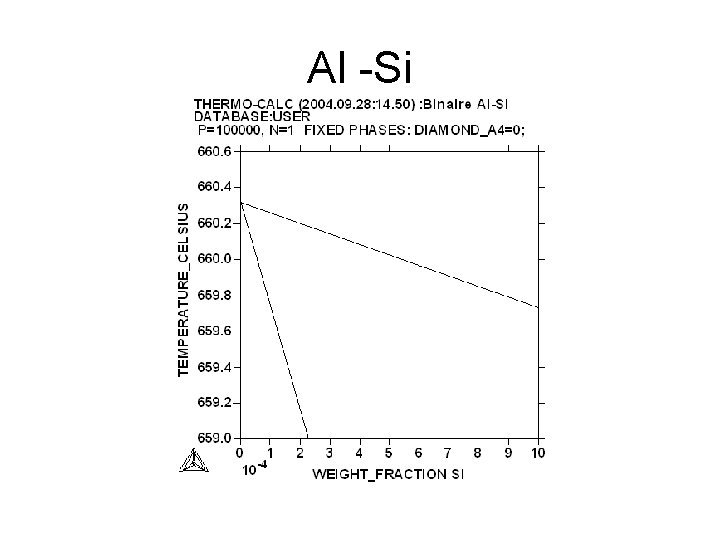
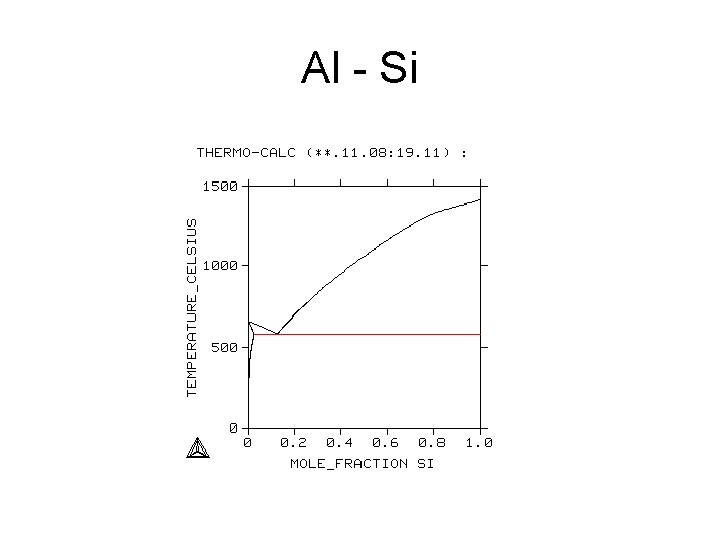
Al - Si

Al - Si

Al -Si

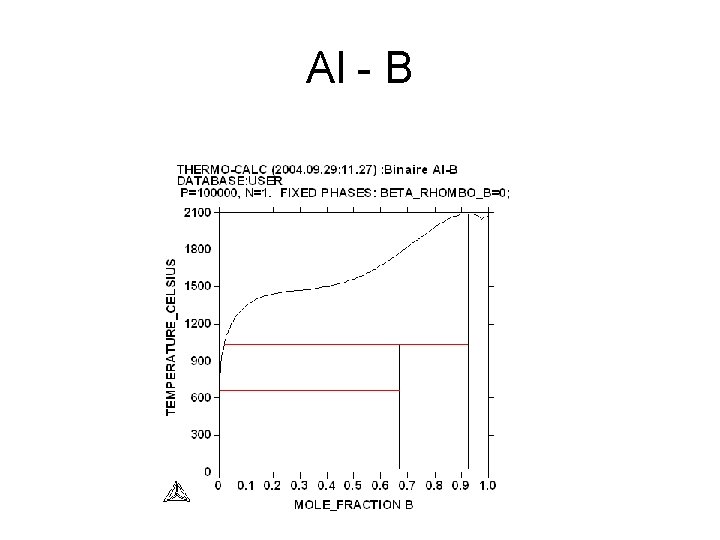
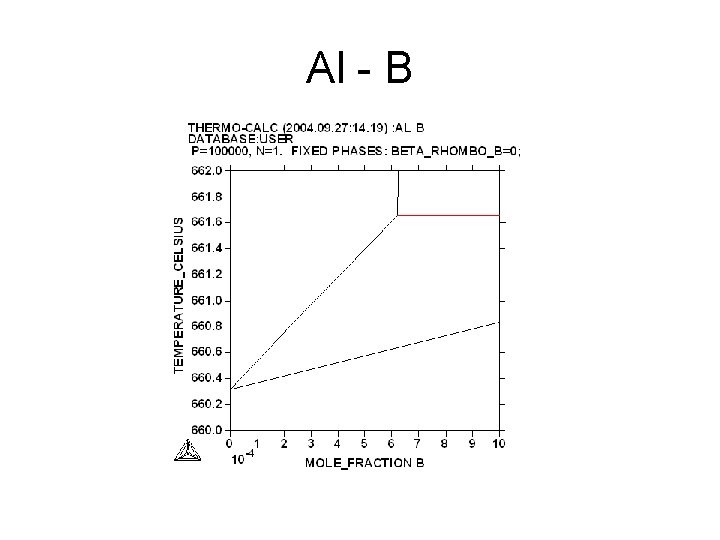
Al - B

Al - B

Al - B

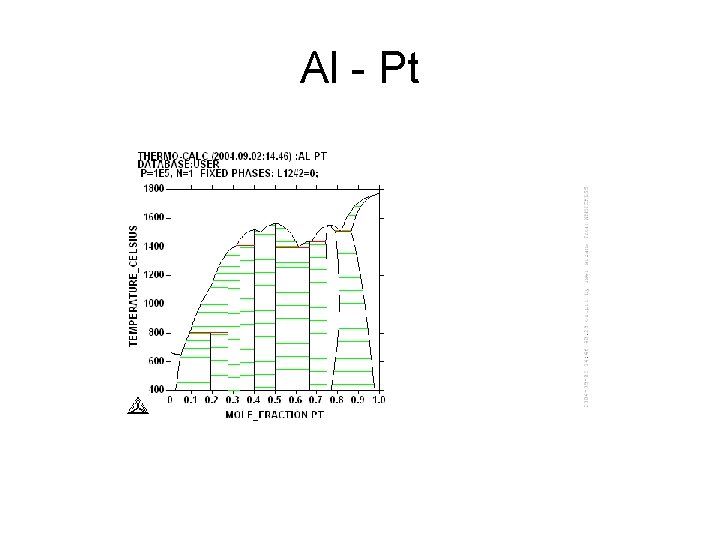
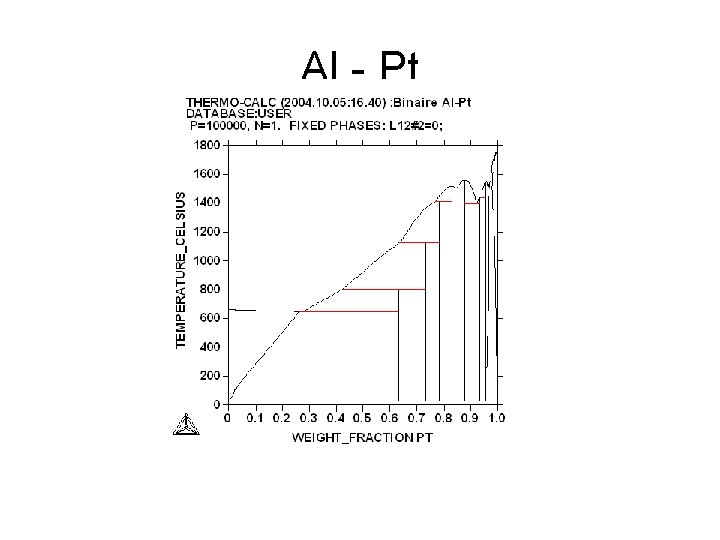
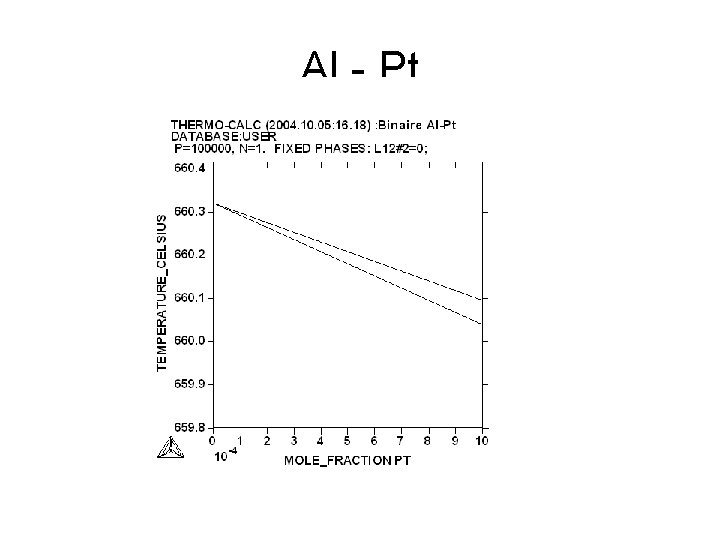
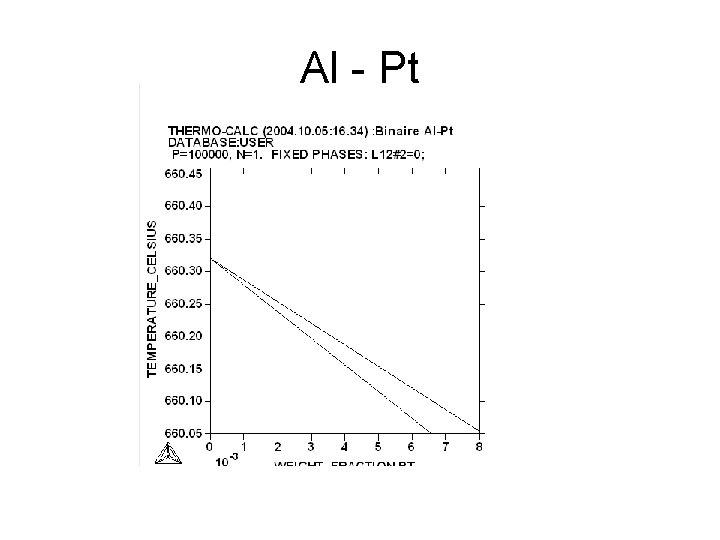
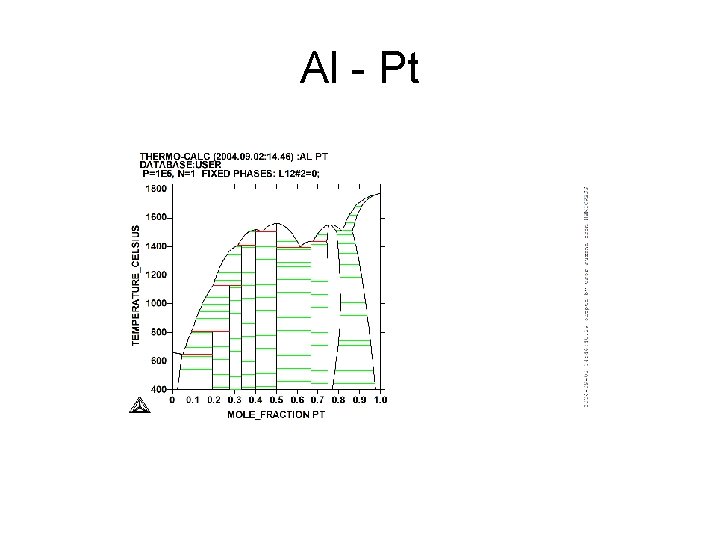
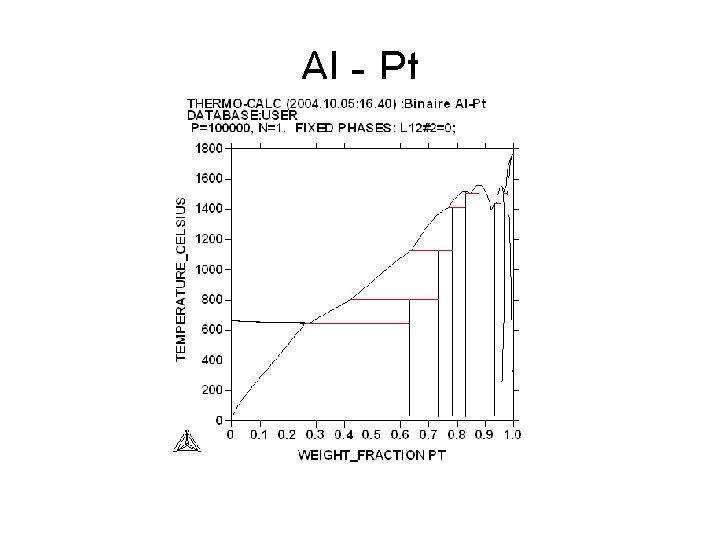
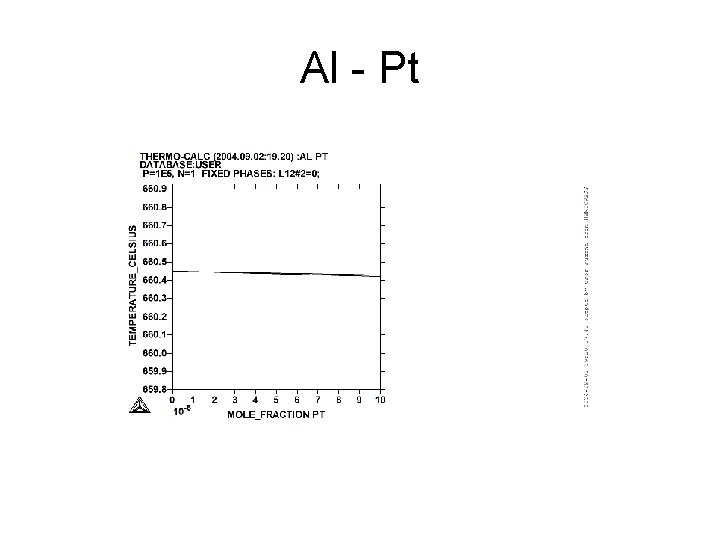
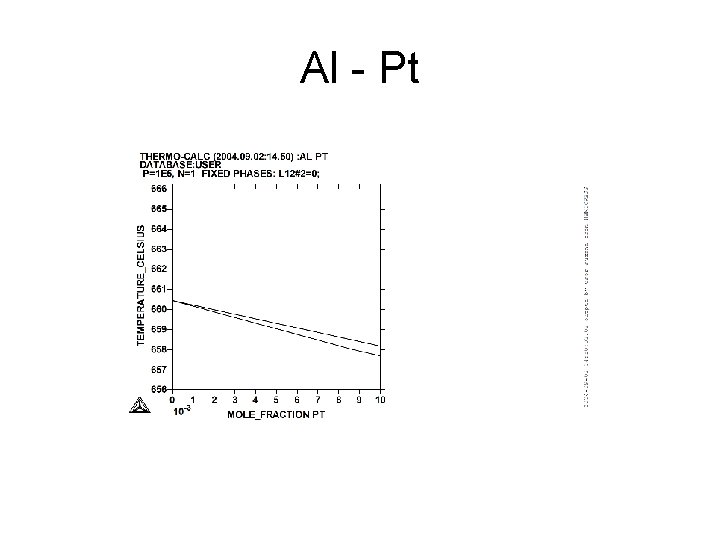
Al - Pt

Al - Pt

Al - Pt

Al - Pt

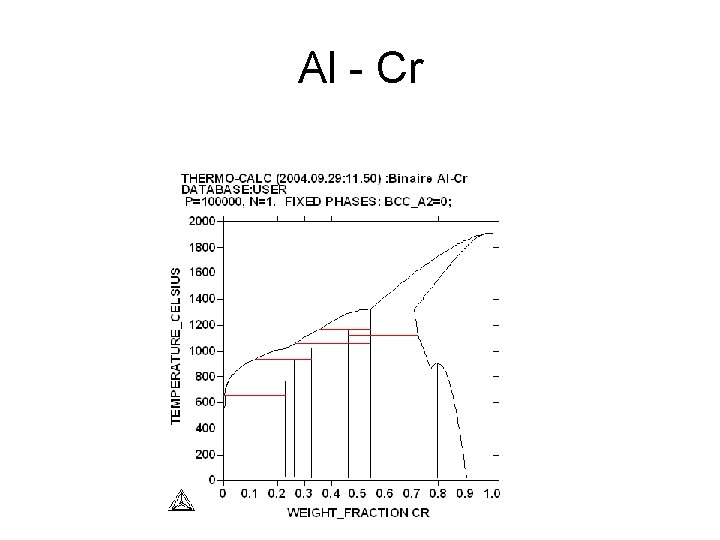
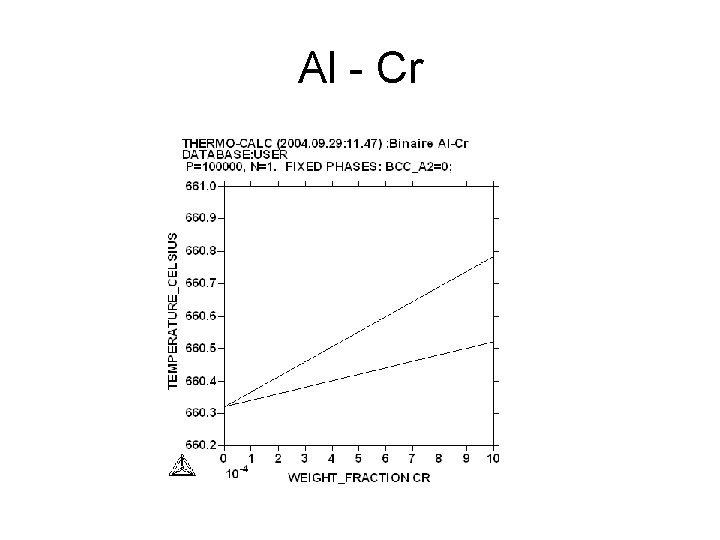
Al - Cr

Al - Cr

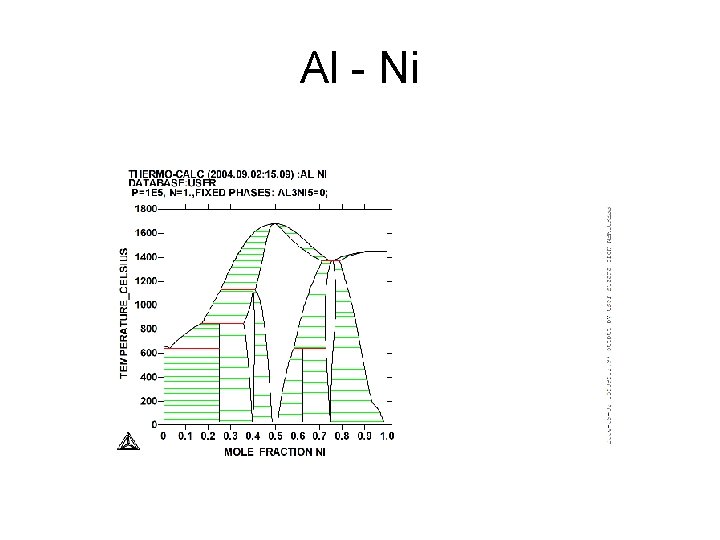
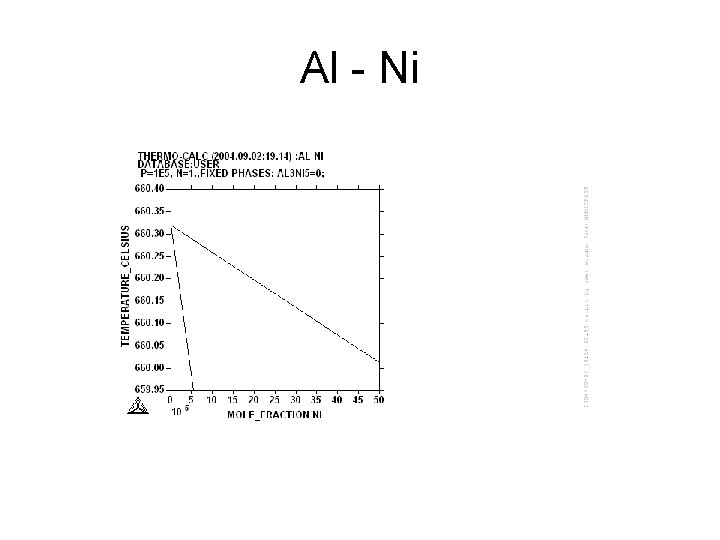
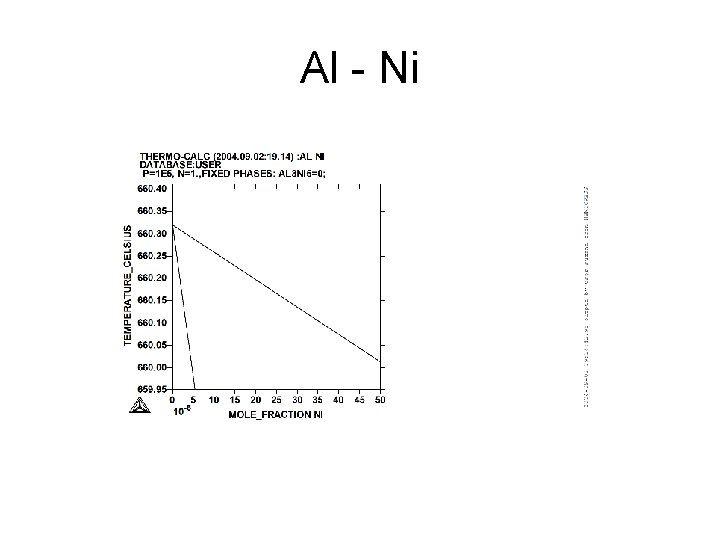
Al - Ni

Al - Ni

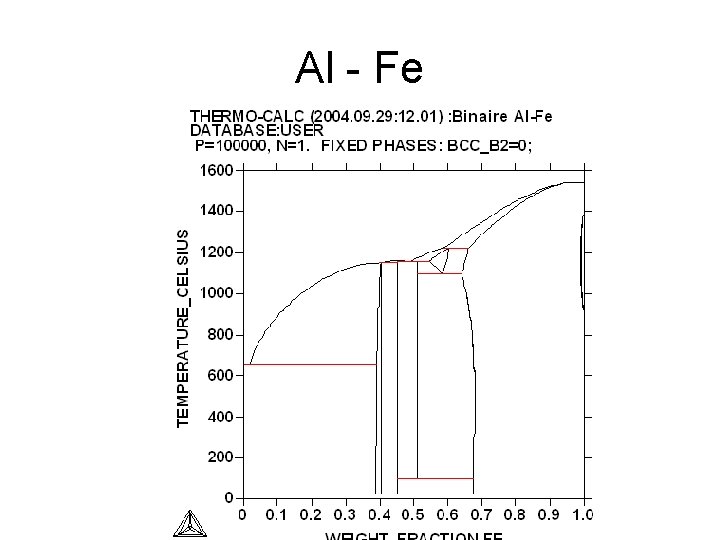
Al - Fe

Al - Fe

Al - Fe

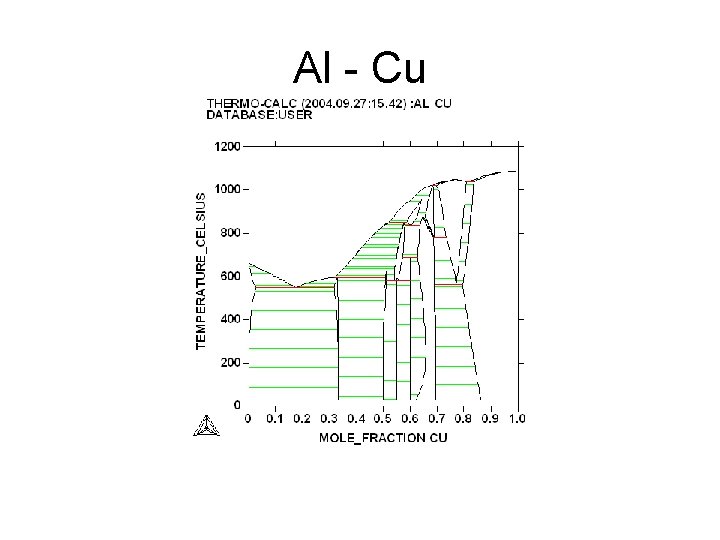
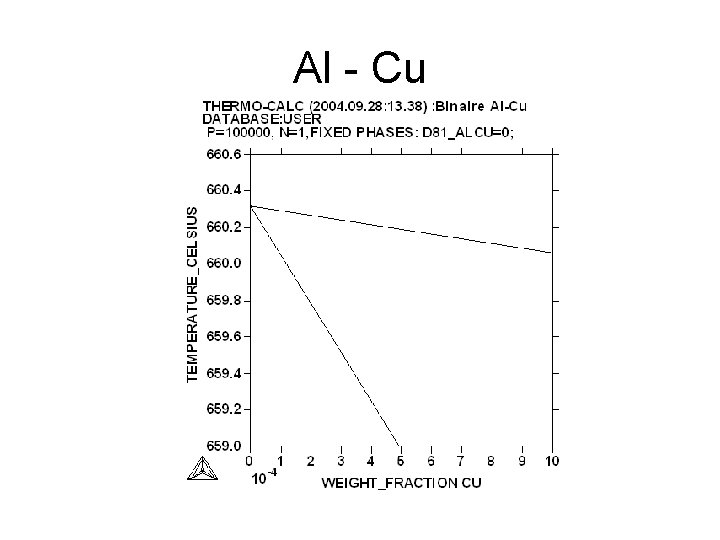
Al - Cu

Al - Cu

Al - Cu

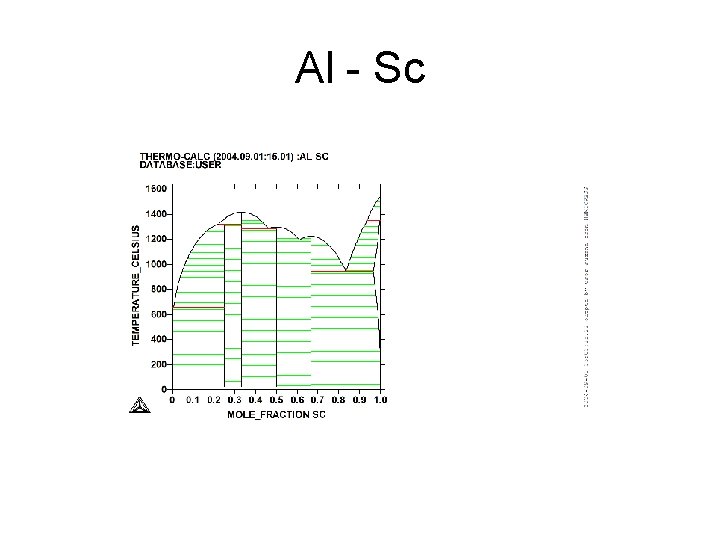
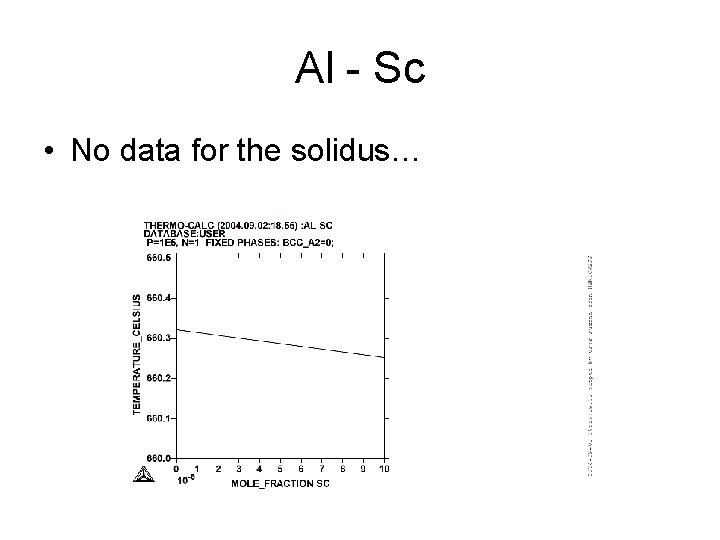
Al - Sc

Al - Sc • No data for the solidus…

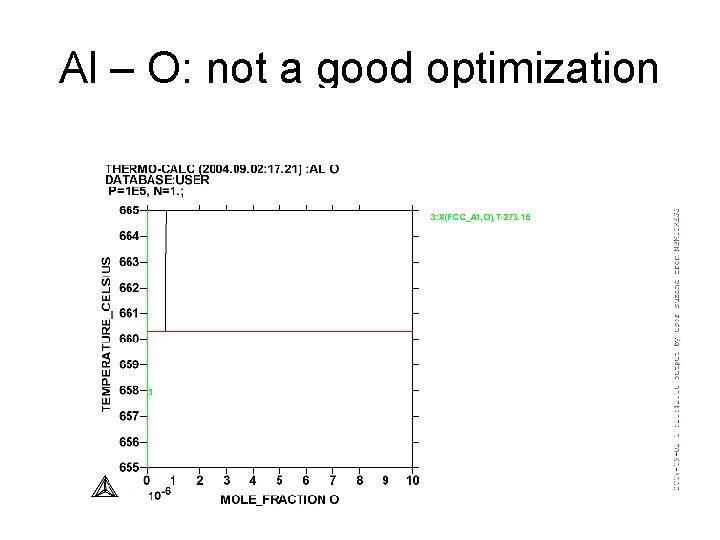
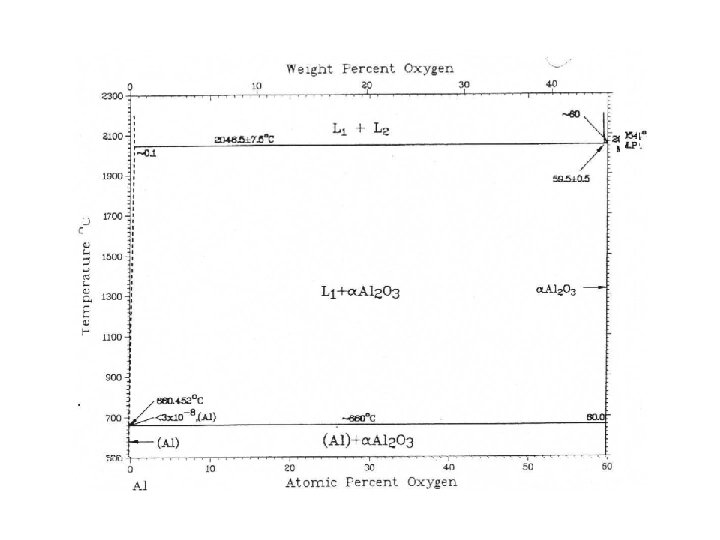
Al – O: not a good optimization


Al - O • Why it is not possible to have a good optimization of the Al-O binary? • The oxygen forms with Al an oxide which is a thin film fixed on the surface of Al and then it is a kind of protection (aluminum pans are used on the gas in a kitchen…) after the fixation of a first layer the oxygen cannot go deeper in Al. There is no homogeneity and no equilibrium.

Al - O • L Al + Al 2 O 3 T =~ 660°C • Van’t Hoff law • dln(a. Al)/d. T = H/RT 2 • a. Al ~ XAl • H = fus. H(Al) = 10711. 04 J/mol • T = 0. 452 K XAl=. 9993316 • d. T/d. X = 676

Results

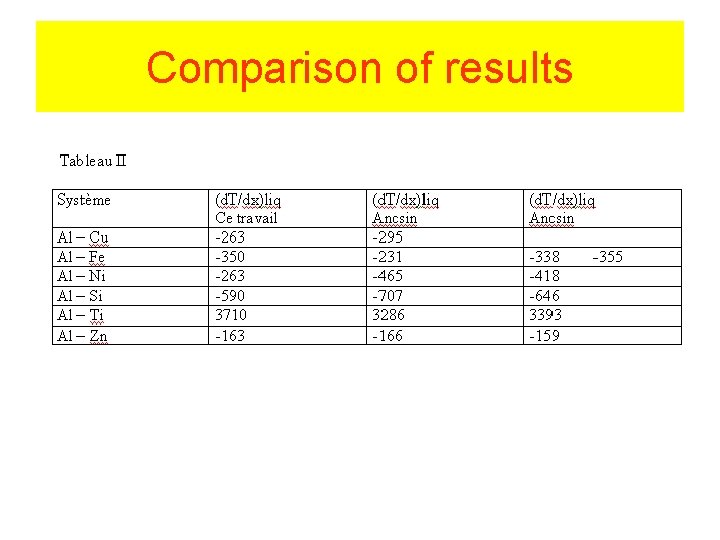
Comparison of results

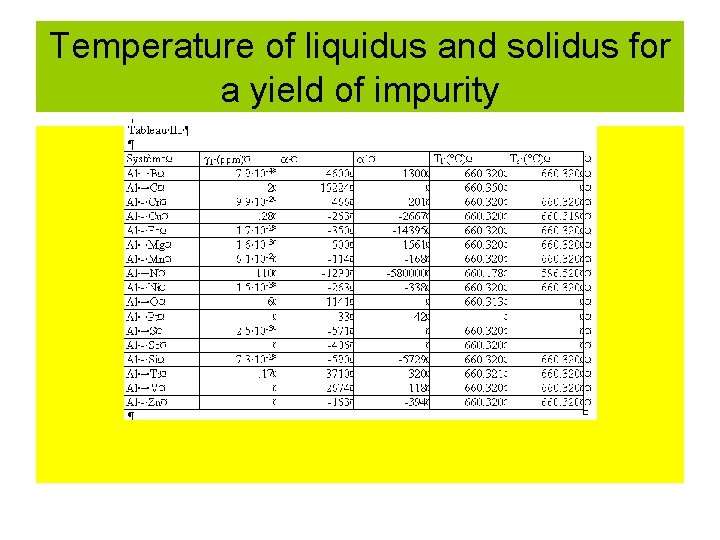
Variation of the liquidus temperature versus of the yield of impurities • • Tl = Tfus + i Tl = temperature of the liquidus Tfus temperature of fusion of pure Al = d. T/dx x : weight fraction i : yield of impurities in ppm*10 -6 Example : for Al-Cr i = 9. 9*10 -2 ppm = 466 Tl = 660. 32°C

Temperature of liquidus and solidus for a yield of impurity

Conclusions • It has been possible to determine the influence of impurities in Al for nearly all the selected elements. • For O and S just an estimation has been possible. • The most important modifications occurs with O and N • It is possible to perform a calculation with two impurities.

Acknowledgement • This work has been financially supported by LNE and BNM • We are very grateful to these organisms for their contribution. •





Al - Pt

Al - Pt

Al - Pt

Al - Ni


Al - Pt

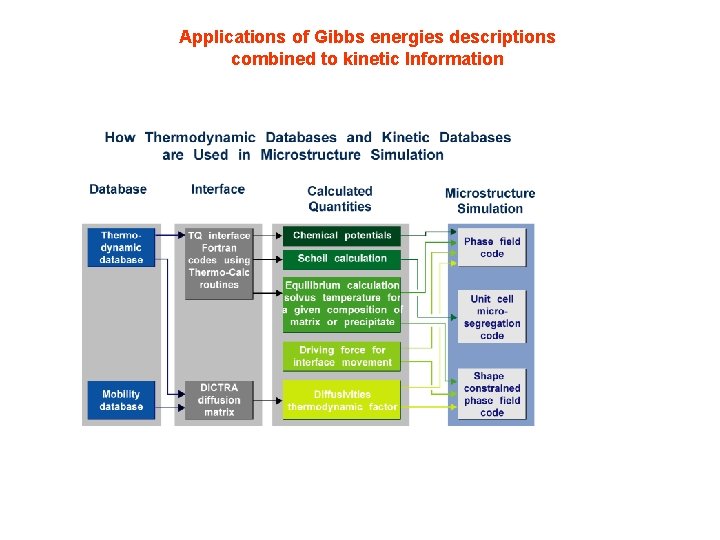
Applications of Gibbs energies descriptions combined to kinetic Information

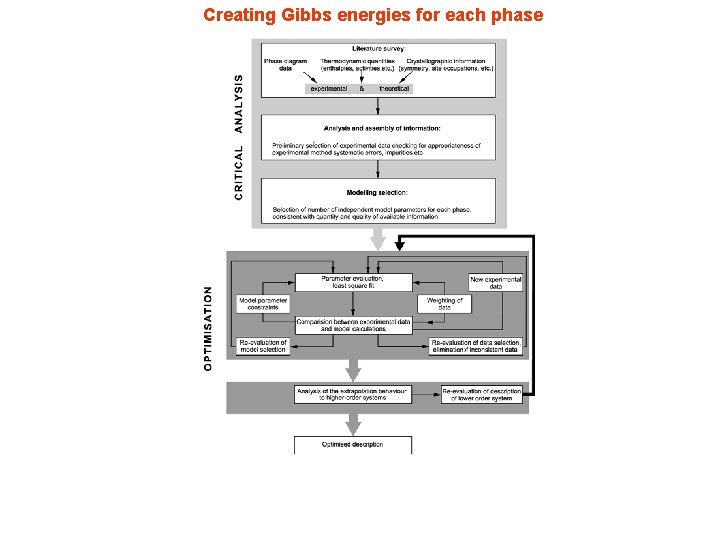
Creating Gibbs energies for each phase

Al - Si